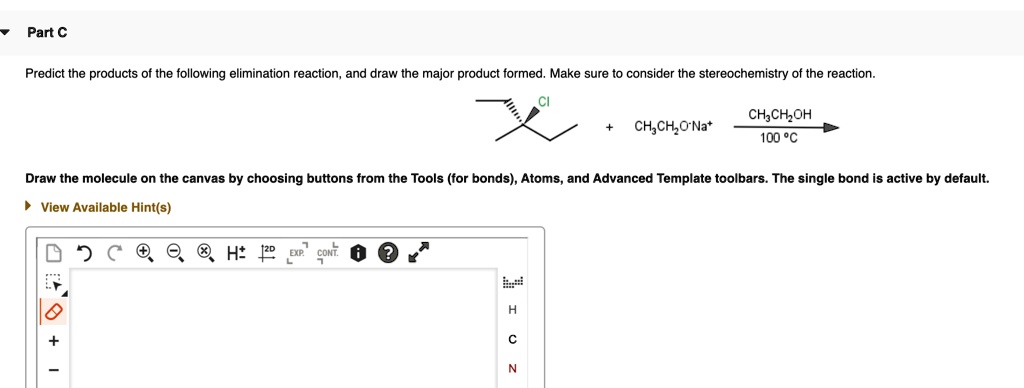
Draw The Major Product Of The Following Elimination Reaction

So, picture this: I’m crammed into my kitchen, surrounded by a whirlwind of flour, sugar, and a vague sense of panic. My mission? To bake the “perfect” chocolate chip cookie. Emphasis on the perfect. You know, the kind that’s chewy in the middle, crispy on the edges, and bursts with chocolatey goodness? Yeah, that kind. I’ve followed the recipe to a tee – or so I thought. I’ve measured, I’ve mixed, I’ve even creamed the butter and sugar, whatever that means in terms of actual magic.
But then comes the crucial step: the baking. The recipe gives instructions, of course, but it doesn’t exactly hand-hold you through the subtle nuances. Is it exactly 10 minutes? Does a slightly browned edge mean it’s done, or is it still a little too… doughy? It’s a delicate balance, a scientific experiment in my own oven, and frankly, sometimes I end up with a batch of vaguely cookie-shaped charcoal briquettes. And other times? Pure, unadulterated cookie bliss. It’s like a culinary gamble, every single time.
This whole cookie saga got me thinking about chemistry, specifically about reactions where the outcome isn't always as straightforward as you'd hope. You know, those moments in the lab where you're trying to coax a molecule into doing what you want, and it’s got a mind of its own. Today, we’re diving into one of those scenarios: an elimination reaction. Think of it as chemistry’s slightly more chaotic cousin to substitution reactions.
Elimination: When Molecules Break Up (and Form Something New!)
In a nutshell, an elimination reaction is all about removing atoms or groups of atoms from a molecule. It’s like a chemical breakup, where two things decide to leave the party, and in their wake, something else forms – often a double bond or a triple bond. Pretty neat, right? Instead of one thing swapping places with another (like in substitution), we’re essentially clearing out some space and letting the remaining atoms get a bit more cozy.
There are a few different types of elimination reactions, but the one we're focusing on today is pretty classic. We've got a starting molecule – let's call it our "substrate." This substrate usually has a leaving group (think of it as the molecule’s baggage, ready to be jettisoned) and a beta-hydrogen (a hydrogen atom on the carbon next to the carbon bearing the leaving group). Our job, with the help of a reagent (usually a base), is to convince both the leaving group and that beta-hydrogen to pack their bags and leave.
When they bail, the carbon atoms they were attached to decide to form a new pi bond. Boom! A double bond. It's a bit like a dance where two people leave the dance floor, and the remaining two decide to get really close and form a special connection. Chemistry is romantic, who knew?
The Tale of the Major Product: Zaitsev's Rule to the Rescue!
Now, here's where things can get a little… interesting. Sometimes, there's more than one beta-hydrogen available for elimination. Imagine our substrate has multiple potential "dance partners" for the leaving group to leave with. This means we could potentially form multiple different alkene products. And this, my friends, is where things get exciting (and sometimes a bit confusing if you’re not paying attention!).
This is like having several different directions you could go on a hike. You can see the same mountain peak from a few different trails. Which one do you choose? Well, in chemistry, there’s a guiding principle, a sort of seasoned hiker’s wisdom that helps us predict which product is more likely to be formed. It's called Zaitsev's Rule (sometimes spelled Saytzeff's Rule, because old-school science names can be a bit… flexible).
![[GET ANSWER] draw the structures of the major products in each of the](https://cdn.numerade.com/ask_images/bd51582171d8494f887e416027c26ed3.jpg)
Zaitsev’s rule basically states that in an elimination reaction, the more substituted alkene is generally the major product. What does "more substituted" mean? It means the alkene with the most alkyl groups (those pesky carbon chains) attached to the carbons involved in the double bond. Think of it as the alkene that's the "most popular" in terms of its neighbors.
Let's break that down with a hypothetical example. Imagine you have a molecule where the leaving group can leave from one carbon, and there are beta-hydrogens on two different adjacent carbons. One of those adjacent carbons might lead to a double bond where one of the carbons is attached to, say, two other alkyl groups. The other adjacent carbon might lead to a double bond where one of the carbons is only attached to one alkyl group.
According to Zaitsev, the one with the more alkyl groups attached to the double bond carbons is the one that's going to be the major player. It’s the more stable alkene, and therefore, it’s the one that forms more readily. The other, less substituted alkene, will still form, but it’ll be the minor product. It’s like the less popular trail; it exists, but not as many people take it.
Applying Zaitsev's Rule: Let’s Get Visual!
Okay, so we’ve talked the talk. Now, let’s walk the walk. Imagine we have the following molecule: (For the sake of this article, I can't actually draw it out for you, but I'll describe it so you can visualize or sketch it yourself. This is where you’ll want a pencil and paper handy, trust me!) Let’s say we have a secondary alkyl halide. Specifically, let’s consider 2-bromobutane.
So, we have a four-carbon chain. On the second carbon, there’s a bromine atom (our leaving group). Now, let’s look at the carbons adjacent to the carbon with the bromine. That’s carbon number 1 (a methyl group, -CH3) and carbon number 3 (a methylene group, -CH2-).
Carbon number 1 has three hydrogens. Carbon number 3 has two hydrogens. Both of these are our potential beta-hydrogens. Our base is going to come in and snatch one of these hydrogens, and the bromine is going to go, "Peace out!"
Now, let's see what happens depending on which beta-hydrogen is removed:
- Scenario 1: The base removes a hydrogen from carbon number 1.
If a hydrogen leaves from carbon 1, and the bromine leaves from carbon 2, what happens? The bond between carbon 1 and carbon 2 will form a double bond. This would result in but-1-ene. Let’s look at the substitution of this double bond. The carbons involved in the double bond are carbon 1 and carbon 2. Carbon 1 is attached to two hydrogens. Carbon 2 is attached to one hydrogen and the rest of the chain (carbon 3 and 4, which are CH2-CH3).
So, the double bond in but-1-ene looks like this: CH2=CH-CH2-CH3. The carbons in the double bond are the CH2 and the CH. The CH2 has two hydrogens. The CH has one hydrogen and is attached to an ethyl group (-CH2CH3). So, in terms of alkyl groups directly attached to the double bond carbons, we have one ethyl group. This makes it a monosubstituted alkene (if we consider the ethyl group as one "substituent").
- Scenario 2: The base removes a hydrogen from carbon number 3.
If a hydrogen leaves from carbon 3, and the bromine leaves from carbon 2, what happens? The bond between carbon 2 and carbon 3 will form a double bond. This would result in but-2-ene. Let’s look at the substitution of this double bond. The carbons involved in the double bond are carbon 2 and carbon 3. The structure is CH3-CH=CH-CH3. The carbons in the double bond are the two central CH groups.

