Draw The More Stable Chair Conformer Of Cis-1-ethyl-2-methylcyclohexane

Alright, settle in, grab a croissant, and let me tell you a tale. A tale of chairs. Not the comfy kind you’re currently sinking into (or maybe it’s one of those stiff café chairs, no judgment here), but the kind that exist in the microscopic, mind-bending world of chemistry. We’re talking about cyclohexane chairs, folks, and specifically, the absolute chillest way to arrange a molecule named cis-1-ethyl-2-methylcyclohexane. Think of it as molecule feng shui, but with a lot more awkward angles and a surprising amount of existential dread for a bunch of atoms.
Now, cyclohexane itself is a pretty standard ring of six carbon atoms. Boring, right? Wrong! These little carbon buddies love to do this fancy little dance, twisting and turning into what chemists affectionately call a “chair” conformation. Why a chair? Well, if you squint really hard and imagine it with your third eye, it kinda looks like a fancy throne from a medieval king who was really into his furniture. It’s got these up and down bits, you know, like legs and a back. Very regal.
But here’s where it gets spicy. Molecules, bless their little hearts, aren't content with just one way to be a chair. Oh no. They can flip-flop! Imagine your comfy armchair doing a full-on somersault, and now the back is the legs and the legs are the back. This is called interconversion, and it’s basically the cyclohexane equivalent of a teenager dramatically changing their entire personality overnight. For cis-1-ethyl-2-methylcyclohexane, this flip-flopping is where the real drama unfolds. Because, unlike us humans who can just wear sweatpants all day and call it a vibe, molecules have preferences. And their preferences are dictated by stability.
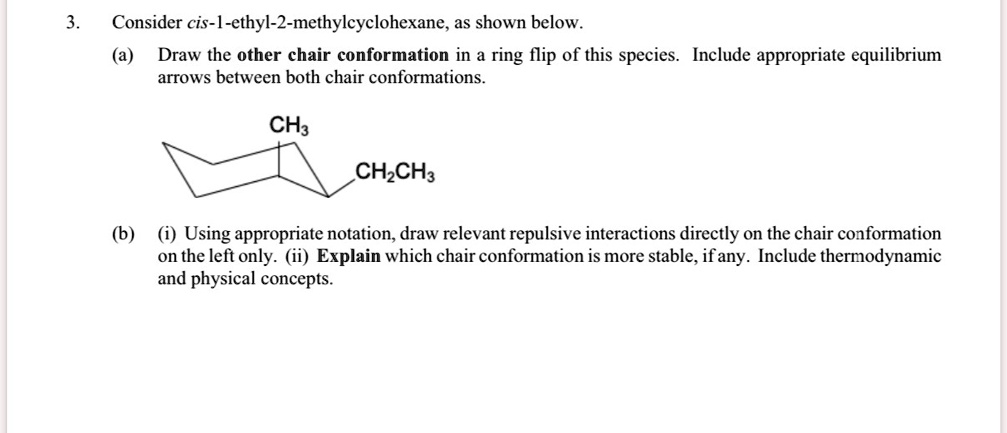
So, what is this mysterious cis-1-ethyl-2-methylcyclohexane? Let’s break it down like a bad breakup. We’ve got a cyclohexane ring, of course. Then, attached to it, we have an ethyl group (that’s two carbons stuck together, like a tiny carbon hot dog) and a methyl group (just one carbon, a lone ranger). The “cis” part is the real kicker. It means that these two little buddies, the ethyl and the methyl, are on the same side of the ring. Imagine two kids sitting on the same side of the couch, trying to share a remote. Awkward, but they’re in it together. If it were “trans,” they’d be on opposite sides, probably giving each other the side-eye.
Now, these groups, our ethyl and methyl friends, don't just float around happily. They have to exist in the chair conformation. And in this chair, the carbon atoms have different positions. We’ve got the axial positions, which are straight up and down, like little flagpole seats. Think of them as the premium, but potentially drafty, seats in the molecular theater. Then we have the equatorial positions, which are more out to the side, like cozy armrests. Much more comfortable, generally speaking.
The catch is, these groups, especially bigger ones like our ethyl friend, really don't like being crammed into the axial positions. It’s like trying to fit a king-sized mattress into a broom closet. They get all bunched up and grumpy. This crowding is called steric strain, and it’s the arch-nemesis of molecular happiness. Molecules, just like us, want to avoid awkward social situations and unnecessary discomfort.
