Draw The Predominant Form Of Histidine At Ph 0
So, I was messing around with some old chemistry textbooks the other day, you know, the kind that smell faintly of dust and desperation? And I stumbled across this diagram of histidine. My first thought was, "Wow, that amino acid looks like it's having an existential crisis." Little did I know, that crisis is directly tied to what happens to it when you dunk it into a solution that’s basically pure acid. Seriously, pH 0? That’s like the chemical equivalent of a toddler throwing a full-blown tantrum.
It got me thinking about how we usually picture amino acids, right? Nice, neat structures, maybe a little zwitterionic charm. But the reality, especially under extreme conditions like a pH of 0, is a whole different beast. It’s not just about drawing pretty shapes; it’s about understanding how these fundamental building blocks of life react to their environment. And histidine, bless its quirky heart, has a particularly interesting story to tell when you crank the acidity up to eleven.
You see, histidine is kind of the oddball of the amino acid family. It’s got this imidazole ring, which is a bit like a tiny, slightly unstable social club. This ring is super important because it can act as both an acid and a base. It’s like that friend who can always see both sides of an argument, but sometimes gets caught in the middle and ends up overloaded. And at pH 0, that imidazole ring is really put to the test.
So, what is pH 0, anyway? Imagine the most concentrated hydrochloric acid you can get your hands on. Yeah, that’s pretty much it. It’s a world where protons (H+) are absolutely everywhere, practically elbowing each other for space. If you were a molecule in that environment, you’d be constantly bombarded, and your own electrons would be in high demand. It’s a really, really proton-rich environment.
When we talk about drawing the predominant form of an amino acid at a specific pH, we're essentially asking: "What does this molecule look like when it's most stable and abundant under these acidic conditions?" It's about identifying which of its possible charged states is the most likely one to be hanging around.
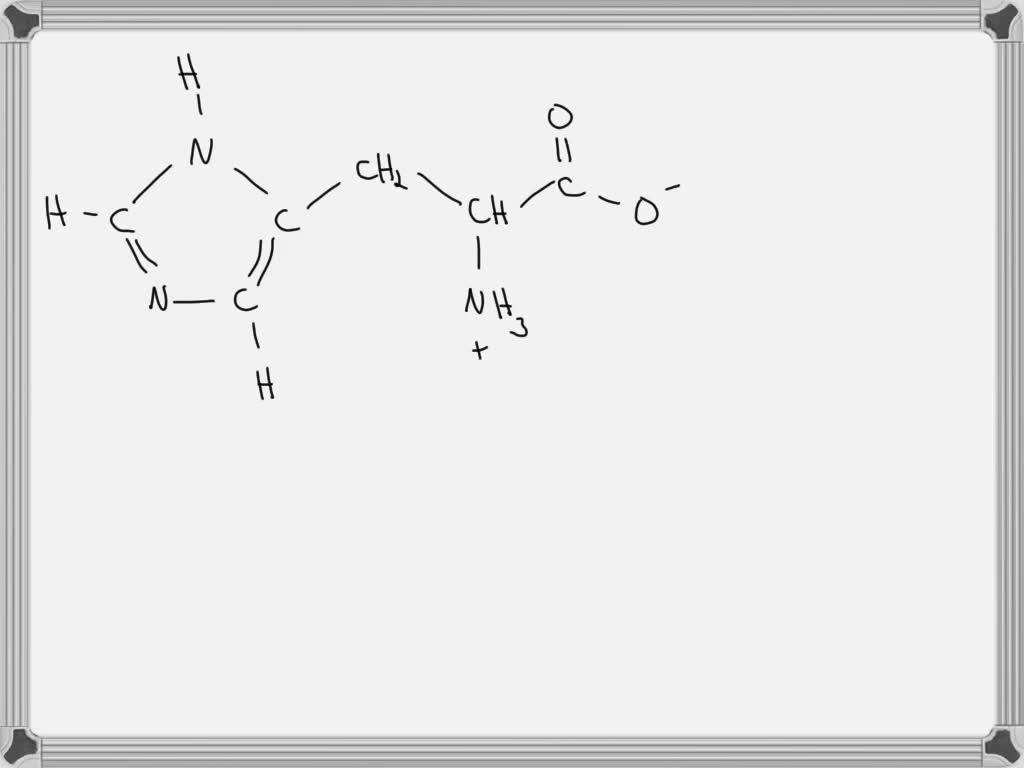
Let's break down histidine itself first. We’ve got the standard amino acid backbone: a central alpha-carbon, an amino group (-NH2), a carboxyl group (-COOH), and a side chain. The side chain is where the magic (or in this case, the pH-dependent drama) happens. For histidine, this side chain is that imidazole ring I mentioned.
