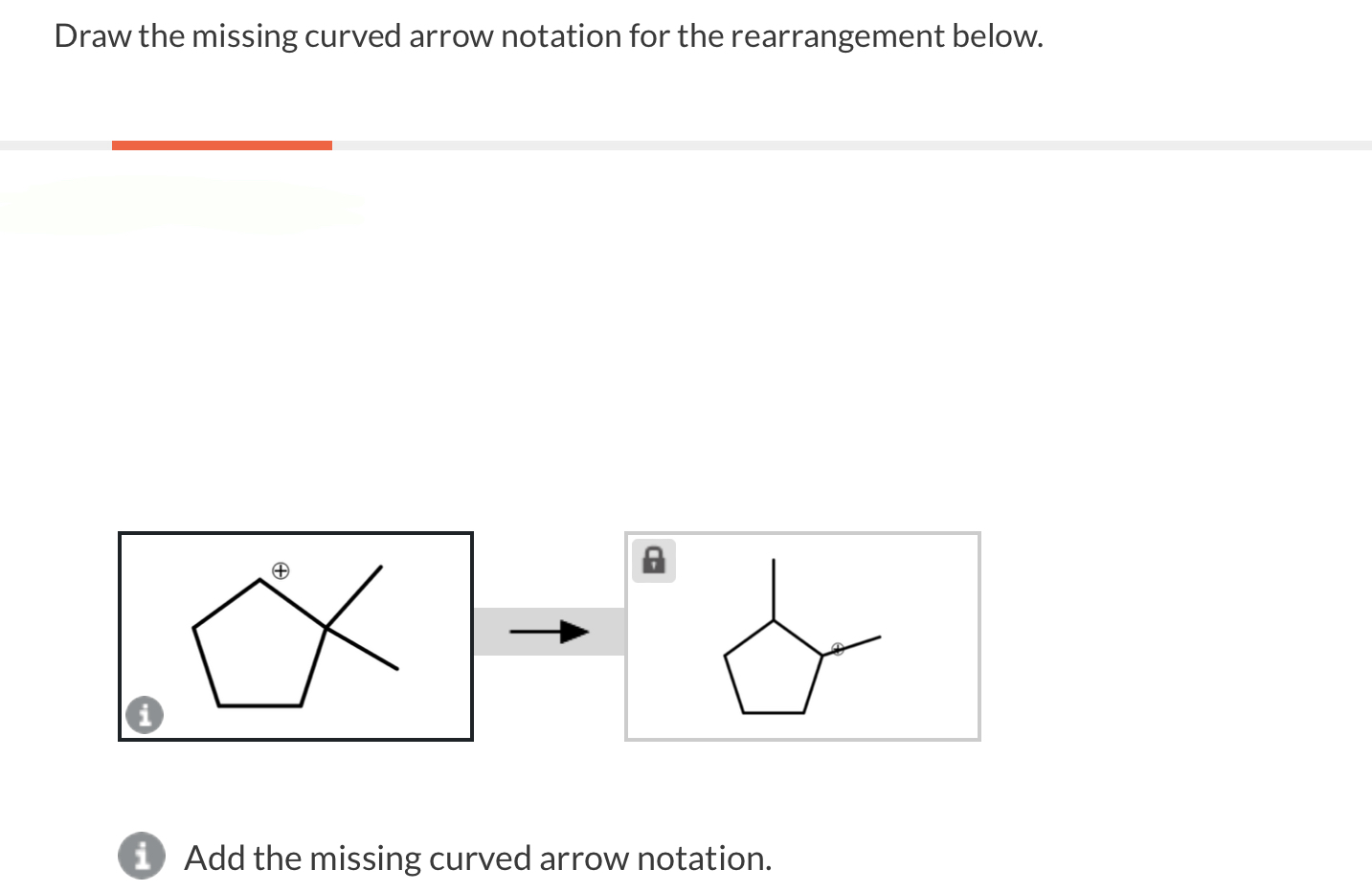
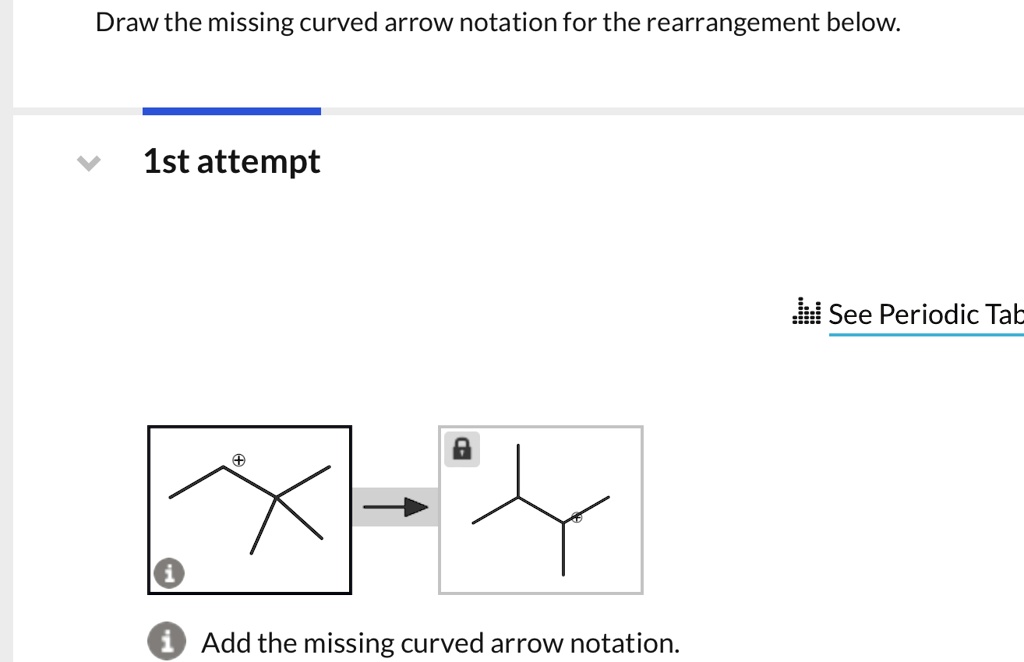
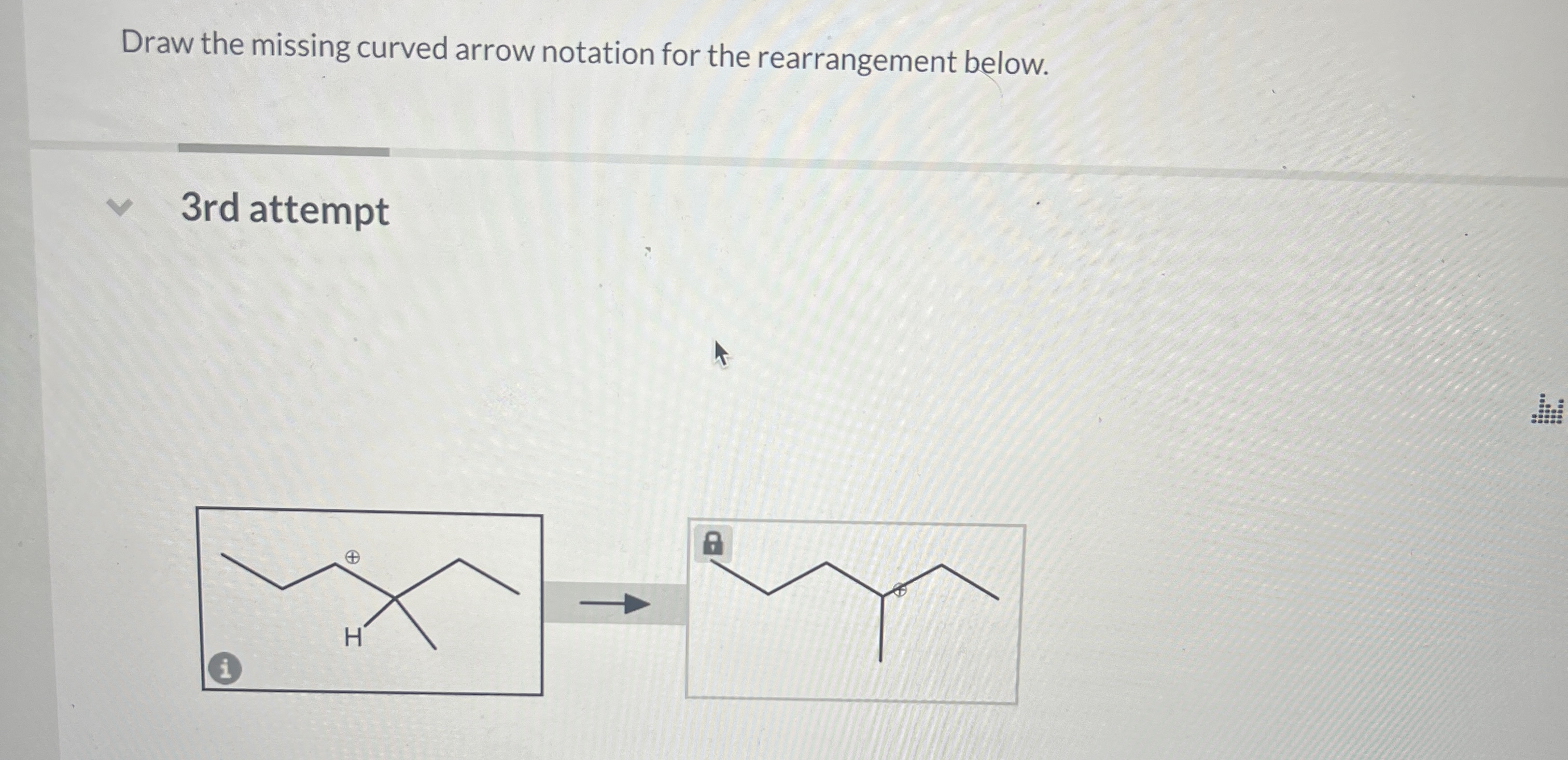
Draw The Missing Curved Arrow Notation For The Rearrangement Below.
Hey there, coffee buddy! Grab your mug, settle in, because we're diving into something super cool today. You know those little curvy arrows in organic chemistry? The ones that look like they're doing a tiny dance on the page? Yeah, those! They're actually our superheroes, showing us where electrons are zipping around and making all the magic happen. Today, we're gonna play a little game of "spot the invisible electron mover" with a classic organic chemistry rearrangement. Get ready to flex those brain muscles, but don't worry, it's gonna be fun. Like solving a puzzle, but with more atoms and way less dusting.
So, we've got this molecule, right? It's all settled in, looking perfectly normal. But then, bam! Something happens. A little internal reorganization. Think of it like your sock drawer. Everything's fine, then