Draw The Major Organic Product For The Reaction Conditions Shown.

Ever looked at a bunch of ingredients and thought, "What on earth is this going to turn into?" That's pretty much what we're diving into today, but instead of a questionable casserole, we're talking about organic chemistry. Think of it as the ultimate culinary experiment, where molecules are the ingredients and the reaction conditions are your oven temperature, cooking time, and secret spice blend. Our mission, should we choose to accept it (and trust me, it's a lot less perilous than trying to assemble IKEA furniture), is to predict the major organic product of a given set of reaction conditions. It’s like being a kitchen detective, but instead of fingerprints, we're looking for clues in the molecular structure.
So, what is a "major organic product"? Imagine you're baking cookies. You put in flour, sugar, butter, eggs. Ideally, you get a delicious cookie. But sometimes, you might get a slightly burnt edge, or maybe one cookie spreads out more than the others. The cookie is your major product. The burnt edge or the flat one? Those are the minor products, the quirky cousins of the main event. In organic chemistry, we're aiming for the star of the show, the molecule that the reaction most enthusiastically wants to create.
Let's break down the "reaction conditions." These are basically the chef's instructions. They tell us what tools we have (like reagents – think of them as your special kitchen gadgets), what kind of heat we're applying (temperature), and how long we're letting things simmer (reaction time). Sometimes, there's a bit of a catalyst in there too, like a helpful sous chef who speeds things up or points the way without getting directly involved in the final dish. Understanding these conditions is key to figuring out what’s going to pop out at the end.
The Art of Molecular Prediction: It's Not Magic, It's Just... Chemistry
Now, some of you might be thinking, "Predicting molecules? That sounds like trying to guess what your cat is thinking. Impossible!" But it’s actually more like following a recipe. You learn the basic cooking techniques, and then you can adapt them. In chemistry, we learn the basic rules of how molecules like to interact. They have preferences, you see. Some are shy and prefer to hang out in groups (like ionic compounds), while others are more outgoing and love to share electrons (covalent compounds). Organic chemistry, specifically, deals with molecules built around carbon atoms, which are like the master builders of the molecular world, able to form all sorts of intricate structures.
When we're given a starting molecule (our initial ingredients) and some reaction conditions, we're essentially asking: "What's the most likely way these molecules will rearrange themselves to become more stable or achieve a preferred configuration?" It’s a bit like how you might tidy your room. You don't just randomly throw things around; you put similar items together, things that are used frequently go where they're accessible. Molecules do something similar, seeking out lower energy states, which is their version of being “tidy” and content.
The Players in Our Molecular Kitchen: Reagents and Catalysts
Let’s chat about the "reagents" for a sec. These are the things we add to our starting material to make the reaction happen. Think of them as the spices, the sauces, the special marinades. Some reagents are mild, like adding a pinch of salt to enhance flavors. Others are more potent, like a dash of chili powder that completely changes the game. For instance, you might see something like "acid" or "base" listed. Acids are like the sour notes in your recipe, while bases are the bitter or alkaline ones. They have a way of donating or accepting little particles called protons (H+), and this can be a big deal for how other molecules behave.
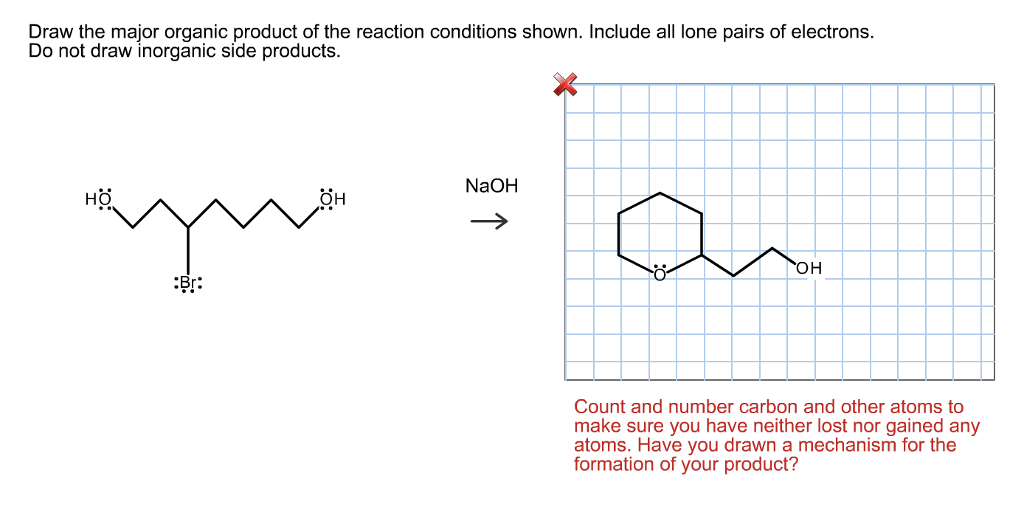
Then there are the "catalysts." These are the unsung heroes of many chemical reactions. They're like the helpful friend who shows you how to perfectly fold a fitted sheet, or the recipe tip that says, "Bake at this temperature for this long, trust me." Catalysts speed up reactions or allow them to happen under milder conditions without being consumed themselves. They're the backstage crew of the molecular theater, making sure the show runs smoothly without ever stepping into the spotlight as a main performer. If you see a catalyst, it’s a big hint that the reaction is designed to be efficient.
