Draw The Product Of The Hydration Of 2-butene.
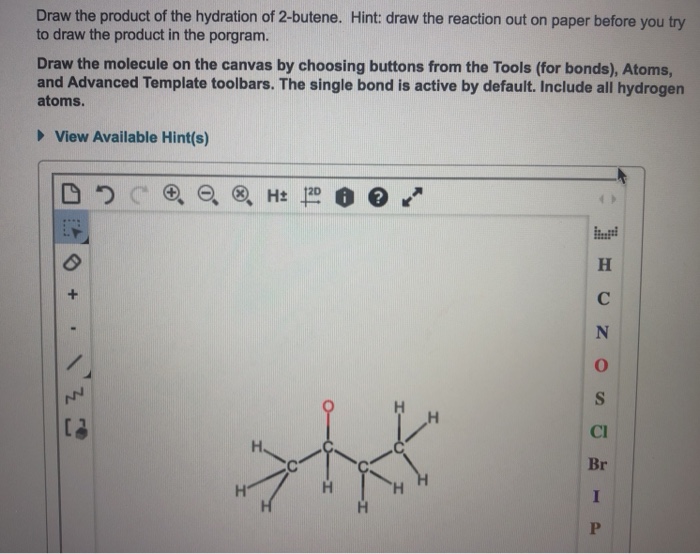
Hey there, curious minds! Ever found yourself staring at a jumble of letters like "2-butene" and wondered, "What in the world is that, and why should I care?" Well, buckle up, buttercup, because we're about to embark on a little adventure into the land of chemistry, and trust me, it's going to be way more fun than you think. We're going to talk about what happens when you "hydrate" something called 2-butene. Think of it like giving it a nice, refreshing drink of water. Simple, right?
Now, "2-butene" might sound like it belongs in a sci-fi movie, but it's actually a pretty common little molecule. Imagine a tiny Lego brick with four carbon atoms all lined up, holding hands with some hydrogen atoms. 2-butene is one specific arrangement of those bricks. The "2" just tells us where the special connection (a double bond, for the chemists among us) is happening between the carbon atoms. It’s like saying, "Hey, the party's happening at the second carbon!"
So, what is "hydration"? In our everyday world, hydration is all about getting enough water, right? For our bodies, it means staying energized and feeling good. For plants, it means staying green and vibrant. For 2-butene, hydration is its own kind of refreshment. It's basically adding a water molecule to it. Imagine you’re at a desert oasis and you finally get a big gulp of cool, clear water. That’s kind of what hydration is for 2-butene – a welcome addition of an H₂O molecule.
Now, let's get to the drawing board, or rather, the digital drawing board! When we talk about "drawing the product," we're not talking about a masterpiece with a frame and everything. We're talking about showing what this 2-butene molecule looks like after it's had its drink of water. It's like taking a "before" and "after" photo of your favorite plant after a good watering.
Let's visualize this. 2-butene, in its original form, has this double bond. Think of it like a handshake between two carbon atoms that's a little extra strong, a double handshake! This double bond is where all the action happens. When water comes along, it’s like a friendly mediator. It breaks that double handshake and helps attach pieces of the water molecule to the carbon atoms.
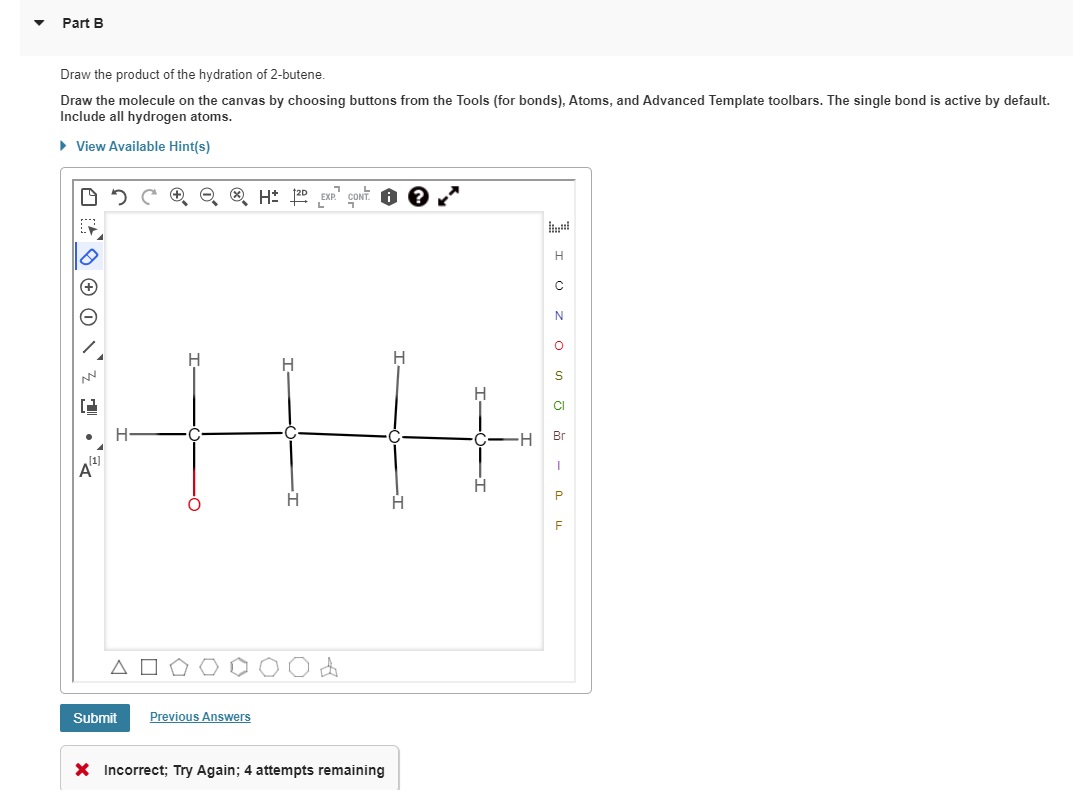
The water molecule (H₂O) is made of one oxygen atom and two hydrogen atoms. When it joins 2-butene, the water molecule splits up. One hydrogen atom often attaches to one of the carbons involved in the original double bond, and the oxygen, along with its remaining hydrogen, attaches to the other carbon. It’s like a polite exchange, where everyone gets a new friend.
The Big Reveal: What Do We Get?
So, what’s the end result? What does this "hydrated" 2-butene look like? When you add water to 2-butene, you get a new molecule called an alcohol. Specifically, you'll get one of two possible alcohols. Think of it like a choose-your-own-adventure story where the ending depends on which path you take. The exact location where the oxygen and hydrogen from the water attach to the carbon chain can vary slightly.
The most common outcome, or the one that usually happens more easily, is where the oxygen atom ends up attached to the middle carbon atom of the original four-carbon chain. This creates a molecule called butan-2-ol. Imagine your four Lego bricks, and the water decided to park its oxygen-friend on the second brick from either end. It’s a very sensible place to settle down!
