Draw The Major Organic Product Of The Reaction. Ignore Stereochemistry.

Hey there, fellow science explorers! Ever looked at a bunch of letters and squiggles, you know, like CH3CH2OH + O2, and thought, "What in the world is HAPPENING here?" Well, buckle up, buttercup, because we're diving into the wonderfully weird world of organic chemistry reactions! And guess what? We're going to draw the major organic product. Sounds fancy, right? But it's really just about figuring out what cool new thing pops out at the end of the chemical party.
And here's the super fun part: we're gonna ignore stereochemistry. What's stereochemistry? Think of it like the 3D arrangement of atoms. For our purposes today, we're keeping it simple. We're not worrying about which way a little wiggle points. We're just focusing on the big picture, the main ingredient that gets formed. It's like baking a cake – we care about the final deliciousness, not exactly how the flour particles are stacked. Though, of course, in real chemistry, that stuff matters a lot! But for our chat, let's keep it light and breezy.
Why is this even cool?
Because it's like a chemical puzzle! You start with some ingredients, and with a little know-how, you can predict the masterpiece that will emerge. It’s like having a secret decoder ring for molecules. How awesome is that?
Think about it. These reactions are happening all the time. In your body, in the air, in the food you eat. They’re the building blocks of everything! And learning to predict the outcome? That’s like having a backstage pass to the universe's coolest show.
Plus, let’s be honest, sometimes those chemical names and formulas look like they were invented by a supervillain. But once you break them down, it’s like, "Oh! That's not so scary after all!" It’s all about patterns and logic. And who doesn’t love a good pattern?
Let’s get our drawing hands ready!
So, how do we actually draw the product? It’s all about understanding what’s going on. We've got reactants – that's the stuff we start with. And then we’ve got products – the stuff we end up with. It's like a recipe: flour, sugar, eggs go in; cake comes out.
The trick is to know what the "cooking" part does. What are the rules of the chemical kitchen? There are different types of reactions, each with its own personality. Some are about adding things together. Others are about breaking things apart. Some are about swapping partners. It's like a molecular dating game!
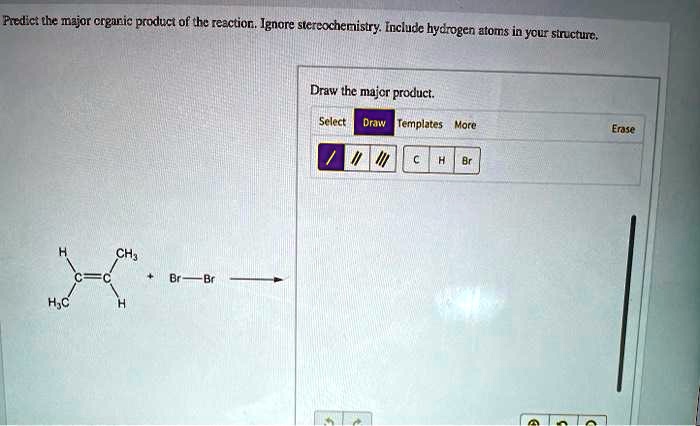
And since we're ignoring stereochemistry, we can just focus on the core structure. Imagine you're building with LEGOs. You've got different colored bricks, but today, we're just concerned with which bricks connect and where they go in the overall structure. We’re not fussing about the exact angle of the connection, just that it is connected.
Let’s take a super simple example, just for kicks. Imagine you have a molecule that looks like a tiny, happy chain. And you add something that wants to break that chain in half. What do you get? Two smaller chains! See? No need for fancy 3D drawings, just the basic shapes of what’s left. Simplicity is key!
The Nitty-Gritty (but not too gritty!)
When we talk about drawing the major organic product, we’re essentially asking: "What’s the most likely thing to form?" Nature, much like us, often likes the path of least resistance. So, reactions tend to follow predictable routes. They’ll form the most stable or the most abundant product.
