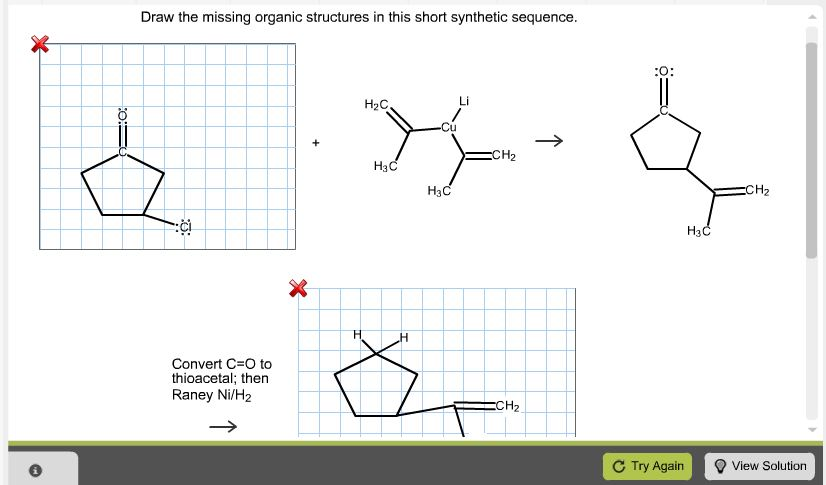
Draw The Missing Organic Structures In This Short Synthetic Sequence
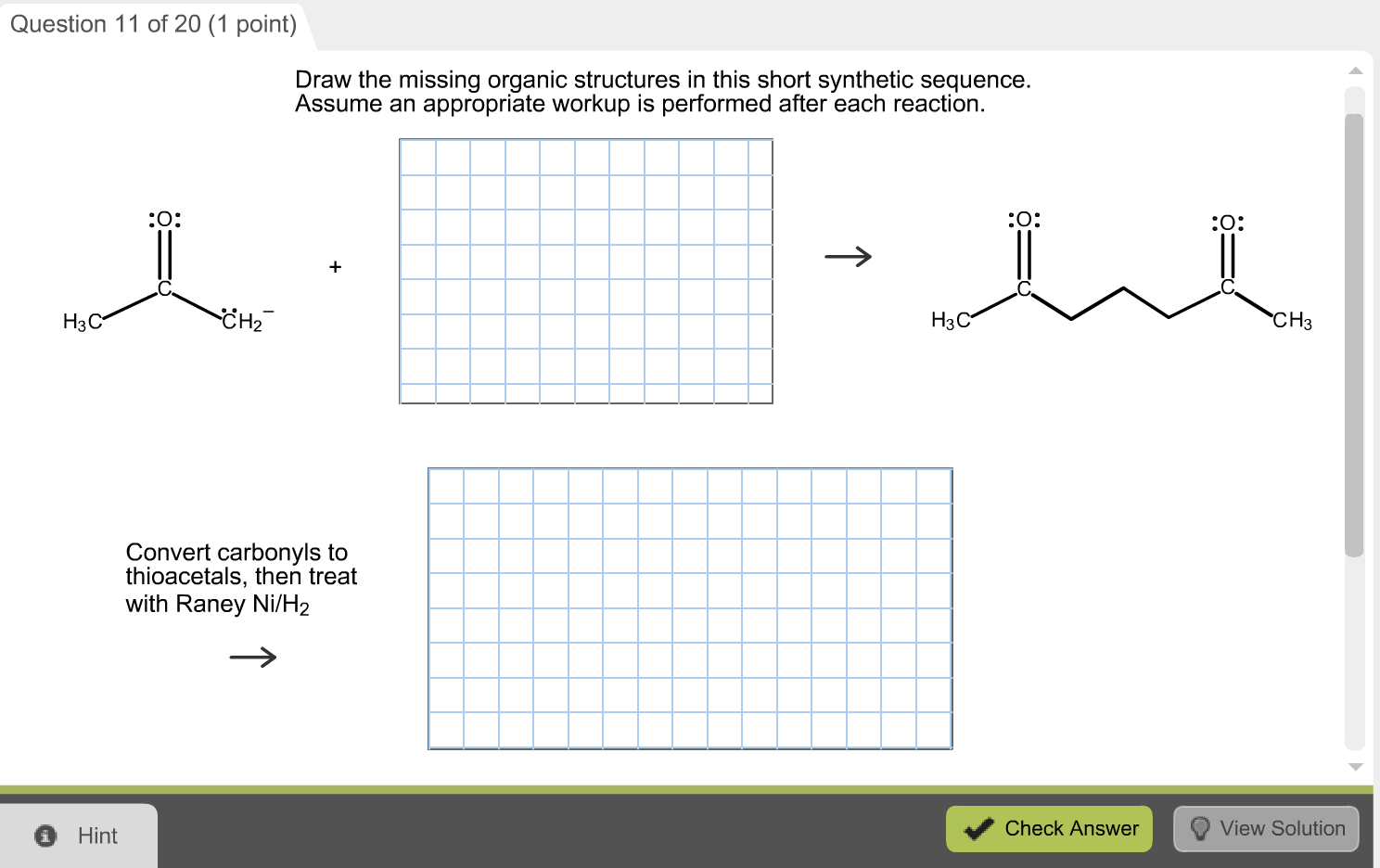
Alright folks, gather ‘round, pull up a beanbag, or just lean precariously off your chair like I usually do. We’re about to embark on a little adventure, a chemical quest, if you will. Think of it like a treasure hunt, but instead of gold doubloons, we’re hunting for… well, missing molecules. Yep, those elusive little guys that are just implied in our grand chemical symphony.
So, imagine you’re a mad scientist, or maybe just someone who’s had too much coffee and decides to doodle on a napkin. You’ve got this elegant little roadmap of a chemical reaction, a sequence, they call it. It’s like a recipe for making something cool, but some of the ingredients are a bit… abstract. They’re the invisible friends of chemistry, the ones you’re supposed to just know are there. Kinda like that feeling when you’re sure you left your keys on the counter, but they’ve mysteriously teleported to the fridge. Happens to the best of us.
Our mission, should we choose to accept it (and we totally have, because, let’s be honest, it’s more fun than doing laundry), is to fill in the blanks in this short synthetic sequence. We’re not talking about building a rocket ship here, just a few nifty steps to create a rather charming organic molecule. Think of it as assembling a tiny LEGO set, but with slightly more dramatic explosions if you get it wrong (kidding! mostly).
The Grand Overture: Setting the Stage
We start with a molecule that’s, shall we say, feeling a little plain. Let’s call it our “Plain Jane”. It’s got a nice, sturdy carbon backbone, a few hydrogen buddies clinging on for dear life, and a functional group that’s just begging for some attention. This functional group is like the diva of the molecule, ready for her close-up. It’s an alcohol, specifically, a primary alcohol. You know, the kind that’s always a bit too eager to react. It’s like that friend who volunteers for everything, even when they probably shouldn’t.
