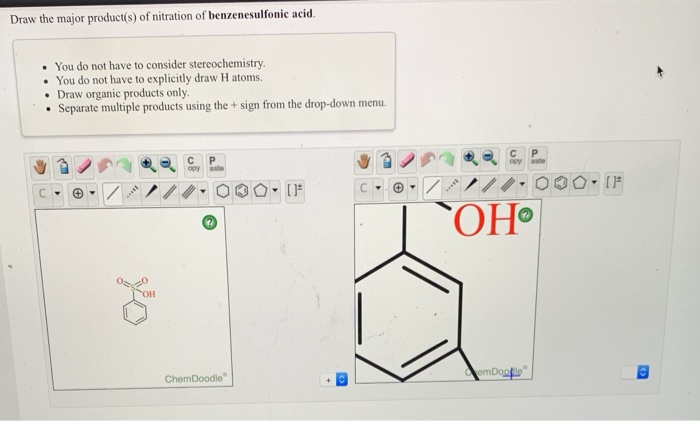
Draw The Major Product S Of Nitration Of Benzenesulfonic Acid
Hey there, chill vibes and chemical curiosity seekers! Ever looked at a molecule and wondered what kind of glow-up it’s going to have? Today, we’re diving into the fascinating world of organic chemistry, but with a laid-back, magazine-worthy spin. Think of this less like a stuffy lecture and more like a peek behind the curtain of how everyday things are made, with a sprinkle of science and a whole lot of fun. We’re going to unpack the nitration of benzenesulfonic acid, and trust me, it’s not as intimidating as it sounds. In fact, it’s a lot like understanding how your favorite influencer crafts their perfect Insta feed – there’s a method to the madness, and the results can be surprisingly… beautiful.
So, what exactly are we talking about? Imagine benzenesulfonic acid as a perfectly organized bookshelf. It’s got its foundational structure, its essential "books" in place. Nitration, on the other hand, is like adding a bold new bookmark or a striking cover sticker to those books. It’s a chemical reaction where a nitro group (-NO2) is introduced onto the benzene ring of benzenesulfonic acid. And the key word here? Major. We’re not just looking for any old outcome; we’re interested in the most likely, the most abundant products. It’s like asking, “What’s the most popular song on repeat right now?”
Before we get our hands dirty (metaphorically, of course – we’re keeping it clean and comfy here!), let’s get acquainted with our starting player: benzenesulfonic acid. Picture a benzene ring – that six-carbon, hexagonal beauty that’s the backbone of so many organic compounds. Now, attach a sulfonic acid group (-SO3H) to it. This group is pretty influential. It’s an electron-withdrawing group, meaning it pulls electron density away from the benzene ring. Think of it as the sensible friend in a group, the one who keeps things grounded. This subtle tug-of-war is crucial because it dictates where the incoming nitro group will decide to settle.
Now, let’s talk about the "nitrating agent." This is the party starter for our reaction. Typically, we use a mixture of concentrated nitric acid (HNO3) and concentrated sulfuric acid (H2SO4). Don't let the "concentrated" scare you; it just means they’re super potent and ready to get things done. Sulfuric acid, in this dynamic duo, acts as a catalyst. It helps to generate the actual nitrating species: the nitronium ion (NO2+). This little ion is the "nitro" part of our equation, the electrophile eager to find a new home on the benzene ring.
The benzene ring, as you might know, has certain "hot spots" or positions that are more or less attractive to incoming electrophiles. These positions are often referred to as ortho, meta, and para. Think of it like picking a seat in a popular cafe. Some spots are prime real estate, others are a bit more tucked away. The sulfonic acid group, being our sensible friend, directs the incoming electrophile to specific locations. Because it’s a meta-director, it guides the nitronium ion to the meta positions of the benzene ring. It’s like the group saying, "Nah, not here, not there… go to the third spot from here, it’s the sweet spot."
So, if we have one sulfonic acid group on the ring, and it's directing things to the meta positions, what does that mean for our products? It means the majority of the nitration will occur at the positions that are meta (or "third") relative to the sulfonic acid group. There are two such positions on a benzene ring, and they are chemically equivalent.
Therefore, the major product of the nitration of benzenesulfonic acid will be 3-nitrobenzenesulfonic acid (or, if you prefer the IUPAC naming convention, benzenesulfonic acid, 3-nitro-). This molecule has the nitro group sitting neatly at the meta position. Imagine our bookshelf again; it’s the book with the striking new cover sticker placed strategically on the third shelf down.
But wait, is it only 3-nitrobenzenesulfonic acid? In the world of chemistry, rarely is anything 100% perfect. While 3-nitrobenzenesulfonic acid is the undisputed heavyweight champion of this reaction, you might also find smaller amounts of the ortho and para isomers. Think of these as the runners-up, the songs that are popular but not the most popular. The ortho positions are adjacent to the sulfonic acid group, and the para position is directly opposite. These are formed in much lower yields because the sulfonic acid group is a strong meta-director.
The relative amounts of these isomers are influenced by the reaction conditions – temperature, concentration of acids, and reaction time. Chemists often tweak these variables to maximize the yield of their desired product. It’s like a chef adjusting the heat and spices to get the perfect flavor profile. For our purposes today, focusing on the major product, we’re really highlighting that 3-nitrobenzenesulfonic acid. It’s the star of the show, the one you’d bet your last dollar on.
