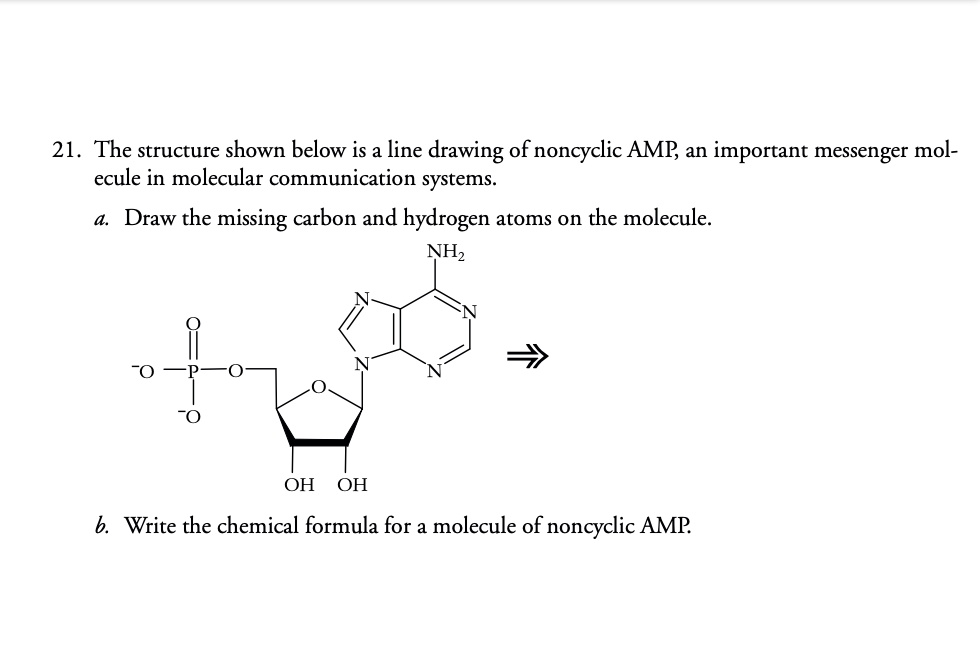
Draw The Missing Carbon And Hydrogen Atoms On The Molecule
Hey there, curious minds! Ever glanced at a molecule and felt like something was… well, missing? Like a puzzle missing a few crucial pieces? That’s exactly what we’re diving into today, and trust me, it’s way more fun than it sounds. We’re talking about drawing in those hidden carbon and hydrogen atoms on a molecule diagram. Sounds a bit like a treasure hunt, doesn’t it?
You see, chemists have this super cool shorthand. When they draw these intricate molecular structures, they often leave out the carbon and hydrogen atoms. Why? Because they’re the absolute backbone of organic chemistry! They’re like the trusty sidekicks in a superhero movie – always there, incredibly important, but sometimes get overshadowed. So, when you see a structure, it’s kind of like a sketch, and your job is to fill in the blanks, adding the characters that make the whole picture make sense.
Let’s break it down. Imagine a line segment in a molecular drawing. That line? It’s not just an empty space, oh no! That line represents a chemical bond. And at the end of that line, where it just stops, or where two lines meet, you can bet your bottom dollar there’s a carbon atom hanging out. Unless, of course, there’s a different atom explicitly written there, like an oxygen (O) or a nitrogen (N). But if it’s just a pointy bit or a junction? Bingo! Carbon!
And what about those hydrogen atoms? These guys are a little shy. They usually hitch a ride with carbon. A carbon atom, to be perfectly stable and happy (chemically speaking, of course!), likes to form four bonds. So, if you see a carbon atom that’s only shown to be connected to, say, two other atoms by lines, you know it’s got some unfinished business. Those missing bonds? They’re almost always filled with hydrogen atoms. You just count up how many more bonds the carbon needs to reach its magical number of four, and that’s how many hydrogens you draw!
Think of it like this: a carbon atom is a very social creature. It loves making friends, and it wants to have exactly four friends (bonds). If it’s already hanging out with a couple of other atoms (lines), it’ll be looking for more friends. Those are the hydrogens! They’re like the extra seats at a party, always filling in the gaps.
This might sound a little dry, but honestly, it’s where the magic of visualization really kicks in. When you start drawing in those missing atoms, you’re not just completing a diagram; you’re starting to see the molecule in its full glory. You’re building it in your mind, understanding its shape, and how it might interact with other molecules. It’s like going from seeing a silhouette to a fully rendered 3D model!
And this skill? It’s not just for super-smart scientists in lab coats (though they’re pretty cool too!). This is fundamental to understanding everything from the medicines you take, to the food you eat, to the materials that make up your world. Seriously, it’s the language of life!
Let’s try a little mental exercise. Imagine a simple shape, like a hexagon. In chemistry, a hexagon with alternating double bonds often represents benzene. If it’s drawn just as a hexagon with those double bonds, where are the carbons and hydrogens? Each corner of that hexagon? That’s a carbon atom, right? And how many bonds does each of those carbons have shown? Each carbon is connected to two other carbons. If it’s part of the ring and has a double bond, it’s already got three bonds accounted for. So, guess what? It needs one more bond! And that one more bond is usually to a hydrogen atom. So, in benzene, each carbon has one hydrogen attached. Voilà! You’ve just drawn in six carbons and six hydrogens without even picking up a pencil!
It’s a bit like learning a new language. At first, the grammar rules might seem a little odd, but once you get them, a whole new world of communication opens up. And in chemistry, that world is filled with incredible discoveries and fascinating processes. This simple act of drawing in missing atoms is your first step towards understanding those complexities.
Think about how much fun you can have once you master this! You can look at the structure of a vitamin C molecule and see all the atoms, understand its shape. You can look at a molecule of sugar and grasp how it fuels your body. It’s empowering! It transforms abstract diagrams into tangible building blocks of the universe. It makes the unseen, seen.

And it's not just about accuracy; it's about intuition. As you practice, you’ll start to develop a feel for how molecules are structured. You’ll anticipate where those carbons and hydrogens should be. It’s like learning to ride a bike – a bit wobbly at first, but soon it feels completely natural and incredibly liberating.
So, next time you encounter a molecular diagram, don’t be intimidated by the seemingly empty spaces. See them as invitations! Invitations to complete the picture, to understand the fundamental components, to unlock the secrets held within those lines and angles. Every corner is a carbon waiting for its friends, and every missing bond is a hydrogen ready to join the party.
This might seem like a small skill, but it’s a giant leap in your understanding of chemistry. It’s the gateway to a world where you can interpret, visualize, and even predict the behavior of matter. It’s a reminder that even in the simplest of representations, there’s a whole lot of fascinating structure and hidden detail waiting to be discovered. So go forth, draw those missing atoms, and let your scientific curiosity flourish. The molecules are waiting for you to bring them to life!
