Draw The Major Organic Product For Each Synthetic Reaction Step
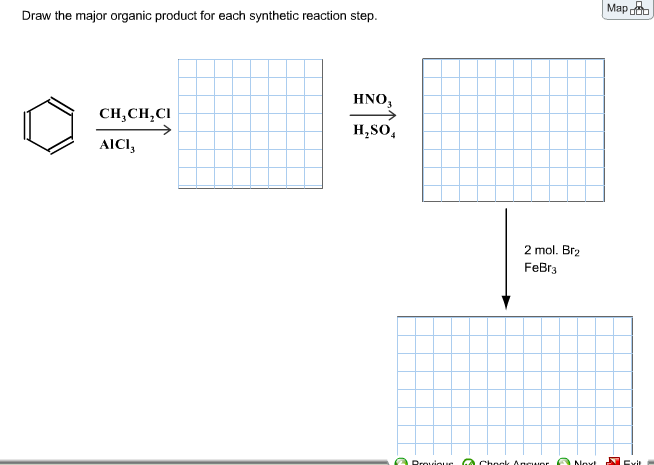
Alright, gather 'round, you lovely humans with brains that, I suspect, are secretly craving a little bit of organized molecular mayhem! We're about to embark on a grand adventure, a quest of epic proportions, where we'll be playing chemist without the risk of accidentally setting off any questionable odors or, you know, blowing up the neighborhood. Today’s mission, should you choose to accept it (and let's be honest, you’re already here, so the deal’s done!), is to draw the major organic product for each synthetic reaction step. Think of it as a delicious chemistry puzzle, served with a side of bewildered amusement and maybe a sprinkle of “Wait, what just happened?”
Now, I know what some of you are thinking. “Organic chemistry? Isn't that the stuff that makes your eyes glaze over faster than a doughnut at a bakery convention?” To which I say, pah-shaw! It’s all about telling a story, a tiny, microscopic drama unfolding on the canvas of a test tube. And we, my friends, are the discerning audience, the ones who get to see the glorious finale, the crowning achievement of each chemical transformation. We're not just drawing molecules; we're witnessing molecular makeovers, chemical celebrities undergoing fabulous transformations. Forget Hollywood, this is where the real drama happens!
Let’s dive right in, shall we? Imagine our first reactant. It’s a perfectly respectable molecule, let's call it "Mildred." Mildred’s got a certain… je ne sais quoi, a subtle elegance. But Mildred is about to meet her match. Enter our first reagent. Now, reagents are like the fairy godmothers (or sometimes the mischievous goblins) of the organic world. They swoop in, do their thing, and Mildred is never quite the same again. For our first act, let's say Mildred is a simple alkene, a double bond just chilling, minding its own business. And then, BAM! Along comes a halogen, like Br₂, a bromine molecule, looking all diatomic and ready to party.
What’s Mildred gonna do? Well, the double bond, you see, is like a little social butterfly. It’s got all this extra electron density, just begging to be noticed. And that bromine molecule? It’s a bit of a flirt. It sees that double bond and thinks, “Ooh, attention!” So, what happens is that the bromine molecule gets a little polarized, one bromine atom gets slightly positive, and the other slightly negative. And that positive bromine atom? It’s like a moth to a flame, drawn irresistibly to the electron-rich double bond. It’s a classic case of opposites attract, just on a much, much smaller scale. We're talking electrophilic addition here, folks, the ABCs of alkene reactions, or as I like to call it, "The Great Double Bond Dissolution."
So, the bromine molecule, in a fit of chemical passion, breaks apart. One bromine atom latches onto one carbon of the double bond, and the other bromine atom? Well, it’s got a negative charge now, like a disgruntled ex. But fear not! Because the first bromine atom, once it's attached, forms a little three-membered ring thingy with the adjacent carbon. It’s called a bromonium ion, and it’s very fancy. This bromonium ion is so electrophilic, so eager for electrons, that it basically screams, “Anyone else got some electron love to share?” And guess who’s lurking nearby, still feeling a bit left out? That’s right, the bromide ion, our disgruntled ex. She sees her chance!
