Draw The Lewis Structures For The Following Ions:

Welcome, brave adventurers, to the thrilling, electrifying, and dare I say, positively sparkling world of Lewis structures! Forget dusty textbooks and intimidating equations; we're about to embark on a joyous quest to map out the secret lives of tiny, powerful particles. Think of it like being a detective, but instead of fingerprints, we're looking for tiny electron buddies hanging out together. It's going to be so much fun, you might even forget you're learning something super important about how the world works!
Today, our mission, should we choose to accept it (and we absolutely should!), is to draw the Lewis structures for a few very special guests: the carbonate ion, the nitrate ion, and the sulfate ion. These aren't just random letters and numbers; oh no! These are the workhorses, the unsung heroes, the rockstars of the chemical universe!
Let's start with our first superstar, the magnificent carbonate ion. Picture this: it's like a tiny, three-piece band, all holding hands (or, you know, electrons). We've got one carbon atom, strutting its stuff, and three oxygen atoms, looking absolutely radiant.
Now, here's the secret handshake. Atoms love to have a full outer shell of electrons, like a perfectly filled candy jar. For most of these guys, that means having eight electrons. It's like a magic number for feeling complete and happy!
So, for our carbonate ion, we're going to arrange our atoms, give them some initial electron buddies, and then start shuffling them around. Think of it like a cosmic dance! We need to make sure everyone's got their eight electrons.
We'll use dots to represent those precious electrons. Single dots are like lonely electrons looking for a friend. When two dots pair up between atoms, that's a covalent bond, like a super-strong handshake holding them together.
Sometimes, atoms need to share more than just one pair. That's when we get a double bond, like a double high-five! It's extra cozy and gives the structure extra stability. It's like adding a safety net, just in case!

And then there are those lone electrons, the ones that haven't found a dance partner. We represent these as pairs of dots floating around an atom, called lone pairs. They're like the shy wallflowers at the party, still important for the overall vibe.
For the carbonate ion, after all our electron shuffling and dancing, you'll notice something super cool. We'll have one carbon atom in the middle, looking very pleased with itself. It will be bonded to three oxygen atoms.
And here's the kicker: one of those oxygen atoms will be rocking a double bond with the carbon. It's like the VIP guest at the party! The other two oxygen atoms will be holding on with a single bond.
But wait, there's more! Those oxygen atoms that are only holding on with a single bond? They've got some extra lone pairs to make up for it. They're looking a little bit like they're carrying a tiny, negative backpack.
This is because our carbonate ion has a total charge of -2. So, those extra electrons, those tiny negative charges, are crucial! We'll see them as lone pairs on the oxygen atoms. We also put the whole thing inside square brackets with a superscript -2, like a fancy label for its superhero identity.
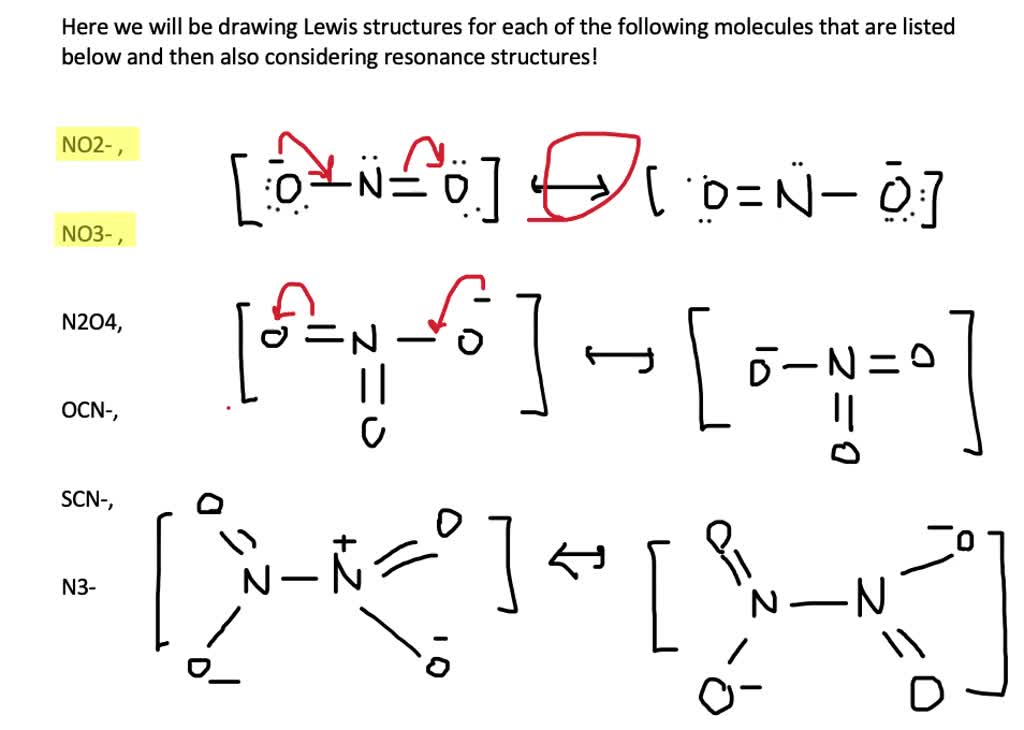
Now, let's move on to our next dazzling performer: the nitrate ion! This one is a bit more streamlined, like a sleek sports car. It's made of one nitrogen atom and three oxygen atoms.
The process is exactly the same, like following a delicious recipe! We count up all our electron buddies, arrange the atoms, and start pairing them up. We want everyone to be happy and have their full set of eight electrons.
For the nitrate ion, the nitrogen atom will be in the center, like the conductor of an orchestra. It will be bonded to the three oxygen atoms. Again, we're aiming for that magical octet!
And guess what? Just like with our carbonate friend, one of the oxygen atoms will get the prestigious double bond. It's like the spotlight is on! The other two oxygen atoms will be connected with single bonds.
And once more, those single-bonded oxygen atoms will be carrying some extra electron baggage, those lovely lone pairs that give our nitrate ion its overall charge of -1. We'll enclose it in brackets with a superscript -1. It's a subtle but important detail!