suddenly, you need that one specific sock, and suddenly, everything’s just… rearranged. That's what's happening here, but with chemical bonds. It's called a rearrangement, and it's basically the molecule saying, "You know what? This current arrangement isn't quite working for me. Let's try this other thing." Pretty sassy, huh?
Now, the crucial bit. These rearrangements don't just happen. They need a little nudge, a little guidance. And that's where our awesome curved arrows come in. They're not just decoration, people! They're the tell-tale signs of electron movement. Seriously, if you see a curved arrow, it means electrons are on the move. Think of it like a tiny, enthusiastic courier service, delivering negative charge from one atom to another. It's all about making things happier, usually by forming more stable bonds or getting rid of a super unstable intermediate. Chemistry is all about that stability, you know? Like us after a good cup of coffee – much more stable!
Let's look at our particular rearrangement. We have this molecule, and at first glance, it might seem like it's just chilling. But oh no, it's got a plan. There's a spot that's a little less than ideal, a little… electron-deficient. You know, like that feeling when you realize you forgot to buy milk. A minor crisis! And then, there's another spot that's practically overflowing with electrons. Like a hoarder's paradise of negative charge. So, what's a molecule to do?
It's time for some electron redistribution! And this is where you come in, my brilliant puzzle-solver friend. We need to draw the missing curved arrows. Imagine you're the conductor of this tiny molecular orchestra. Each arrow is a baton stroke, telling a pair of electrons exactly where to go. And it's not just random flailing! There are rules, like a really polite game of musical chairs. Electrons always move from a place of high electron density to a place of low electron density. It's like they're trying to even things out, a chemical form of sharing is caring.