Why is this important? Well, these nitro-substituted sulfonic acids are valuable intermediates in the synthesis of dyes, pharmaceuticals, and other fine chemicals. Think of the vibrant colors in your favorite clothes, or the life-saving medications in your cabinet – some of them have a lineage that includes reactions like this. It's a behind-the-scenes glimpse into how the colorful and complex world we live in is constructed, molecule by molecule.
Let’s break down the "why" of the meta-direction a little further. The sulfonic acid group is electron-withdrawing through two main mechanisms: induction and resonance. Inductively, it pulls electron density towards itself. Through resonance, it can also withdraw electron density from the ring. This makes the ortho and para positions less electron-rich and therefore less attractive to the electron-seeking nitronium ion. The meta positions, while also affected, are relatively more electron-rich compared to the ortho and para positions under these circumstances. It’s a bit like how certain parts of a room might be better lit than others depending on where the lamp is placed. The nitronium ion is looking for the brightest spots, and the sulfonic acid group is casting a bit of a shadow on the ortho and para positions.

Imagine you’re decorating your living room. You have a large, elegant rug (the benzene ring) and you want to place a statement lamp (the sulfonic acid group). You know that placing the lamp in a certain spot will create the best ambiance. The sulfonic acid group is like that lamp, influencing where the "light" (the nitro group) will shine brightest on the rug. It's all about directing the energy.
In a more modern context, think of social media algorithms. They direct content to you based on your past interactions, preferences, and the overall structure of the platform. The sulfonic acid group acts as a sort of "algorithm" for the incoming nitro group, dictating where it’s most likely to "engage."
So, to recap for our visual learners and those who appreciate a good summary: the major product of the nitration of benzenesulfonic acid is 3-nitrobenzenesulfonic acid. The nitro group (-NO2) attaches to the benzene ring at the position meta to the sulfonic acid group (-SO3H). The other products, 2-nitrobenzenesulfonic acid and 4-nitrobenzenesulfonic acid, are formed in significantly smaller amounts.
Let's add a fun fact: the discovery of these directing effects was a huge breakthrough in organic chemistry. It allowed chemists to predict and control the outcome of reactions, paving the way for the synthesis of a vast array of complex molecules. It’s like figuring out the secret recipe for success in the molecular world.

This knowledge is not just for lab coats and beakers. Understanding how substituents on a benzene ring influence reactivity is a fundamental concept that pops up in many areas. It's like learning basic grammar – once you know the rules, you can understand and even create complex sentences. In chemistry, these "sentences" can be the building blocks for everything from advanced materials to life-saving drugs.
Consider the process of making your morning coffee. The beans (our starting materials), the roasting process (our reaction conditions), and the brewing method (our directing effects) all contribute to the final flavor in your cup. The sulfonic acid group is like the specific way you roast your beans, setting the stage for the final flavor profile of your coffee – or in our case, the final molecular structure.
It’s fascinating to think about how these precise chemical arrangements lead to the properties of substances we encounter daily. The colors we see, the medicines we take, the materials we use – they all owe their existence to the controlled dance of atoms and molecules. The nitration of benzenesulfonic acid is just one small, albeit important, step in this grand chemical ballet.
And the beauty of it all is that even these seemingly complex reactions are governed by fundamental principles. It's about understanding the electron distribution, the push and pull of charges, and the inherent stability of different molecular arrangements. It’s a bit like understanding the psychology behind why certain trends become popular – there are underlying patterns and preferences at play.
For those who are a bit more hands-on, or just enjoy a good analogy, imagine you’re building with LEGOs. The benzene ring is your baseplate. The sulfonic acid group is like a specific connector piece you've already attached. When you go to add the next piece (the nitro group), you’re naturally going to look for the best place to connect it, and the existing connector piece will heavily influence your choices. It's not random; it's strategic!
In our case, the sulfonic acid group is a bit like a bulky, rule-following connector. It prefers the "middle" spots for the next connection, avoiding the tight spaces right next to itself (ortho) or the directly opposite spot (para) if there are more favorable meta spots available. This preference is all about minimizing steric hindrance (physical crowding) and optimizing electronic interactions.
So, next time you see a brightly colored fabric or a sleek piece of technology, remember the intricate chemical processes that made them possible. The nitration of benzenesulfonic acid, leading to our main star, 3-nitrobenzenesulfonic acid, is a testament to the elegance and predictability of organic chemistry. It's a reminder that even in the microscopic world, there's order, there are patterns, and there are beautiful, functional outcomes.
It’s a humbling thought, really. We’re all just complex arrangements of these very same building blocks. And understanding how these blocks interact, how they get modified, and what new structures they can form, gives us a deeper appreciation for the world around us. From the vibrant hues of a sunset to the intricate design of a smartphone, it all stems from the fundamental interactions of atoms and molecules. The nitration of benzenesulfonic acid is a small window into this vast and wondrous universe of chemical transformations.
This journey into the nitration of benzenesulfonic acid, focusing on the 3-nitrobenzenesulfonic acid as the major product, is more than just memorizing a chemical reaction. It’s about appreciating the guiding principles, the directing effects, and the elegance of molecular design. It’s about recognizing that even seemingly complex processes have underlying logic and predictable outcomes. And in our daily lives, isn't that what we're often seeking? To understand the patterns, to predict the outcomes, and to find that elegant simplicity in the midst of complexity? Whether it's navigating social dynamics or understanding the world around us, a little bit of chemistry can go a long way in helping us appreciate the intricate, yet often predictable, dance of existence.