Now, our Plain Jane is about to undergo a transformation. The first step in our little chemical drama involves treating this poor, innocent alcohol with something called PCC. PCC, my friends, stands for Pyridinium Chlorochromate. Sounds fancy, right? It’s basically a polite way of saying “let’s oxidize this guy, but not too much.” Think of it as a gentle nudge, not a full-blown shove. PCC is like the well-behaved older sibling who makes sure you don’t get into too much trouble.
So, what happens when Plain Jane meets PCC? Well, our humble primary alcohol, with its two hydrogens attached to the carbon bearing the -OH group, is going to lose those hydrogens. One goes with the oxygen, forming water (yes, water, the universal solvent and occasional beverage!), and the other hydrogen, well, it just ghosts. Poof!
The First Transformation: From Booze to Bang!
When an alcohol loses those two hydrogens, it doesn’t just sit there feeling naked. Oh no! The carbon and oxygen atoms get a little… closer. They decide to form a double bond. This is where the magic really starts to happen. Our Plain Jane, the primary alcohol, is now becoming an aldehyde. An aldehyde is like the cooler, more reactive cousin of the alcohol. It’s got a carbonyl group (that’s a C=O, for the uninitiated) at the end of the chain. It’s got a certain je ne sais quoi, a little spark of excitement. Think of it as going from a quiet library reading to a lively karaoke night.
So, the first missing structure is this aldehyde. It’s a crucial step because aldehydes are like the chameleons of organic chemistry, ready to transform into a million different things. They’re the little black dresses of the molecule world – versatile and always in style.
