Draw The Major Product S Of Nitration Of Benzonitrile
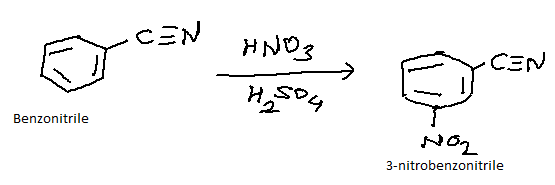
Hey there, future organic chemistry wizards! So, we're diving into the wacky world of electrophilic aromatic substitution today, specifically, the nitration of benzonitrile. Don't let the fancy words scare you; it's actually pretty straightforward, like figuring out which sock goes with which shoe (though arguably more predictable!).
Think of benzonitrile as a benzene ring chilling with a little cyano (-CN) group attached. This cyano group is our guest star, and it's going to play a huge role in where our incoming nitro group (-NO2) decides to park itself. It's like a picky landlord, this cyano group!
Before we get our hands (or molecules, rather) dirty, let's chat about what "nitration" even means. Basically, we're taking a benzene ring and sticking a nitro group onto it. The common recipe for this involves a mix of concentrated nitric acid (HNO3) and sulfuric acid (H2SO4). The sulfuric acid is like the wingman for nitric acid; it helps it get ready to do its job.
The sulfuric acid protonates the nitric acid, making it a super-duper electrophile – that's the fancy term for something that loves electrons. This potent little nitrosonium ion (NO2+) is the actual star of the show, ready to attack our electron-rich benzene ring.
Now, back to our benzonitrile. The cyano group (-CN) is interesting. It's what we call a meta-director. This means it's not the friendliest neighbor to the ortho and para positions on the benzene ring. Why? Well, the cyano group is electron-withdrawing. Imagine it's constantly tugging on the electrons in the benzene ring, making those ortho and para spots a bit less appealing for our electron-hungry nitrosonium ion.
So, where does that leave us? If the ortho and para positions are a bit like a "no soliciting" zone, then the meta position is the only place left for our nitro group to hang out. Think of it as the only available parking spot in a very popular (and slightly intimidating) neighborhood.
When we nitrate benzonitrile, the nitro group will preferentially attack the positions that are meta to the cyano group. On a benzene ring, the meta positions are the ones that are two carbons away from the substituent. So, if our cyano group is at position 1, the meta positions are 3 and 5. They're essentially mirror images of each other in terms of their electronic environment relative to the cyano group.

Therefore, the major product of the nitration of benzonitrile is 3-nitrobenzonitrile (or meta-nitrobenzonitrile). It’s the most stable and therefore the most favored product of the reaction.
Let's break down why this happens a little more. When the electrophilic nitrosonium ion approaches the benzonitrile molecule, it can theoretically attack at the ortho, meta, or para positions. However, the electron-withdrawing nature of the cyano group destabilizes the intermediate carbocations formed when the attack happens at the ortho and para positions. These intermediates have a positive charge closer to the electron-withdrawing cyano group, which is like trying to balance a heavy weight on a wobbly table – not ideal!
On the other hand, when the nitrosonium ion attacks at the meta position, the positive charge in the intermediate carbocation is further away from the electron-withdrawing cyano group. This makes the intermediate more stable. And in the world of organic chemistry, stability is king! Molecules always want to find the path of least resistance, the most stable arrangement.
So, while you might get tiny, almost undetectable amounts of ortho and para isomers, they're so negligible that we can pretty much ignore them when talking about the major product. It's like finding a single blue M&M in a bag of red ones – technically there, but you wouldn't call the bag "red and blue," would you? You'd just say it's a bag of red M&Ms.

The reaction conditions also play a role. We typically use strong acids and heat to drive this reaction. This provides enough energy for the nitrosonium ion to be generated and to overcome any activation energy barriers. But even with all this energy, the inherent electronic effects of the cyano group steer the reaction towards the meta position.
Think of it as a social gathering. The cyano group is the host, and it's not really keen on having guests right next to it (ortho) or directly across the room (para). It prefers people to mingle at a comfortable distance (meta). Our nitro group, the guest, is polite and respects the host's preferences. Voila! Meta-substitution.
So, to recap our little adventure: we have benzonitrile, a benzene ring with a cyano group. We're adding a nitro group using nitric and sulfuric acids. The cyano group is an electron-withdrawing bad boy, making it a meta-director. This means it tells the incoming nitro group, "Hey, the meta spots are where it's at!"
The result? The major product is 3-nitrobenzonitrile. You might also see it written as m-nitrobenzonitrile. The "m" stands for meta, just in case you were wondering. It’s a nice little shorthand!
Now, what if the question asked for all possible products? Well, in a strict sense, you could have small amounts of 2-nitrobenzonitrile (ortho) and 4-nitrobenzonitrile (para). But when the question says "major product," it's like asking for the headliner of a concert – you're interested in the star of the show, not the opening acts that played to an empty room.
Let's consider the structure of benzonitrile again. The benzene ring has six carbons. Let's number them starting from the carbon attached to the cyano group as carbon 1. The carbons next to it are carbons 2 and 6 (ortho). The carbons further away, but not opposite, are carbons 3 and 5 (meta). The carbon directly opposite is carbon 4 (para).
So, the cyano group at position 1 has two ortho neighbors (2 and 6), two meta neighbors (3 and 5), and one para neighbor (4). Because the cyano group is a meta-director, it "pushes" the incoming nitro group towards positions 3 and 5. Since positions 3 and 5 are chemically equivalent in this molecule (due to symmetry), we only get one major product, 3-nitrobenzonitrile.
It's like having two identical doors that lead to the same party room – you only count it as one entrance to the party, even though there are two physical doors. Pretty neat, huh?
The mechanism itself involves the formation of the sigma complex (or arenium ion) where the nitrosonium ion is attached to the benzene ring. The stability of these sigma complexes is what dictates the regioselectivity (where the substitution occurs). As we discussed, the sigma complexes formed from meta attack are more stable than those formed from ortho or para attack because the positive charge is delocalized onto carbons that are not directly adjacent to the electron-withdrawing cyano group.
This is a fundamental concept in organic chemistry, and understanding how substituents influence the reactivity and regioselectivity of electrophilic aromatic substitution reactions is super important. It's like learning the rules of the game before you start playing!
So, the next time you see benzonitrile and hear the word "nitration," you can confidently say, "Ah ha! That cyano group is a meta-director, so the nitro group is heading to the meta position. 3-nitrobenzonitrile, here we come!" You'll be like a seasoned chemist, effortlessly predicting the outcome. High fives all around!
It's amazing how these tiny molecules, with their specific electronic properties, follow these predictable patterns. It's like a complex dance choreographed by nature itself. And you, my friend, are learning the steps to this beautiful molecular ballet.
So, don't be intimidated by the big words or complex structures. With a little understanding of how substituents influence reactions, you can unlock the secrets of organic chemistry. Keep exploring, keep learning, and remember that every molecule has a story to tell – you just need to know how to listen!
And with that, you've conquered the nitration of benzonitrile! You've tamed the beast, understood the director, and predicted the product. Go forth and apply this knowledge, knowing that you've made significant progress in your chemical journey. The world of organic molecules is vast and exciting, and you're well on your way to exploring its wonders. Keep that curiosity alive, and you'll achieve amazing things!