It’s like when you’re tidying up your room. You’re not going to meticulously place every sock in its exact perfect orientation. You’re going to shove them in the drawer. That’s the major product of your tidying efforts – a drawer full of socks!
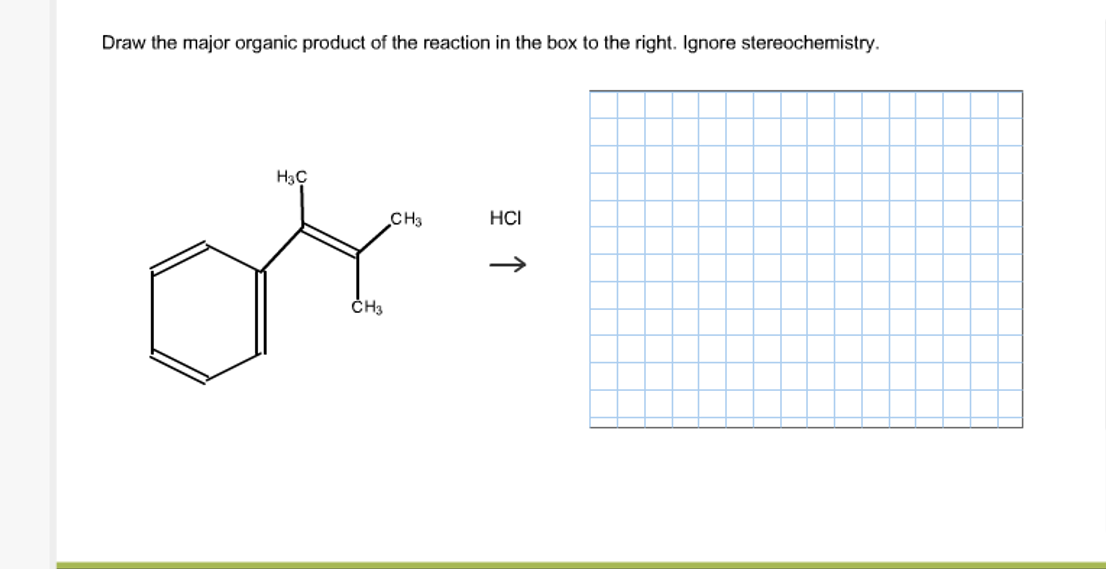
In organic chemistry, certain atoms love to bond with other specific atoms. Oxygen wants electrons. Carbon is pretty flexible and likes to bond with lots of things. Hydrogen is a bit of a tag-along. Understanding these little quirks is your superpower.
And sometimes, you'll see little symbols above or below the reaction arrow. These are like secret instructions. Maybe it means "add heat" or "use this special catalyst." They’re the chef’s special notes. We don’t need to memorize them all for today, but it’s good to know they exist and they’re telling us something important.
A Peek at the Chemical Playground
There are some classic reaction types that are super common. You’ve got addition reactions, where two molecules join to make a bigger one. Like two friends hugging. So sweet!
Then there are elimination reactions. This is where a small molecule, like water or ammonia, peaces out from a larger one, often leaving behind a double bond. It’s like someone leaving a party, and the remaining guests get closer.
Substitution reactions are also a big deal. One atom or group gets swapped out for another. Think of it as a musical chairs game for atoms. Someone loses their seat, and someone else takes it.

And don't forget redox reactions! These involve the transfer of electrons. It’s like a sharing economy for electrons. Some molecules gain electrons, others lose them. It’s all about the electron hustle.
Each of these reaction types has its own set of rules for predicting what happens. And once you get the hang of it, it’s like unlocking a new level in a video game. Suddenly, those squiggly lines start to make sense!
Making it Fun, Not Frightening
So, how do we get better at this? Practice, practice, practice! And the best practice? Drawing! Grab some paper, grab some pens, and start sketching. Don’t be afraid to make mistakes. Mistakes are just detours on the road to discovery.
Imagine each atom is a little character. Give them personalities. What do they like? What do they hate? What are they looking for in a chemical partner? This makes it way more engaging than just staring at abstract symbols.
