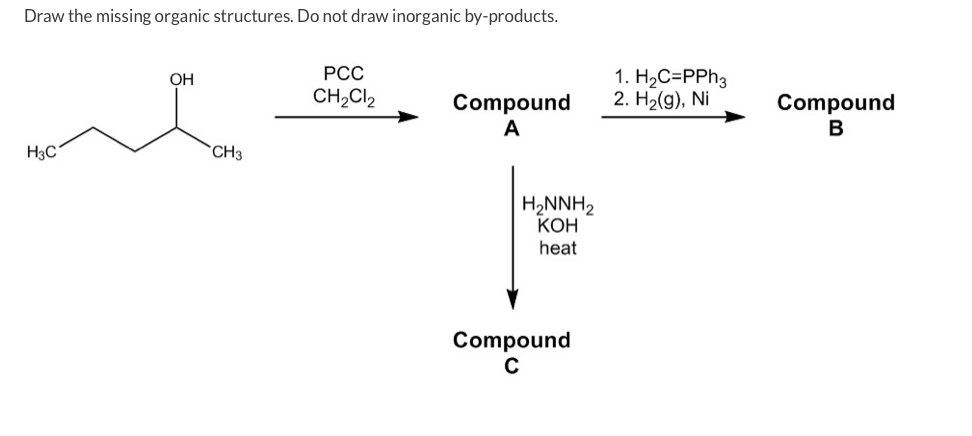
Draw The Missing Organic Structures. Do Not Draw Inorganic By-products.

Ever stared at a chemistry problem and felt a little… bewildered? You're not alone. Sometimes it feels like those organic chemistry textbooks are written in a secret code. And then, the dreaded "Draw the Missing Organic Structure" instruction appears.
It's like a cryptic crossword puzzle, but with more atoms and less Agnes from accounting winning the prize. The goal is to fill in the blanks, but only with the cool stuff. We're talking about the carbon chains, the rings, the bits that make molecules look like tiny, intricate sculptures.
And the best part? We get to ignore the "boring" stuff. You know, the inorganic by-products. Think of them as the background characters in a blockbuster movie. Important, sure, but nobody's really tweeting about the extra who was holding a tray of drinks.
So, we're on a mission to find our missing organic stars. These are the molecules that have personalities, that do the heavy lifting in reactions. They're the ones with names that sound like they belong in a fantasy novel, like "Benzene" or "Cyclohexane".
Sometimes, you see a reaction laid out, like a culinary experiment gone slightly awry. There are reactants, there are products, and then… a big, gaping hole where a molecule should be. It’s our job to figure out who’s supposed to be there.
This isn't about understanding the deep, dark secrets of quantum mechanics. No, this is more like playing a really sophisticated game of connect-the-dots. You look at what went in, you look at what came out, and you deduce what must have happened in between.
And the rule is crystal clear: only organic. We're talking about the molecules that are the building blocks of life, the basis of plastics, fuels, and pretty much everything you can touch. They've got that lovely carbon backbone.
So, that stuff that looks like a salt shaker exploded? The white powder that appears out of nowhere? Yeah, that's usually an inorganic by-product. Our instructions politely tell us to ignore it. It's like saying, "We appreciate your contribution, little salt crystal, but we're focusing on the main act here."
It’s a bit of an unpopular opinion, but I think this is the best part of organic chemistry. It’s where you get to be a detective. You’re not just memorizing formulas; you’re figuring out a story.
Imagine a chef preparing a complex dish. They've got their main ingredients, and then they’ve got the little bits and bobs that season and enhance. The missing organic structure is like the secret sauce that brings the whole thing together. We're tasked with figuring out what that secret sauce is made of.
And when you finally draw it, and it fits, and it makes sense? It’s a tiny victory. It’s a moment of clarity in a sea of confusing chemical notation. You feel a little bit like a genius.
Sometimes the missing structure is something simple, like adding a hydroxyl group to a molecule. Other times, it's a whole new ring system, a complex arrangement of atoms that looks like a miniature molecular city.
We might see a reaction with "Sodium Hydroxide" as a reactant. Now, that’s inorganic. It’s a useful tool, but it’s not our missing organic structure. We nod to it, acknowledge its presence, and then move on to the real prize.
The real prize is the organic molecule that was either created, transformed, or consumed during the process. It’s the one with the carbon skeleton, the hydrogen atoms, and maybe some oxygen, nitrogen, or halogens hanging around.
Think of it like this: you’re assembling a LEGO set. The instructions show you the final model. You have some pieces, and there are some empty spots. You have to figure out which LEGO bricks go into those empty spots to complete the model.

The inorganic by-products are like the little plastic bags the LEGOs came in. You open them, you use the bricks, and then you discard the bags. They’re part of the process, but not part of the final, impressive structure.
And sometimes, the missing structure is so obvious once you see it, you wonder how you missed it. It's like looking for your glasses when they're on your head. That's the beauty of it.
The names can be intimidating, though. Words like "Aldehyde", "Ketone", or "Ester" might sound like they require a secret handshake to understand. But in the context of a reaction, they often become your best clues.
You look at the functional groups on the reactants and products. Functional groups are like the "features" of organic molecules, the bits that give them their reactivity. Identifying these is key to solving the puzzle.
For example, if you see a reaction where an alcohol is converted into a carbonyl compound, and you're missing the carbonyl compound, your brain automatically goes, "Ah, this is probably a ketone or an aldehyde!"
Then, you just need to draw the correct carbon chain attached to that carbonyl. It’s about keeping track of the carbon atoms and making sure everything adds up. It's like a very precise accounting ledger for molecules.

And the beauty of it is, even if you're not a chemistry whiz, you can often intuit the missing piece. Your brain starts to recognize patterns. You’ve seen similar reactions before.
It’s a skill that develops with practice. The more you stare at these reaction schemes, the more your brain gets trained to see the gaps and fill them in. It’s like learning to spot Waldo in a crowded picture.
So, when you see that instruction, "Draw the Missing Organic Structure. Do Not Draw Inorganic By-products," don't panic. Take a deep breath. Look at the molecules that are already there. Think about what kind of transformations are happening.
Focus on the carbon-based heroes of the story. Ignore the bit players and the stagehands. You’re there to complete the masterpiece, the intricate dance of organic molecules.
And that feeling of satisfaction when you finally draw the correct structure? Priceless. It’s a small win, but in the world of chemistry, sometimes the smallest wins are the most rewarding. It's like finding a perfectly shaped piece for your puzzle.
So, embrace the challenge. Embrace the deduction. Embrace the exclusion of the mundane. Because in the grand theatre of organic chemistry, you are the director, and you decide which molecules get their spotlight.

And honestly, who wants to draw a bunch of generic salt molecules anyway? They don’t have the same flair as a complex "Carboxylic Acid" or a zippy "Alkene".
It’s all about the structure, the shape, the functionality. It’s about the magic of organic chemistry, where carbon is king and the missing pieces are just waiting for you to bring them to life.
So go forth, my fellow problem-solvers. Fill in those blanks. Create those structures. And leave the inorganic detritus to the footnotes of chemical history. Your organic creations deserve the applause.
"Chemistry is not just about reactions; it's about the story those reactions tell."
And we, my friends, are here to help write the most interesting chapters of that story by drawing the stars. The organic stars, of course.
It’s a simple rule, but it makes the whole process so much more enjoyable. Less clutter, more focus on the fascinating world of organic molecules.
So next time you see that prompt, remember: you're not just drawing atoms; you're completing a molecular narrative. And that, in itself, is pretty darn cool.
