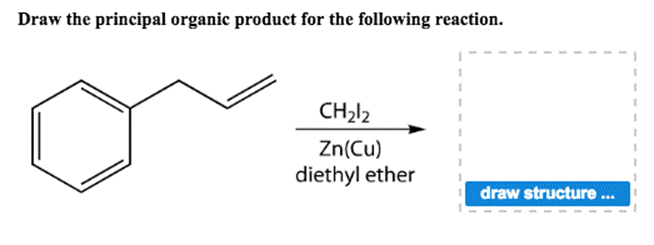
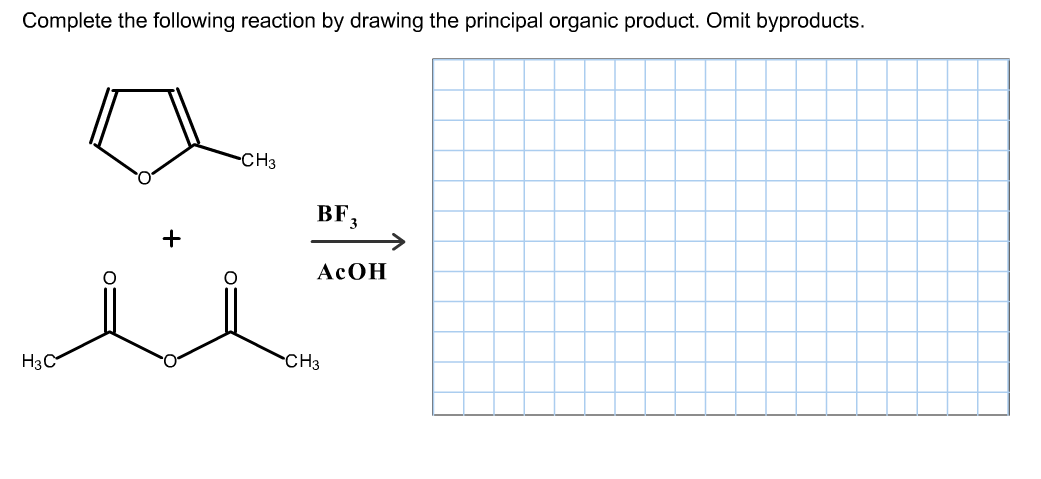
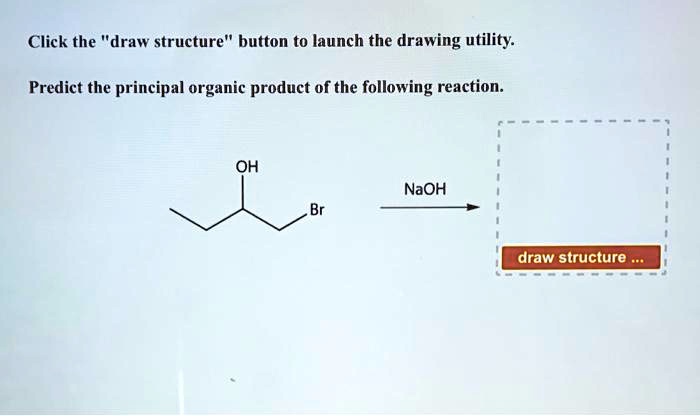
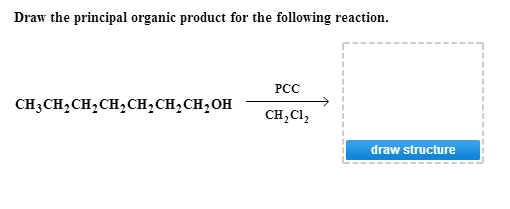
Draw The Principal Organic Product For The Following Reaction
Hey there, science explorers! Ever feel like chemistry is this big, scary monster with a million complicated rules? Well, buckle up, because today we're going to tackle a little puzzle that's more like a fun game of "what happens next?" It’s all about predicting the principal organic product. Think of it as being a detective, but instead of clues, you're looking at little molecular ingredients, and instead of a culprit, you're figuring out what they'll magically transform into!
Imagine you have a bunch of LEGO bricks. You know what each brick looks like, right? Now, imagine you're told to connect certain bricks together. You don't need a degree in LEGO-engineering to figure out that a couple of red bricks might join up to make a small red wall. Chemistry is kind of like that, but with tiny, invisible building blocks called atoms!
So, when we talk about drawing the principal organic product, we're essentially asking: "Given these starting molecules, what's the most likely and biggest thing they're going to form when they hang out together?" It’s like asking what's the most obvious sandwich you can make with bread and peanut butter – a peanut butter sandwich, duh!
Let’s say we have our starting materials, which are like our ingredients. They’re minding their own business, but then, poof, we add a little something extra, a reagent. This reagent is like the chef in our kitchen, or the magic spell that makes things happen. It nudges, it shoves, it encourages the starting molecules to get together and form something new.
And what are these "starting molecules" made of? Mostly carbon and hydrogen, hence the term organic. Think of all the amazing things carbon can do! It's the backbone of life, the stuff in your pencil, and even the sparkle in diamonds. It's a superstar element, and when it teams up with hydrogen, and sometimes other friends like oxygen or nitrogen, it creates a whole universe of fascinating compounds.