The Plot Thickens: A New Challenger Appears
Now, our newly formed aldehyde is feeling pretty good about itself. It’s strutting its stuff, ready for its next adventure. And what’s next? Oh, just another awesome reagent! This time, we’re bringing in the big guns, or at least, the moderately-sized, very reactive guns. We’re talking about Grignard reagents.
Grignard reagents are like the ultimate party guests. They’re organometallic compounds, meaning they have a carbon atom directly bonded to a magnesium atom. Think of it as a carbon atom that’s found its wealthy, metal-loving sugar daddy. These guys are nucleophilic, which is a fancy way of saying they’re super eager to donate electrons and attack positive centers. They’re the chemical equivalent of a hungry teenager spotting a pizza delivery.
In our sequence, we’re going to use a specific Grignard reagent. Let’s say it’s methyl magnesium bromide (CH₃MgBr). That little methyl group (CH₃) is the star player here. It’s the one with the electrons ready to rumble. Our aldehyde, with its electrophilic carbon in the carbonyl group (remember that C=O?), is just begging to be attacked by this electron-rich methyl group.
The Second Encounter: A Carbon-Carbon Connection!
So, the methyl group from our Grignard reagent swoops in and attacks the carbonyl carbon of the aldehyde. It’s a classic case of attraction! The double bond in the carbonyl group breaks, and the methyl group forms a new carbon-carbon bond. This is a HUGE deal in organic chemistry, people! We’re building bigger, more complex molecules by connecting these little carbon pieces. It’s like finally getting to use the really cool, oddly shaped LEGO bricks.
When this happens, the oxygen atom in the former carbonyl group grabs onto a proton (an H⁺) from the solution during a subsequent workup (which is basically the chemical equivalent of cleaning up after a messy party). This protonation turns the oxygen back into an alcohol group. But wait! This isn’t just any alcohol anymore. Because the Grignard reagent added a carbon chain, we now have a secondary alcohol. Two carbon atoms are attached to the carbon bearing the -OH group. It’s like our molecule has gone from being a solo artist to being in a dynamic duo.
This secondary alcohol is our second missing structure. It’s a beautiful thing, this new carbon-carbon bond. It shows that our molecule has grown, it has evolved. It’s no longer Plain Jane; it’s “Jane and a new friend,” ready for whatever the chemical world throws at them.
The Grand Finale: A Touch of Class
We’re almost there! We’ve got our secondary alcohol, looking all sophisticated. But our sequence isn’t quite finished. There’s one last tweak to be made. The final step involves treating our secondary alcohol with a reagent that sounds a bit like a villain from a superhero movie: CrO₃ in aqueous acid. Yes, we’re bringing in chromium trioxide, the more aggressive cousin of PCC. This guy doesn’t play around.
Just like PCC, CrO₃ is an oxidizing agent. But unlike PCC, which stopped at the aldehyde for primary alcohols, CrO₃ is not afraid to push things further. And for secondary alcohols? Oh, it has a specific target in mind.
The Final Flourish: The Ketone Creation!
Our secondary alcohol has two carbon atoms attached to the carbon bearing the -OH group. The CrO₃ comes in and strips away the hydrogen from the -OH group and also strips away a hydrogen from one of the adjacent carbon atoms. Again, with the ghosting hydrogens! This process results in the formation of another double bond between the carbon and the oxygen. And what do we call a carbon atom double-bonded to an oxygen, with two other carbon atoms attached? You guessed it: a ketone!
Ketones are like the sophisticated evening wear of the organic molecule world. They have that C=O group, just like aldehydes, but they’re tucked neatly in the middle of carbon chains. They’re not as reactive as aldehydes, but they’ve got their own unique charm and are used in all sorts of cool applications, from solvents to flavorings. Think of it as moving from a lively karaoke night to a classy jazz club.
This ketone is our final, third missing structure. It’s the culmination of our little synthetic journey. We started with a simple alcohol, gently oxidized it to an aldehyde, used a Grignard reagent to build a new carbon-carbon bond and create a secondary alcohol, and finally, with a more potent oxidizing agent, transformed it into a ketone. It’s a testament to the power of organic reactions, transforming simple molecules into more complex and useful ones, one missing structure at a time.
So there you have it! The mystery is solved, the blanks are filled, and our molecule has gone through a rather impressive makeover. Next time you see a chemical reaction sequence, remember the invisible steps, the implied ingredients, and the sheer, unadulterated fun of figuring out where they all came from. Now, who wants more coffee?
