Draw The Product Of The Complete Hydrogenation Of Ethyne
Ever heard of ethyne? It’s a super simple molecule, like a tiny building block in the world of chemistry. Think of it as the most basic of the alkynes, with a really energetic triple bond holding it all together. It’s a bit of a rebel, always ready for a chemical adventure!
Now, imagine we decide to give this little ethyne molecule a makeover. We're going to do something called complete hydrogenation. Sounds fancy, right? But it’s actually pretty straightforward. We're basically adding hydrogen atoms to it, and we’re going to do it until it can't take any more.
Think of hydrogenation like giving our ethyne friend a hug. A very, very full hug, where it grabs onto as many hydrogen atoms as it can hold. It’s like going from a packed party to a really, really comfortable, fully occupied couch.
What happens when we do this complete hydrogenation to ethyne? That's where the magic really starts. We take away that feisty triple bond, which is the source of ethyne's extra reactivity. It's like calming down a very excited puppy and giving it a cozy bed.
Instead of that wild triple bond, our molecule ends up with a much more relaxed structure. It becomes a molecule where all the connections are single. No more triple threat! It’s like going from a juggling act with three balls to a simple toss with one.
And the product of this transformation? It’s a molecule called ethane. Yup, just good old ethane. It’s like the chill cousin who shows up to the party after all the wild dancing is done, and just wants to relax. Ethane is pretty stable, not as eager to react as ethyne was.
So, what makes drawing this whole process so entertaining? It’s the journey! We start with something that looks a bit like a dash with two dots on either side, representing that triple bond. It's a visual representation of pure energy, ready to burst!
Then, we introduce our hydrogen friends. They’re like little happy circles, eager to join the party. We show them approaching our ethyne molecule, and they start to break apart that triple bond.

Imagine the triple bond as three tightly wound springs. Hydrogen atoms come along and gently uncoil them, one by one. It’s a gradual process, a visual unravelling of tension.
With complete hydrogenation, we’re not just uncoiling one spring, or even two. We’re uncoiling all of them! We add enough hydrogen to make sure every available spot is filled.
The final drawing of ethane looks so different! It’s a simple chain, usually depicted as a zig-zag line. Each point in the zig-zag is a carbon atom, and the lines connecting them are single bonds. And all around, you see those little hydrogen circles attached.
It’s a visual story of transformation. From a compact, high-energy molecule to a more spread-out, relaxed one. The triple bond, once the star of the show, is completely gone. It’s like the difference between a firework display and a calm starry night.
The beauty of it is in its simplicity. You don't need to be a chemistry whiz to appreciate the visual change. It’s like watching a caterpillar transform into a butterfly. The starting point is so different from the end result, but the process makes it all understandable.
The “complete hydrogenation of ethyne” is a fun little drama in the molecular world. It’s about taking something super reactive and making it calm and steady. It’s a chemical journey from excitement to tranquility.
When you see the drawings, you can really visualize the change. On one side, you have ethyne, looking tense and ready for action. On the other, you have ethane, looking peaceful and content.
What makes it special is that it’s a fundamental reaction. It shows us how molecules can be changed and how we can control those changes. It’s like learning to bake – you start with simple ingredients and end up with something delicious and different.
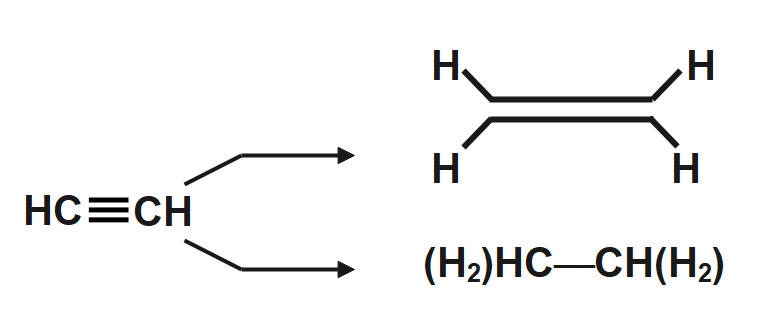
Think about the triple bond in ethyne. It’s represented by three lines between the carbon atoms. That’s a lot of connection, a lot of shared electrons. It’s a really strong, but also very reactive, bond.
Hydrogenation is like adding more people to a dance floor that’s already packed. But instead of getting more crowded, the dancers actually spread out and become more relaxed. In our case, the “dancers” are the hydrogen atoms.

With complete hydrogenation, we add two molecules of hydrogen gas (H₂). That’s a total of four hydrogen atoms being added to the ethyne. They all jump in to fill the spots created by breaking those triple bonds.
So, ethyne (C₂H₂) becomes ethane (C₂H₆). The formula itself tells part of the story: we went from 2 carbons and 2 hydrogens to 2 carbons and 6 hydrogens. That extra four hydrogens are the key!
Drawing it out is like telling a story with pictures. You have the starting scene with the high-energy ethyne. Then, you show the process of adding hydrogen, maybe with arrows and the hydrogen molecules. Finally, you reveal the peaceful ethane at the end.
It’s the visual contrast that’s so engaging. The dense, tightly bonded ethyne versus the looser, single-bonded ethane. It’s the difference between a coiled spring and a stretched-out spring.
And the “complete” part is important. It means we’re not stopping halfway. We’re not leaving any of those triple bonds partially intact. We’re going all the way to saturation, to maximum hydrogen content.
Imagine you have a very small, very bouncy ball (ethyne). Hydrogenation is like wrapping it in layers of soft padding. Complete hydrogenation means you keep adding padding until the ball can barely bounce anymore, becoming a soft, stable cushion (ethane).
It’s a simple concept with a dramatic visual outcome. From something that looks like `HC≡CH` (if we draw it linearly) to something that looks like `CH₃-CH₃`. The change in bonding is the real spectacle.
This reaction is fundamental in chemistry. It’s how we can convert less stable compounds into more stable ones. It’s a basic building block for understanding organic chemistry.
The entertainment comes from seeing the ‘before’ and ‘after’. The vibrant, reactive ethyne and the calm, steady ethane. It’s a transformation that’s both visually striking and conceptually satisfying.
So, next time you hear about the “complete hydrogenation of ethyne,” picture that energetic little molecule getting a full hydrogen hug, transforming into the chill ethane. It’s a simple process, but the visual story it tells is wonderfully engaging!