The other, less common, possibility is that the oxygen ends up on one of the outer carbon atoms. This would give you butan-1-ol. It’s like the water decided to take a little detour and set up camp at the very end of the line. Both are perfectly valid outcomes, just like having two different flavors of ice cream – you can’t go wrong!
So, when you draw the product of the hydration of 2-butene, you're essentially drawing these alcohol molecules. You'll see a chain of four carbon atoms, and attached to one of those carbons will be an -OH group. That -OH group is the signature of an alcohol, the little stamp that says, "Yep, this is an alcohol!"

Why Should You Even Bother?
Okay, okay, I hear you. "But why should I care about drawing an alcohol from 2-butene? Does it impact my life of Netflix binges and pizza nights?" The answer is a surprising yes! These seemingly tiny chemical transformations are the building blocks for so much of what we use and experience every single day.
Alcohols, like the ones we get from hydrating 2-butene, are incredibly useful. Think about solvents. You know those things that help dissolve other things? Alcohols are fantastic at that. They're used in cleaning products, paints, and even in some of your favorite perfumes to help the scent spread evenly. Imagine trying to mix oil and water – it’s a mess! But alcohols can often bridge that gap, helping things blend together smoothly, just like a good mediator at a party.
And it's not just about dissolving. Alcohols are also crucial ingredients in the pharmaceutical industry. Many medicines, from your basic painkillers to more complex treatments, rely on alcohol-based compounds. The journey of a drug from a lab to your medicine cabinet often involves steps where molecules like the ones we're talking about are formed or used.

Furthermore, these types of reactions are fundamental to creating plastics and other materials. The world of polymers, the long chains that make up plastics, is deeply intertwined with the chemistry of simple molecules like butenes and their derivatives. So, that water bottle you're holding, the car you drive, even your smartphone – they all have a lineage that can be traced back to these fundamental chemical processes.
It’s like learning your ABCs. You might not think knowing your "A" is going to change the world, but it’s the first step to reading a great novel, sending a heartfelt letter, or even writing your own amazing story. Understanding how molecules like 2-butene react with water helps chemists design new materials, create better medicines, and develop more sustainable processes. It’s about building a better future, one molecule at a time.
So, the next time you hear about "hydration of 2-butene" or see a chemical drawing, don't just glaze over. Think of it as a tiny, fascinating step in a much larger, incredibly important story. It's the story of how we create the world around us, a world that’s made of these tiny, interactive building blocks. And knowing a little bit about it can make you appreciate the complexity and ingenuity that goes into everything from your morning coffee to the advanced technology you use every day. Pretty cool, right?