Now, the principal part is super important. Sometimes, a reaction can lead to a few different possible outcomes. It’s like having a recipe that says you can make either a cake or cookies with the same ingredients, depending on how you mix them. But in our chemistry adventure, we're usually interested in the one that happens the most. It's the main event, the star of the show, the one that shouts, "Here I am!"
So, when you're given a reaction, you're looking at the starting materials, the reagent that’s going to stir things up, and then you get to play detective and figure out what the main creation will be. It’s about understanding how these little molecular buddies like to connect and rearrange themselves. Do they want to grab an extra atom? Do they want to kick another atom out? Do they want to link up and form a bigger chain?
Let’s consider a super simple example, though we won't draw it out fully (that's your job, detective!). Imagine you have something called an alkene. Think of it like a molecule with a little double bond party going on. It's a bit more reactive, a bit more eager to make new friends than its single-bonded buddies. When we bring a simple molecule like bromine (Br2) to this party, the bromine atoms just can't resist!
The double bond in the alkene is like an open invitation. It breaks open, and each carbon atom in that double bond happily accepts a bromine atom. So, instead of a double bond, you get two new single bonds, and the alkene has now "added" the bromine to itself. The principal organic product here is a molecule where the bromines have joined the original carbon chain.
It’s like taking two people holding hands (the double bond) and then introducing two new people who also want to hold hands with them. Now, instead of holding hands with each other, they're each holding hands with a new person. The original two are still connected, but now they have new partners!
Sometimes, the reagent is a bit more specific. Imagine you have a molecule with a particular "functional group." Think of functional groups as the personality traits of molecules. Some are shy, some are outgoing, some are a bit mischievous. When a reagent comes along, it might be specifically looking for a certain personality trait to interact with.
For instance, if you have an alcohol, which has an -OH group (that's an oxygen atom bonded to a hydrogen atom), and you add a strong acid, the -OH group might get a bit grumpy and decide to leave! The acid helps it to depart, and then something else might hop in to take its place. The principal organic product will be whatever replaces that -OH group, forming a new functional group.
It’s like a molecule has a favorite toy, and a reagent comes along and says, "Hey, that's a nice toy, but I have an even cooler toy! Want to trade?" The molecule, being open to new experiences, might just make the trade, and you get a new molecule with the cooler toy!
The beauty of predicting the principal organic product is that there are underlying principles at play. It’s not just random; it’s like following the laws of molecular attraction and stability. Molecules tend to go towards arrangements that are more stable, or where the atoms have the most satisfying bonds.
Think about magnets. They have positive and negative ends, and they are attracted to each other. Molecules have similar kinds of "charges" or electron densities, and they interact in predictable ways. Your job as the budding chemist is to understand these attractions and repulsions.
We're not asking you to be a full-blown molecular matchmaker on your first try! It's a skill that builds with practice. Each time you look at a reaction, you're adding another piece of knowledge to your mental LEGO box. You start to recognize patterns, like how certain reagents always bring certain atoms along for the ride, or how certain functional groups are always ready for a transformation.
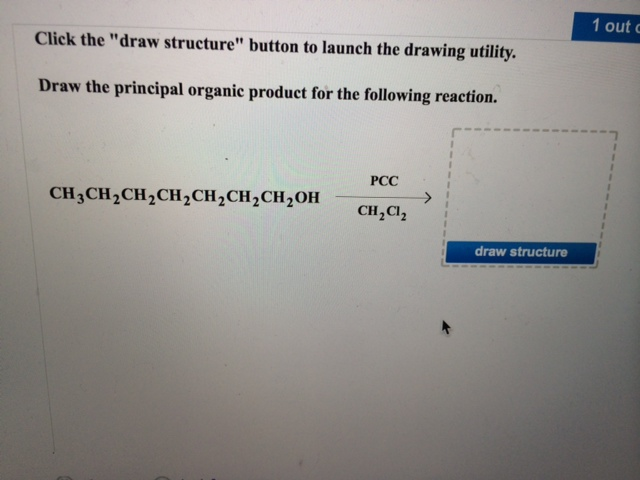
So, when you're presented with a reaction and asked to draw the principal organic product, take a deep breath. Look at your starting materials. What are they made of? What are their key features? Then, look at the reagent. What's its job? What does it seem to be looking for?
Imagine you're watching a play. You see the actors (the starting materials) and the scriptwriter (the reagent). Your task is to predict the most likely scene that will unfold. What will the characters say and do? Who will end up paired with whom? The principal organic product is the final scene, the most prominent outcome of the actors following the script.
It's a wonderful way to understand how the world around us is constantly changing and transforming at a molecular level. From the food we eat to the air we breathe, these little organic molecules are the unsung heroes, constantly reacting and creating new things. And you, by learning to predict the principal organic product, are becoming a master of understanding these fundamental transformations.
So, don't be intimidated! Think of it as a fun challenge, a molecular puzzle waiting to be solved. With each reaction you analyze, you're becoming a more confident molecular detective, ready to uncover the hidden treasures of organic chemistry. Let the drawing commence, and may your products be principal and perfectly predicted!