So, in our cis-1-ethyl-2-methylcyclohexane, we have an ethyl group and a methyl group on the same side of the ring. They’re the cis-couple, remember? Now, let’s imagine our chair. When they’re both axial, it’s a party nobody asked for. The ethyl is sticking straight up, bumping elbows with its neighbor, and the methyl is doing the same. It’s a molecular mosh pit, and nobody’s having a good time. This conformation is unstable, like trying to balance a bowling ball on a unicycle.
But remember that flip-flopping chair? When the chair flips, the axial positions become equatorial and vice-versa. So, what was up becomes out, and what was out becomes up. It’s a molecular identity crisis, but it often leads to a better arrangement. For our cis-molecule, there’s a conformation where both the ethyl and the methyl can get into the more spacious equatorial positions. Think of it as finding the perfect spot on the couch where nobody’s knees are getting poked.
Imagine drawing this. You draw your cyclohexane ring in that classic chair shape. Then you have to decide where to put the ethyl and methyl. Since they are cis, they need to be on the same side. Let’s say you put one going up (axial) and the other going down (axial) relative to the plane of the ring. They're on opposite sides of the ring, but in this chair drawing, one might appear "up" and the other "down" relative to the plane. This is where it gets tricky and people often get confused. The key is to visualize them in relation to each other across the ring.
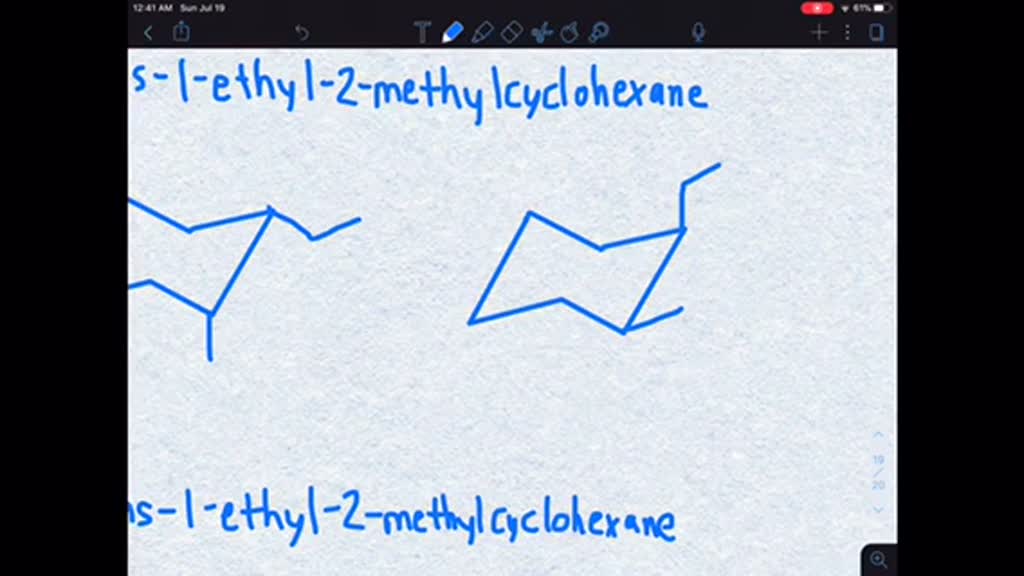
But the more stable conformer? Oh, that’s where our ethyl and methyl groups are both chillin’ in their equatorial spots. One will be sticking out from a carbon at an angle pointing roughly to the left and a bit up, and the other will be sticking out from the next carbon, angled roughly to the right and a bit down. They’re both on the same side of the ring itself, but in their respective chair positions, they’re not bumping into each other in the axial positions. They're like two friends who have found the perfect balance of personal space while still being close enough to gossip.
Think of it like this: you’ve got your cyclohexane chair. The carbons are numbered. Let’s say ethyl is on carbon 1 and methyl is on carbon 2. If they are cis, and you draw them both equatorial, one will be pointing generally "up" from its carbon and the other generally "down" from its carbon. But they are on the same side of the overall molecule. It’s a subtle, but crucial, distinction. This arrangement minimizes that pesky steric strain. The bigger the group, the more it hates being axial. Ethyl is bigger than methyl, so it really wants to be equatorial.
So, when you’re asked to draw the more stable conformer, you’re looking for the chair where the ethyl and methyl groups are as far away from each other as possible, ideally in those comfy equatorial positions. It’s a molecular negotiation, a quest for the least amount of awkward molecular bumping. And the winner, the champion of chill, is the conformation where our cis-ethyl and cis-methyl are both rocking the equatorial lifestyle. They’ve found their molecular happy place, and frankly, who are we to argue with that?