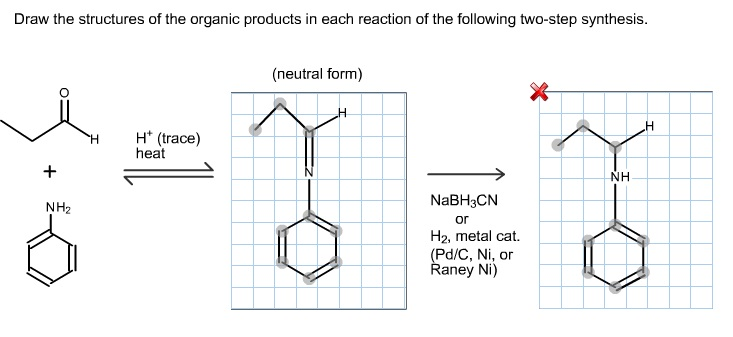
The bromide ion, with her abundance of electron negativity, swoops in from the opposite side (this is important, it’s called anti-addition, and it’s a real snob about it) and attacks one of the carbons in the bromonium ring. The ring cracks open, and voila! You’ve got yourself a vicinal dibromide. Two bromines, happily situated on adjacent carbons. Mildred is no longer Mildred; she’s now… Barbara. Barbara the dibromide. And if you were to draw her, you’d see two carbons, each holding a bromine atom, and all the original hydrogen atoms still dutifully attached. Easy peasy, right? If you can manage to remember that the bromines go on opposite sides. It’s like a chemical handshake, but with a bit more molecular drama.
Now, let's spice things up a bit. What if our Mildred the alkene is a bit more complex? What if it has different groups attached? This is where things get really interesting, and where we start to appreciate the nuances of chemical destiny. Imagine Mildred has a choice: she can react with a reagent that prefers to attach to one carbon over another. This, my friends, is the realm of regioselectivity. It’s like Mildred has to decide which side of the party to join, and the reagent has its own VIP list.
Let’s say we’re dealing with a reaction involving a hydrogen halide, like HCl. Hydrogen chloride, a classic for adding across double bonds. Now, that double bond is still the social butterfly, but the HCl molecule is a bit different. It’s polarized too, with a slightly positive hydrogen and a slightly negative chlorine. When the hydrogen (the positively charged bit, the electrophile) approaches the double bond, it has a choice. It can attach to one carbon or the other.
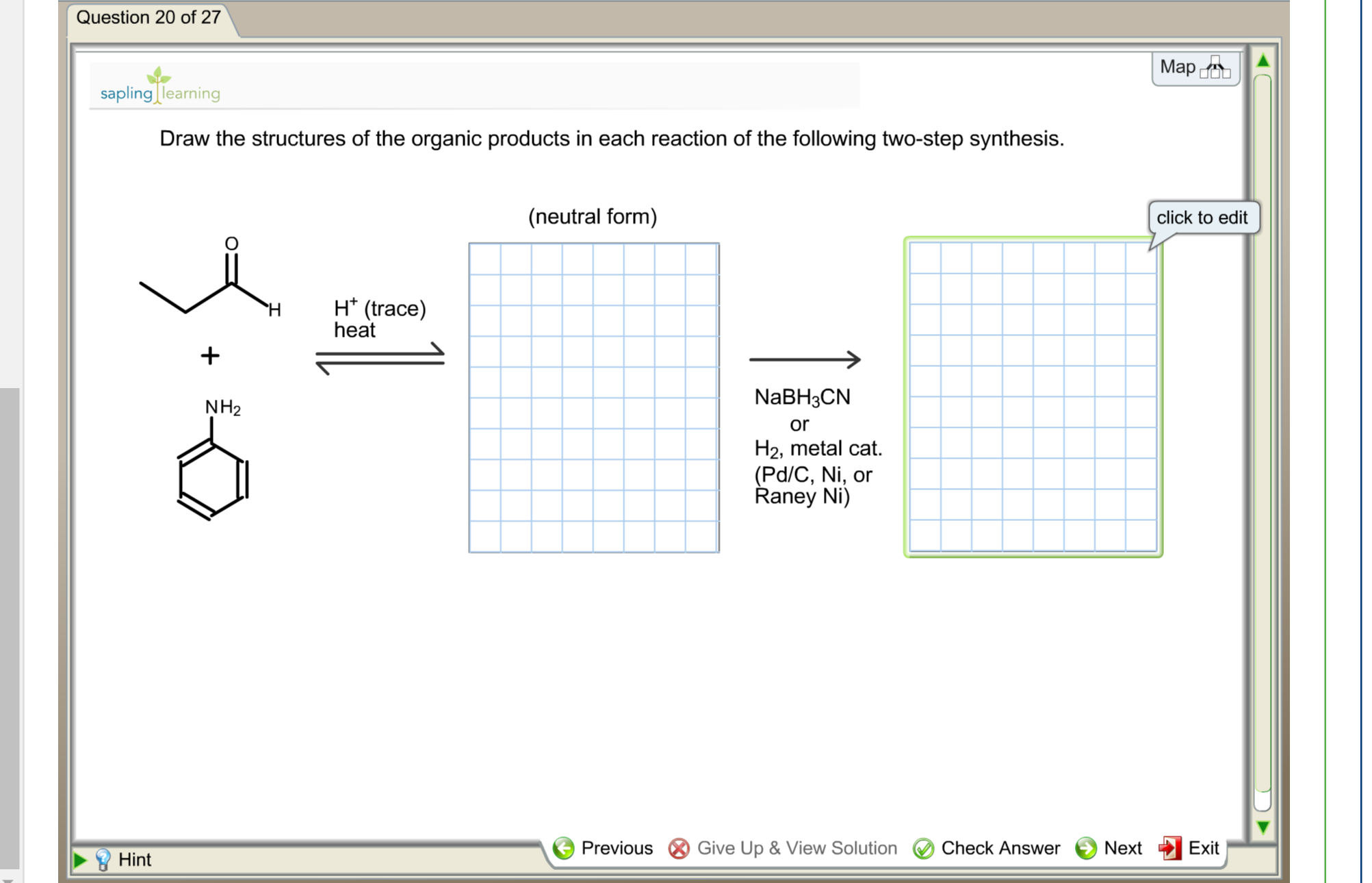
And here’s the kicker: the universe, in its infinite wisdom (or perhaps just the laws of thermodynamics), prefers stability. The hydrogen will preferentially attach to the carbon that results in the most stable intermediate. In organic chemistry, stability is often linked to carbocations. The more substituted a carbocation, the more stable it is. Think of it like a popularity contest. A tertiary carbocation (attached to three other carbons) is way more popular and stable than a primary carbocation (attached to only one other carbon). So, the hydrogen will add to the carbon that leaves behind the most stable carbocation. This is Markovnikov's Rule in action, the legendary guiding principle that basically says, “The rich get richer!” The hydrogen goes to the carbon that already has more hydrogens, leaving the more substituted carbon to form the more stable carbocation, which then gets attacked by the halide.
So, if your alkene looks like a wonky bicycle, and the double bond is between a carbon with one hydrogen and a carbon with two hydrogens, the hydrogen from HCl will go to the carbon with two hydrogens, creating a more stable carbocation on the carbon with only one hydrogen. Then, the chloride ion comes in and ding ding ding! it attacks that carbocation. The product? You’ve got your chlorine atom sitting on the more substituted carbon, and the hydrogen on the less substituted carbon. It’s like the hydrogen showed up at the party and immediately found the most popular spot, leaving the less popular carbon to host the guest of honor.