It’s important to remember that these structures are a bit like snapshots. The actual electrons are zipping around so fast, it's like a blur! Lewis structures give us a simplified, yet incredibly useful, way to visualize their arrangements. They help us understand how these ions will interact with other atoms and molecules.
Finally, let's meet the grand finale, the mighty sulfate ion! This one is a bit of a powerhouse, and it shows us some really cool chemistry. We've got one sulfur atom at the heart of it all, surrounded by four oxygen atoms.
Here, things get a little more interesting. Sulfur is in a special category called an "expanded octet." It's like it's so popular and has so many friends that it can actually have more than eight electrons around it! It’s like having an all-you-can-eat electron buffet.
So, for the sulfate ion, we'll start with the usual electron counting and atom arrangement. Sulfur will be the central atom, looking very busy with its four oxygen companions. We're aiming for those happy octets, but with sulfur, we have a bit more flexibility.
In the most common and stable Lewis structure for the sulfate ion, you'll find that sulfur is connected to all four oxygen atoms with single bonds initially. It’s like everyone’s holding hands in a big circle. But to make everyone truly happy and fulfill their electron desires, we need a little bit of extra magic.
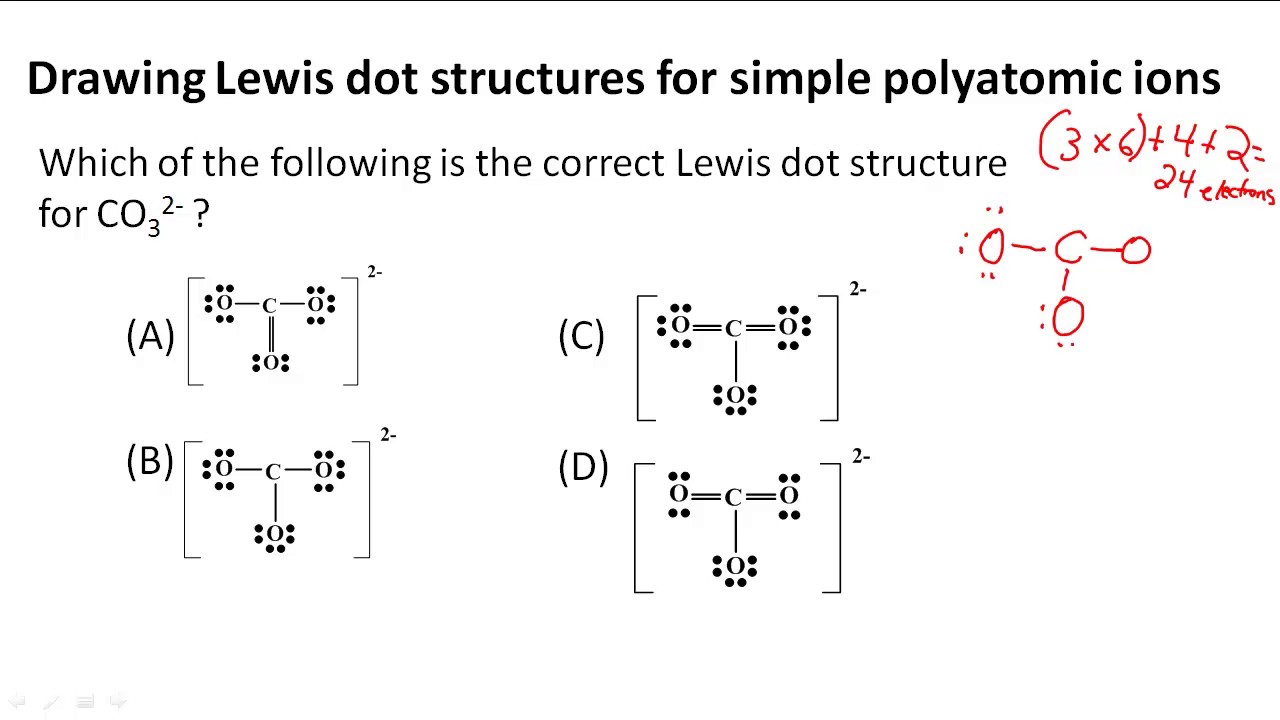
To achieve the overall charge of -2 for the sulfate ion, we'll find that two of the oxygen atoms will have a double bond, while the other two will have a single bond. This arrangement allows the sulfur to accommodate more than eight electrons around it, making it a super-stable molecule. It’s a beautiful compromise in the electron world!
And just like our previous ions, those oxygen atoms with the single bonds will be sporting their characteristic lone pairs, carrying the negative charge. The whole structure, of course, gets its protective bracket embrace with a superscript -2. It’s a testament to the incredible variety and adaptability of atoms!
Drawing these Lewis structures might seem like a puzzle at first, but with a little practice, you’ll be a pro in no time! It’s all about counting electrons, connecting atoms with bonds, and making sure everyone has a full outer shell (or a happy expanded octet!).
So there you have it, the delightful doodles of the carbonate ion, the elegant lines of the nitrate ion, and the robust structure of the sulfate ion. Each one tells a story about how these charged atoms behave and interact. It’s like learning a secret language that explains so much of the world around us!
Keep practicing, keep exploring, and remember that even the smallest particles have incredibly fascinating lives. Happy drawing, and may your electrons always find their happy octets! You've conquered another piece of the amazing chemical universe!