Spotting the Clues: What to Look For in Your Molecular Recipe
So, how do we actually do this prediction thing? It’s all about recognizing patterns and understanding the basic personalities of different functional groups (these are specific arrangements of atoms within a molecule that give it particular properties). Think of functional groups like different types of kitchen appliances. You have your oven (a way to add heat), your mixer (a way to combine things vigorously), and your blender (a way to break things down). Each functional group has its own set of reactions it’s prone to.
One of the most common scenarios involves something called a nucleophile. Don't let the fancy name scare you! A nucleophile is basically a molecule or a part of a molecule that is electron-rich. Think of it as someone who has an abundance of spare change and is always looking to share or spend it. Nucleophiles love to attack areas that are electron-deficient, like a magnet being drawn to metal. They have negative charges or lone pairs of electrons that are just itching to form a new bond. You can often spot them by looking for atoms with negative charges, or atoms like oxygen or nitrogen that have unshared pairs of electrons.
On the flip side, we have electrophiles. These are the opposite – they are electron-deficient. Think of them as someone who is perpetually short on cash and looking for a handout. They have a positive charge or a partial positive charge, making them attractive targets for our electron-rich nucleophiles. Where do you find these guys? Often, they’re associated with atoms that are bonded to very electronegative elements (like oxygen or halogens), which pull electron density away from them, making them hungry for electrons. The carbon atom in a carbonyl group (C=O) is a classic electrophile.
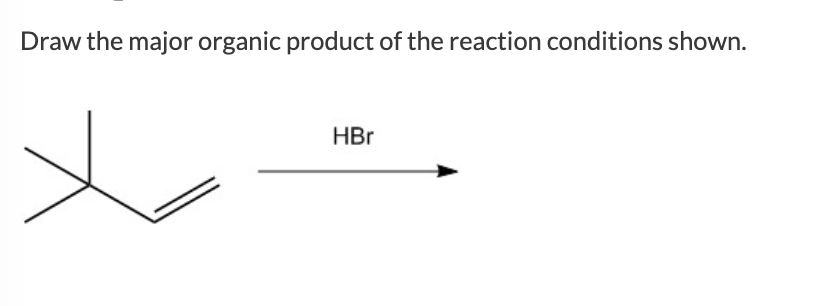
When a nucleophile and an electrophile meet, especially under the right conditions (like the presence of a good leaving group – a part of the molecule that's happy to depart once a new bond forms), you often get a substitution reaction. This is like swapping out an ingredient. Imagine your recipe calls for sugar, but you accidentally grab salt. You realize your mistake and swap the salt for sugar. In a substitution reaction, one group is replaced by another. It’s a very common and straightforward way for molecules to rearrange.
Another big player is elimination reactions. These are a bit more dramatic. Instead of swapping one thing for another, you're essentially removing two groups from adjacent atoms, often creating a new double or triple bond in the process. Think of it like taking two things out of your pantry to make space for a new, larger appliance. You’re sacrificing those two items to gain something new. Elimination reactions usually happen when you have a good base present, which can help "pull off" a proton, and a good leaving group ready to depart. This often happens when there are too many groups crowded around and the molecule wants to simplify and become more rigid with a double or triple bond.
Then there are addition reactions. These are like adding toppings to a pizza. You start with your base (often a molecule with a double or triple bond, which is like a slightly stretchy or less rigid structure), and you add new atoms or groups across that bond. The double or triple bond "opens up" to accommodate the new additions, turning into a single bond. These are particularly common when you have reagents that can easily break apart and add to the carbons of the double or triple bond, like halogens (think chlorine, bromine) or hydrogen halides (like HCl).
Putting It All Together: A Mini-Workout
Let's try a little mental exercise, shall we? Imagine we have a molecule with a hydroxyl group (-OH) attached to a carbon. And our reaction conditions involve treating it with a strong acid, like concentrated sulfuric acid (H2SO4), and heating it up. What do you think is going to happen?