So, take a good look at our starting molecule. Where do you see that electron-rich spot? Maybe a lone pair of electrons chilling on an atom? Or a pi bond, which is basically two atoms sharing electrons a little too enthusiastically? That's your starting point for an arrow. That's where the arrow's tail should go. Got it? Good! Now, where is this electron-poor spot? Is there a positively charged atom hanging around, looking all forlorn and desperate for some electron love? Or maybe an atom that's bonded to more things than it probably should be, making it a bit strapped for electrons?
The tip of your curved arrow needs to point directly at that electron-deficient spot. That's where the electrons are going. It's like a tiny roadmap for our electron couriers. They start at the source, and they end up at the destination, delivering their precious cargo of negative charge. And the beauty of it? Sometimes, one arrow isn't enough! Rearrangements can be a whole sequence of electron movements. One arrow leads to another, like a domino effect, but way more elegant and with less potential for knocking things over.
Let's break down the common types of moves these arrows make. Sometimes, you'll see an arrow starting from a lone pair. That's like taking a couple of electrons that were just sitting pretty and saying, "Okay guys, time for an adventure!" And off they go, usually to form a new bond. It's a beautiful thing. Other times, the arrow will start from a pi bond. Pi bonds are inherently a bit more diffuse and reactive than sigma bonds, so they're like the readily available energy source for these movements. Think of it as tapping into the readily available snack drawer.
And where do these arrows end? Well, they usually point to form a new bond. This is where the action is! You're literally showing the formation of a new connection between atoms. But they can also point to a lone pair on an atom, showing that the electrons from a bond are now becoming a lone pair. This often happens when a bond breaks. It's all about conserving those electrons, keeping the total number the same, just redistributed. It’s like a magic trick, but the magic is real chemistry!
Now, a really important rule to remember, and this is key for our puzzle. You can't just move electrons from nowhere! You always need a source. A lone pair or a pi bond. And you can't create or destroy electrons, obviously. So, the number of electrons around each atom might change, but the overall count for the molecule stays the same. It's like you're taking apart a Lego castle and rebuilding it into a different, perhaps even cooler, structure. Same bricks, different design.
Think about the charges. If you start with a neutral molecule and move electrons, you’re going to end up with some charged species, right? A positive charge means an atom is a bit electron-shy, and a negative charge means it has a surplus. When you draw your arrow from a high-electron-density area to a low-electron-density area, you're essentially neutralizing or stabilizing those charges, or creating new ones that are more stable. It's like the universe's way of tidying up. Very satisfying when you get it right!
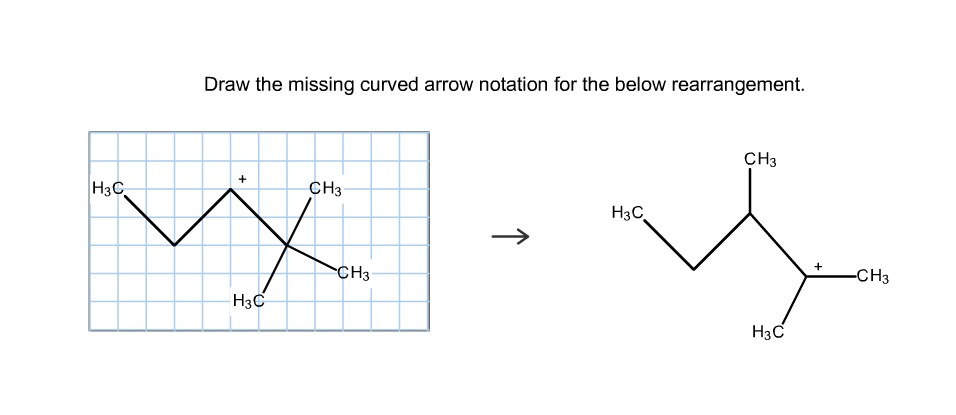
In our specific case, we're looking at a rearrangement where a carbocation is likely involved. Carbocations are those positively charged carbon atoms. They're like the little orphans of the molecule, desperately needing some electron attention. And often, they're not the most stable form. So, the molecule will often try to rearrange itself to form a more stable carbocation. That's the driving force behind many of these reactions. Stability is king, my friends!
