Draw The Major Organic Product Of The Sn1 Reaction:
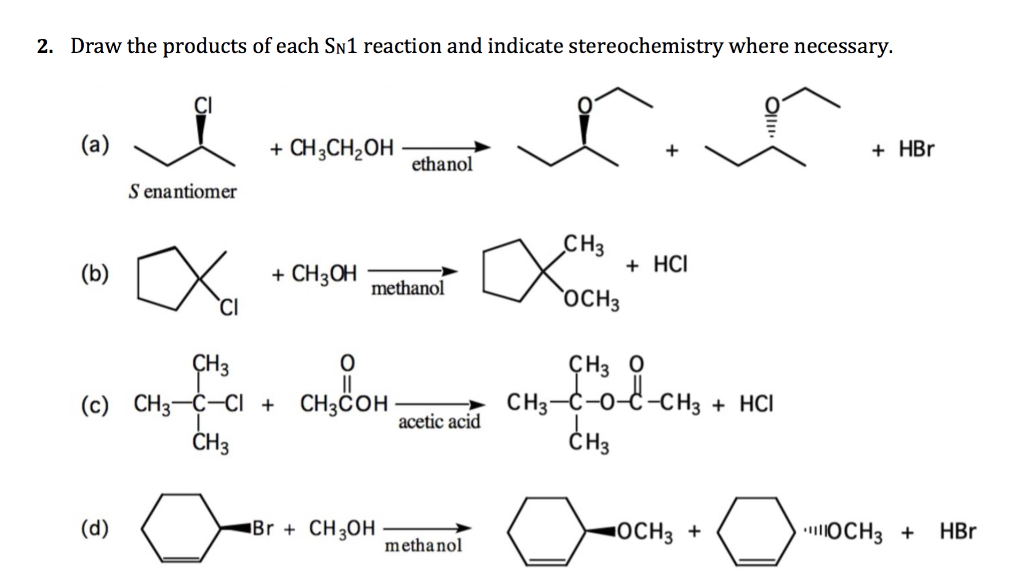
Ever wondered what happens when molecules decide to shake things up in a chemical reaction? If you're nodding along, you're in for a treat! Today, we're diving into the exciting world of SN1 reactions, a process that's as fascinating as it is fundamental in organic chemistry. Think of it as a molecular dance where bonds break and new ones form, leading to some pretty cool transformations. It’s a topic that pops up frequently in chemistry classrooms and labs, and for good reason – understanding it unlocks a deeper appreciation for how the building blocks of matter rearrange themselves.
The beauty of studying SN1 reactions lies in their predictability and the valuable insights they offer into reaction mechanisms. When you’re asked to draw the major organic product of an SN1 reaction, you’re essentially being challenged to become a molecular detective. You’re not just memorizing facts; you’re applying a set of logical steps to predict the outcome of a chemical transformation. This skill is incredibly useful, not only for acing chemistry exams but also for anyone involved in fields like pharmaceuticals, materials science, or even environmental chemistry, where understanding how molecules interact is key.
The SN1 Dance: A Step-by-Step Ballet
So, what exactly is this "SN1" all about? The "SN" stands for nucleophilic substitution, meaning a nucleophile (an electron-rich species that loves to donate electrons) replaces a leaving group (an atom or group of atoms that readily departs from a molecule). The "1" signifies that the rate-determining step (the slowest step in the reaction) involves only one molecule. This is a crucial distinction from SN2 reactions, which involve two molecules in their rate-determining step.
The SN1 reaction typically proceeds in two main steps. The first, and slowest, step is the dissociation of the substrate. The bond between the carbon atom and the leaving group breaks heterolytically, meaning the leaving group takes both electrons from the bond. This results in the formation of a positively charged carbocation intermediate. This carbocation is planar, with the positive charge residing on the carbon atom.
The formation of the carbocation is the pivotal moment in an SN1 reaction. Its stability heavily influences the reaction rate.
The stability of this carbocation is paramount. Tertiary carbocations are the most stable, followed by secondary, and then primary. This is because the alkyl groups attached to the positively charged carbon can donate electron density, helping to delocalize and stabilize the positive charge. This is why SN1 reactions are most common with tertiary and secondary substrates.

The second step is the attack of the nucleophile on the carbocation. Since the carbocation is planar, the nucleophile can attack from either face of the plane. This leads to the possibility of forming stereoisomers, specifically a mixture of enantiomers if the starting material was chiral. This phenomenon is known as racemization, where a 50:50 mixture of R and S configurations is often observed. However, depending on the solvent and reaction conditions, there might be a slight preference for one enantiomer over the other due to ion pairing effects with the departing leaving group.
Predicting the Major Product: Becoming a Molecular Maestro
When you're presented with an SN1 reaction scenario and asked to draw the major organic product, follow these key principles:

- Identify the Leaving Group: The first step is to spot the atom or group that is most likely to depart. Good leaving groups are typically weak bases, like halides (Cl-, Br-, I-) or tosylates.
- Form the Carbocation: Imagine the leaving group breaking away, taking its electrons, and leaving behind a positively charged carbon – the carbocation. Note its structure and hybridization.
- Consider Carbocation Rearrangements: Carbocations are highly reactive intermediates. If a more stable carbocation can be formed through a hydride shift or an alkyl shift, this rearrangement will likely occur. This is a crucial step in predicting the major product, as rearrangements often lead to more stable intermediates and thus favored products.
- Nucleophilic Attack: Now, bring in the nucleophile. It will attack the positively charged carbon of the carbocation (or the rearranged carbocation). Remember, the nucleophile can attack from either side of a planar carbocation.
- Determine Stereochemistry (if applicable): If the carbon that forms the carbocation is a stereocenter, the attack of the nucleophile can lead to a mixture of stereoisomers. For SN1 reactions, a racemic mixture (equal amounts of both enantiomers) is common, although partial inversion or retention of configuration can sometimes be observed.
For instance, if you have a tertiary alkyl halide reacting with a weak nucleophile in a polar protic solvent (like water or alcohol), you can expect an SN1 reaction to occur. The leaving group will depart, forming a tertiary carbocation. This carbocation is relatively stable. Then, the nucleophile will attack. If the carbon was also part of a chiral center, you’d anticipate a racemic mixture of products. If there was an opportunity for a rearrangement to form an even more stable carbocation (e.g., going from a secondary to a tertiary carbocation), that rearrangement would definitely happen before the nucleophile attacks.
Mastering the art of drawing the major organic product of an SN1 reaction is a rewarding journey. It involves understanding the nuances of carbocation stability, the possibility of rearrangements, and the stereochemical outcomes. So, the next time you encounter an SN1 reaction, embrace the challenge – you're about to orchestrate a fascinating molecular transformation!