Now, amino acids have functional groups that can gain or lose protons. The main ones we usually worry about are the amino group and the carboxyl group. At neutral pH (around 7), the amino group is typically protonated (NH3+) and the carboxyl group is deprotonated (COO-), giving us the zwitterion. This is the image most people have in their heads, right? Looks stable, balanced, all good.
But as you lower the pH, you’re adding more and more protons to the solution. And these protons aren’t shy. They’ll go looking for any electron-rich spot to latch onto. This is where that imidazole ring in histidine becomes crucial. This ring has a nitrogen atom with a lone pair of electrons, just itching to grab a proton.
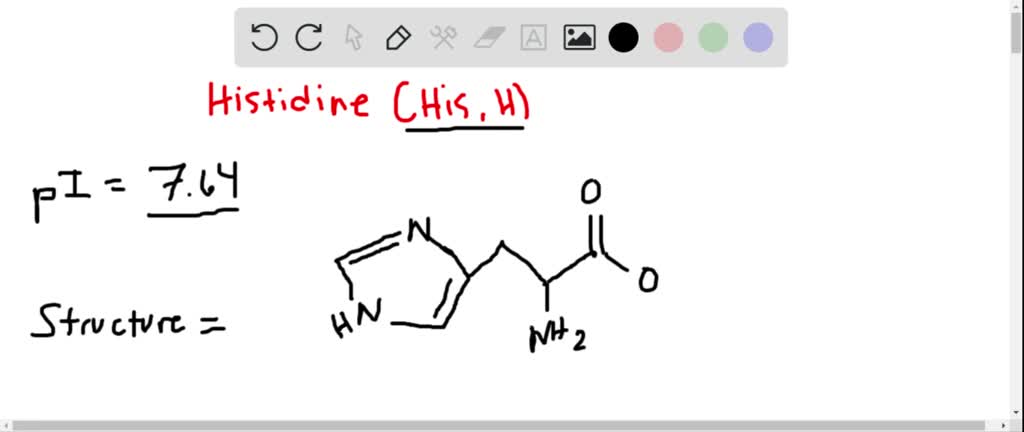
At a very low pH, like pH 0, everything that can be protonated, will be protonated. Think of it as a proton-grabbing free-for-all. So, let's consider each part of histidine and see what happens.
First, the alpha-amino group. This is a pretty basic group. At pH 0, it’s definitely going to be fully protonated. So, instead of -NH2, we’ll have a positively charged -NH3+. This is pretty standard for any amino acid in strong acid.
Next, the alpha-carboxyl group. This group is acidic. Normally, at neutral pH, it’s -COO-. But in a super acidic environment, where there are tons of protons, it’s going to snatch one back. So, it will be in its protonated form, -COOH. This also makes sense – the more acidic the environment, the less likely an acidic group is to give away its proton.
Now, for the star of the show: the imidazole ring. This is where histidine really shines (or maybe just gets a bit overwhelmed). The imidazole ring has two nitrogen atoms. One has a hydrogen attached, and the other has a lone pair of electrons. The key nitrogen atom here is the one with the lone pair. This nitrogen has a pKa value of around 6.0. This means that around pH 6, it’s about 50% protonated and 50% deprotonated.
But we're not at pH 6, are we? We're at pH 0. That's a massive difference. Remember how pKa works? It's a measure of acidity. If the pH is much lower than the pKa, the protonated form is favored. If the pH is much higher than the pKa, the deprotonated form is favored.
So, at pH 0, which is drastically lower than the pKa of the imidazole nitrogen (around 6.0), that nitrogen will be extremely protonated. It’s basically a magnet for protons at this point.
This means the imidazole ring essentially gains an extra proton on that key nitrogen. The ring structure itself will remain, but the nitrogen will now have a positive charge. So, the imidazole side chain, which is normally neutral or slightly basic depending on the pH, becomes positively charged at pH 0.