Each of these central CH groups is attached to one methyl group (-CH3) and one hydrogen. So, we have two methyl groups attached to the carbons forming the double bond. This makes it a disubstituted alkene.
The Verdict: Who Wins the Elimination Race?
Now, let’s bring in Zaitsev’s rule! We have two potential products: but-1-ene (monosubstituted) and but-2-ene (disubstituted).
According to Zaitsev’s rule, the more substituted alkene is the major product. In our case, but-2-ene is the disubstituted alkene, and but-1-ene is the monosubstituted alkene. Therefore, but-2-ene is the major product of the elimination reaction of 2-bromobutane.
It’s the more stable molecule, and thus, it’s the one that the reaction "prefers" to make. It’s like that perfectly baked cookie – it’s just… better, more satisfying, and therefore, more likely to be the result when conditions are right. The minor product, but-1-ene, will still be formed, but in smaller quantities.
Now, you might be thinking, “What about the cis and trans isomers of but-2-ene?” Ah, excellent question! But-2-ene can exist as cis-but-2-ene (where the methyl groups are on the same side of the double bond) and trans-but-2-ene (where the methyl groups are on opposite sides). Generally, the trans isomer is even more stable than the cis isomer due to less steric hindrance. So, if we were being super precise, trans-but-2-ene would likely be the most abundant product, followed by cis-but-2-ene, and then the minor product, but-1-ene. But for the purpose of identifying the major alkene type, Zaitsev’s rule focusing on the degree of substitution is our primary tool.

When Zaitsev Takes a Backseat: The Hofmann Elimination
Now, before you get too comfortable with Zaitsev ruling the roost, it's important to know that there are exceptions! Sometimes, the less substituted alkene is actually the major product. This happens in what's called a Hofmann elimination. This usually occurs when you have a very bulky base or when the leaving group is poor (like a quaternary ammonium salt).
Think of it this way: if the base is really big and clumsy, it might struggle to reach for that beta-hydrogen that’s in a more crowded spot (which is often the one that leads to the more substituted alkene). It’ll go for the easier-to-reach hydrogen on the less substituted side. It’s like trying to find a parking spot in a crowded city – sometimes you have to settle for the one that's further away but easier to get into.
So, while Zaitsev’s rule is our go-to for most standard elimination reactions, it’s always good to keep an eye out for those special conditions that might favor the Hofmann product.
The Takeaway: Predicting the Unexpected
Understanding elimination reactions and Zaitsev’s rule is a super powerful tool in your organic chemistry arsenal. It allows you to predict the major product of a reaction, which is crucial for planning syntheses and understanding reaction mechanisms. It’s like having a cheat sheet for molecular breakups!
So, next time you’re presented with an elimination reaction, remember to:
- Identify the leaving group.
- Identify all the beta-hydrogens.
- Determine the alkene products that could form from the removal of each beta-hydrogen.
- Apply Zaitsev’s rule: the more substituted alkene is generally the major product.
It’s not always as straightforward as a cookie recipe, but with a little practice and a solid understanding of the rules, you’ll be predicting major products like a seasoned pro. And hey, if all else fails, at least you’ll know what to aim for when you’re trying to bake those perfect cookies – the ones that are just right, not too burnt, not too doughy, but just so. Happy eliminating!