This is crucial, folks. It’s not just random splashing around. There’s a method to this molecular madness. And when you’re drawing these products, you need to be thinking: where is the most likely place for this new group to land? Where will it create the most stable intermediate? If you get that right, you’re well on your way to becoming a molecular maestro.
Let's try another one. Imagine you have a lovely little alcohol, like ethanol. Ethanol’s got that hydroxyl group (-OH), which is pretty stable. But what if we want to make it even more interesting? What if we want to replace that -OH with something else, like a halogen? Well, alcohols are a bit stubborn. That -OH group doesn't just up and leave. You need a special kind of persuasion.
Enter, our trusty friend, the hydrogen halide again, say HBr this time. Now, if you just mix ethanol and HBr, not much happens. The -OH is too strong a bond. But, if you heat them up, things start to get interesting. The hydrogen from HBr will first protonate the oxygen of the -OH group. Think of it as giving the -OH group a little electrical charge boost, making it a better leaving group. So, it becomes -OH₂⁺, which is basically water with an extra proton, and water is a fantastic leaving group. Water loves to bail! So, that water molecule just pops off.

And what’s left behind? You guessed it! A carbocation. In the case of ethanol, if the H from HBr adds to the oxygen, the whole -OH₂ group leaves, leaving a positively charged carbon. This is a primary carbocation, and as we discussed, it's not the most thrilled about existing. But it's there! And what's floating around, feeling a bit lonely and electron-deficient? The bromide ion, Br⁻. So, the bromide ion swoops in and poof attaches to that carbocation. The product? Ethyl bromide. You’ve successfully turned an alcohol into an alkyl halide. It's like giving Mildred a whole new career, from graceful dancer to… well, a slightly more reactive dancer.
The key here is understanding the leaving group ability. Some groups are much happier to ditch the molecule than others. Hydroxyl groups on their own? Not so much. But when you protonate them, they become water, and water is like the ultimate escape artist. So, when you see an alcohol and a hydrogen halide, especially with heat, think: protonation, water leaves, carbocation forms, halide attacks. Simple, right? If you can follow that chain reaction, you’re practically a molecular sorcerer.
So, there you have it! A little peek into the glamorous world of drawing organic products. It’s all about understanding the personalities of the molecules, their desires, their fears, and how they interact. It's a story of attraction, of stability, and sometimes, of just plain ol' brute force with a bit of heat. Keep practicing, keep imagining those tiny dramas unfolding, and soon, you'll be drawing these products with the same effortless cool as a seasoned barista crafting the perfect latte. Now, who’s ready for the next round of molecular mysteries?