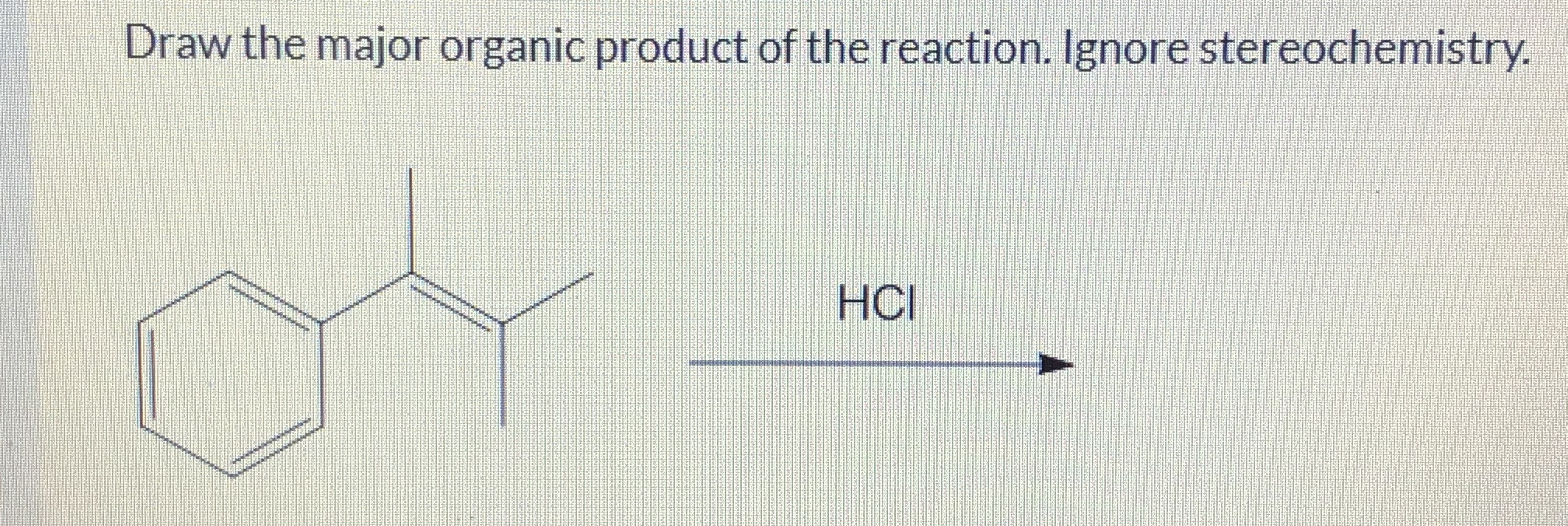
And when you’re working on a specific problem, ask yourself: "What’s the main thing this reaction is trying to do?" Is it trying to make things bigger? Smaller? Swap bits around? Focus on that core purpose.
Remember, we’re ignoring stereochemistry. This means you can draw your carbons and hydrogens in the most convenient way possible on the page. No need to worry about wedges and dashes. Just get the connections right. It’s the difference between drawing a stick figure and trying to render a photorealistic portrait – we’re going for the stick figure today, but a structurally accurate stick figure!
Think of the molecules as having sticky bits and not-so-sticky bits. The sticky bits want to connect! And the reaction conditions – like heat or a catalyst – are like the extra energy or the special handshake that helps those sticky bits find each other.
The "major" part of "major organic product" is also important. Sometimes, a reaction could make a few different things. But usually, one product is way more common, way more stable, or just easier to form. That's our main event. The rockstar of the reaction!
So, next time you see a chemical reaction, don't just see a jumble of letters. See a story waiting to unfold. See a puzzle with a satisfying solution. See a chance to predict the outcome of a molecular dance. And most importantly, have fun drawing that major organic product! It’s your chance to be a chemical artist, even if it’s just with a pen and some paper. Keep exploring, keep drawing, and keep enjoying the amazing world of chemistry!