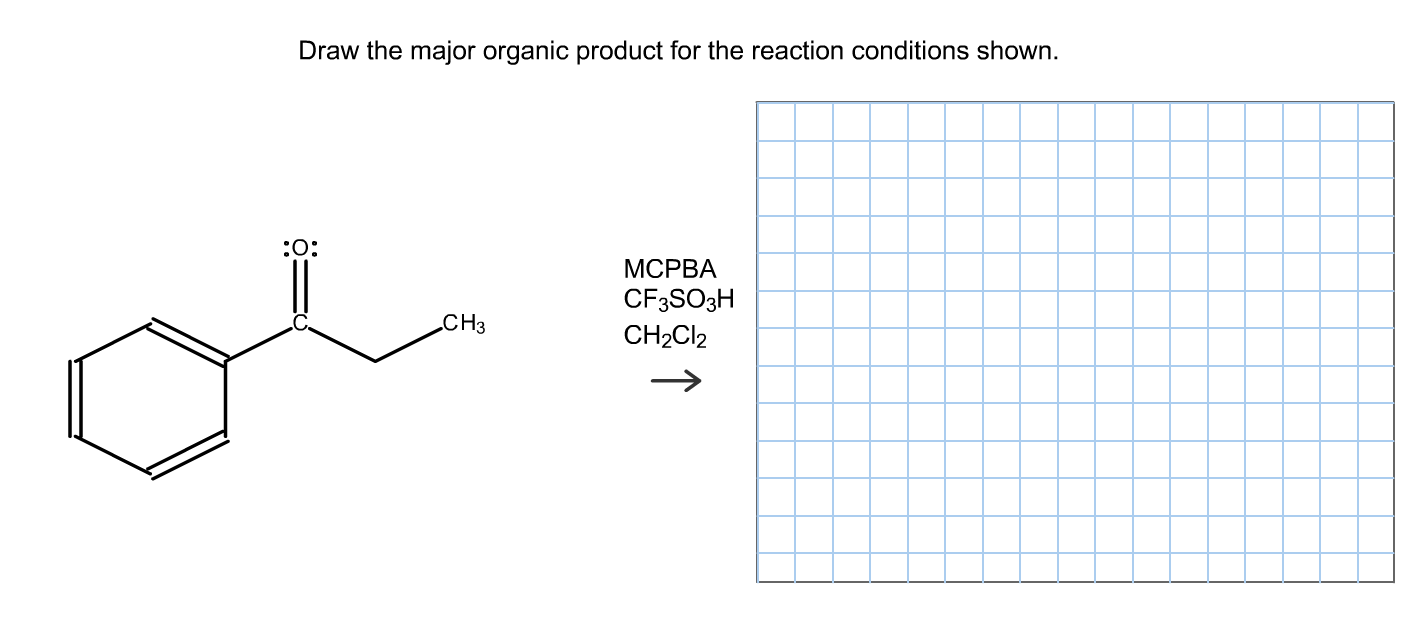
Okay, deep breaths. First, we have the hydroxyl group. On its own, it's not a great leaving group – oxygen doesn't like to leave without a good reason. But here's where the strong acid comes in. The oxygen atom in the -OH group has those lovely lone pairs of electrons (remember our nucleophile analogy? Oxygen is often a bit electron-rich). The acid's protons (H+) are like little positive charges looking for electron density. So, the oxygen grabs a proton, turning the -OH into -OH2+. Now, that + charge on the oxygen is a bit unhappy. What's even better is that water (H2O) is a fantastic leaving group. It's very stable on its own.
So, step one: Protonation of the -OH group. Step two: The -OH2+ group, now a great leaving group, decides to leave as a molecule of water. As it departs, it leaves behind a positively charged carbon atom – a carbocation. This carbocation is super electron-deficient and is just begging for something to happen.
Now, what else is floating around? Well, we're heating things up, which gives molecules more energy to move and react. If there's another molecule of our starting material nearby, its electrons might be attracted to this positively charged carbocation. Or, if we're doing an elimination reaction, a proton from an adjacent carbon might be grabbed by a base (even a weak one like water, especially when heated), causing the electrons from the C-H bond to shift and form a double bond between the carbon that lost the leaving group and the carbon that lost the proton.
In this specific scenario with heating and a strong acid, the most likely outcome is an elimination reaction. The water molecule leaves, a carbocation forms, and then a proton is removed from an adjacent carbon to form a new double bond. Ta-da! We’ve just gone from an alcohol to an alkene. It's like turning a simple fruit into a more complex pastry by baking it and letting some moisture evaporate.

It's All About the Most Stable Outcome
The overarching principle guiding all these transformations is the drive towards greater stability. Molecules, like people, generally prefer to be in a relaxed, low-energy state. Double and triple bonds, for example, are often more stable than a crowded arrangement of single bonds. Complete octets of electrons (where each atom has a full outer shell of eight electrons) are much happier than atoms with incomplete shells. Leaving groups that are stable on their own are more likely to depart.
When you look at a problem, the first thing to do is identify the functional groups present in your starting material and the reagents you're using. Then, consider the conditions: Is it acidic? Basic? Hot? Cold? Does it involve a catalyst? These are your clues. Think about what each reagent wants to do. Does it want to donate electrons? Accept electrons? Grab a proton? Let go of something? Your task is to anticipate the most logical and energetically favorable way these interactions will play out to form the major organic product.
It might seem daunting at first, like learning a new language. You see a bunch of strange symbols and have no idea what they mean. But as you learn the alphabet (the atoms), the grammar (how they bond), and the common phrases (the typical reactions), it starts to make sense. Soon, you’ll be able to look at a chemical equation and have a pretty good idea of what’s going to happen, just like you can glance at a few ingredients and know if you’re about to make a soufflé or a rock-hard biscuit.
So, next time you see a chemical reaction, don't panic. Grab your mental apron, examine your molecular ingredients, and follow the chef's instructions. You might just surprise yourself with what delicious (or at least, predictable) molecular dish you can create! It’s a fascinating puzzle, and the more you practice, the better you get at seeing the underlying order in what might initially seem like chemical chaos. Happy predicting!