So, let's put it all together. At pH 0:
- The alpha-amino group is protonated: -NH3+
- The alpha-carboxyl group is protonated: -COOH
- The imidazole ring nitrogen is protonated: The ring gains a positive charge.
This means that histidine at pH 0 carries three positive charges! Yes, you read that right. Three! One on the amino group, one on the imidazole ring, and if you really want to be pedantic about it, you could say the carboxyl group has a "net zero" charge in its protonated form, so it contributes to the overall charge balance but isn't a discrete positive charge. But for the purpose of understanding the molecule's behavior, it’s important that it's not an anion like it would be at high pH. The key takeaway is that both the amino and imidazole groups are highly protonated and positively charged.
So, if you were to draw this, you’d have a histidine molecule that looks quite different from its zwitterionic cousin. The backbone would be the standard structure, but with a fully protonated amino group (NH3+). The carboxyl group would be COOH. And the imidazole ring would have that extra proton on the nitrogen, giving it a formal positive charge. You can imagine it looking a bit like a small, positively charged hedgehog!
This triple-positive charge is what makes histidine so unique and so interesting. It's incredibly hydrophilic and will readily interact with negatively charged molecules. This is actually super important in biology. In the active sites of enzymes, for example, histidine's ability to become positively charged at low pH (or even just protonated at physiological pH) allows it to interact with negatively charged substrates or amino acid residues, playing a critical role in catalysis and binding.
Think about it this way: at pH 0, histidine is practically screaming for electrons and is a powerful positively charged entity. This isn't just a chemistry quirk; it's fundamental to how histidine participates in the complex dance of biological molecules.
The pKa values of the ionizable groups in an amino acid are key to understanding its behavior at different pH levels. For histidine, the relevant pKa values are:
- Alpha-carboxyl group: ~2.1
- Imidazole side chain: ~6.0
- Alpha-amino group: ~9.3
Let's revisit these with our pH 0 scenario. When the pH is significantly lower than the pKa of a group, that group will be predominantly in its protonated form. When the pH is significantly higher than the pKa of a group, that group will be predominantly in its deprotonated form.
For the alpha-carboxyl group (pKa ~2.1): At pH 0, pH << pKa. So, it's fully protonated (-COOH). For the imidazole side chain (pKa ~6.0): At pH 0, pH << pKa. So, it's fully protonated (the imidazole ring accepts an extra proton, giving it a positive charge). For the alpha-amino group (pKa ~9.3): At pH 0, pH << pKa. So, it's fully protonated (-NH3+).
So, yes, three positive charges are the order of the day at pH 0. It's a powerful demonstration of how the environment can drastically alter the charge and therefore the chemical properties of a molecule.
The “predominant form” is essentially the form that has the lowest free energy and is most stable under those specific conditions. At pH 0, the universe is awash in protons, and histidine’s structure is such that it can accommodate several of them, becoming highly positively charged in the process.
It’s a bit like having a sponge soaking up as much water as possible. At pH 0, histidine is a proton sponge. It’s not just a theoretical exercise; understanding this helps us appreciate how amino acids function in different cellular compartments, which can have varying pH levels, or even in pathological conditions where pH might be altered.
It’s also a good reminder that the chemical structures we often see in textbooks are simplifications. The reality is dynamic and responsive. Histidine’s imidazole ring is particularly adept at this dynamic responsiveness, making it a versatile player in protein chemistry.
So, next time you see a diagram of histidine, don't just think of the zwitterion. Remember its alter ego: the highly protonated, triple-charged entity that emerges when it’s dropped into the chemical equivalent of a mosh pit. It’s a testament to the power of protonation and the fascinating adaptability of these fundamental building blocks of life.
And honestly, who knew that a little molecule could have such a dramatic transformation? It's like a superhero origin story, but with more protons and fewer capes. Just a humble amino acid, flexing its chemical muscles under extreme pressure.
It really makes you appreciate the elegance of chemistry, doesn't it? Even in the most acidic environments, there's a predictable (if slightly intense) outcome. And for histidine, that outcome at pH 0 is a seriously positive personality.
So, if you ever find yourself in a situation where you need to draw histidine at pH 0, just remember: think protons, think positive charges, and think of that amazing little imidazole ring working overtime.