So, where’s the most stable carbocation likely to form? Generally, tertiary carbocations (where the positive carbon is attached to three other carbons) are more stable than secondary (two other carbons), which are more stable than primary (one other carbon). It's like a hierarchy of awesomeness. So, if your molecule can shift things around to get a positive charge onto a more substituted carbon, it probably will. It’s just good sense, molecule-wise.
Now, let's get specific with our puzzle. You’ve got your starting molecule. Can you see a carbon that's positively charged, or perhaps a part of the molecule that could become positively charged if some electrons moved away? That's your target. And can you see a nearby group of atoms that has some extra electrons to spare? Maybe a lone pair on an oxygen or a nitrogen? Or even a pi bond that’s ripe for the picking?
The first arrow in a rearrangement like this often involves a group migrating. This means an entire atom or group of atoms, along with its bonding electrons, moves over to form a new bond. It’s like a whole little family moving house! So, you might see an arrow starting from a bond (often between the migrating group and the atom it's leaving) and pointing to the electron-deficient atom where it's going to land. This simultaneously breaks the old bond and forms the new one. Pretty neat, right? It’s a coordinated effort!

Once that group has moved, and a new bond has formed, what’s the charge situation now? Did you just get rid of one carbocation and create another, perhaps a more stable one? Or maybe you've just shifted a positive charge. The key is to look at the atom that the migrating group left behind. If it was part of a bond that just broke, and it's now left with fewer electrons than it started with, it might become positively charged. That's your cue for the next arrow!
Sometimes, after the initial migration, you might have a situation where a positive charge is still not in the most ideal spot. Or maybe a lone pair is now in a perfect position to attack something. This is where you get those sequential arrows. You might see an arrow starting from a pi bond in the newly formed molecule, attacking a positively charged atom. Or a lone pair swooping in to form a new bond. It’s a cascade of electron movement, all leading to the final, most stable product.
Let's think about the geometry, too. Curved arrows aren't just about electron numbers; they also imply a certain spatial arrangement. When a group migrates, it often does so in a way that keeps the transition state somewhat stable. It's like they're trying to move smoothly, not awkwardly bumping into things. Though, honestly, the visual representation of the arrow is usually enough. Don't get too lost in the molecular ballet just yet, unless you want to!
The beauty of drawing these arrows is that it forces you to think about the mechanism. You can't just guess. You have to follow the electron flow logically. It’s like being a detective, piecing together clues. The starting materials are your crime scene, the curved arrows are your witness testimonies, and the product is your solved case. And the reward? A deeper understanding of how molecules actually work. Pretty cool payoff for a little bit of drawing, right?
So, for our specific rearrangement, I want you to focus on where that positive charge is, or where it wants to be. Then, find the nearest source of electrons that can make that happen. Can you see a group that can "jump" over and take that positive charge? Or perhaps a pi bond that can "reach out" and stabilize it? Visualize that movement. Imagine the electrons leaping from their comfortable electron cloud to the needy, electron-starved atom. It’s a heroic act, really.
Remember, each arrow represents two electrons. That's a pair. So, when you see a pi bond, that's two electrons. When you see a lone pair, that's two electrons. And when you draw an arrow to form a new bond, you're essentially saying, "This atom is now sharing these two electrons with that atom." And if an arrow points to an atom and results in it gaining a lone pair, you're saying, "These two electrons are now exclusively yours, buddy!"
Don't be afraid to erase and redraw. It's part of the process! Sometimes, your first instinct might be slightly off. That's totally normal. The trick is to look at the product and work backward, or at least to ensure your arrows lead you logically to a plausible product. Does the final arrangement make sense in terms of stability? Are the charges distributed in a reasonable way?
Think about resonance structures, too. Sometimes, a rearrangement can lead to a molecule that can be stabilized by resonance. If you see a positive charge adjacent to a pi bond or a lone pair, that's a potential site for electron delocalization, which can be very stabilizing. The arrows you draw should reflect this possibility, or lead towards it.
So, take another sip of your coffee, look at the molecule, and channel your inner electron whisperer. Where do those precious negative charges want to go? What's the most logical path they can take to achieve a more stable configuration? It's all about following the energy. Molecules are lazy, in a good way. They want to be in the lowest energy state possible. And these rearrangements are their clever ways of getting there. You’ve got this! Let those arrows flow!
