Draw The Lewis Structures For Resonance Forms Of Acetamide
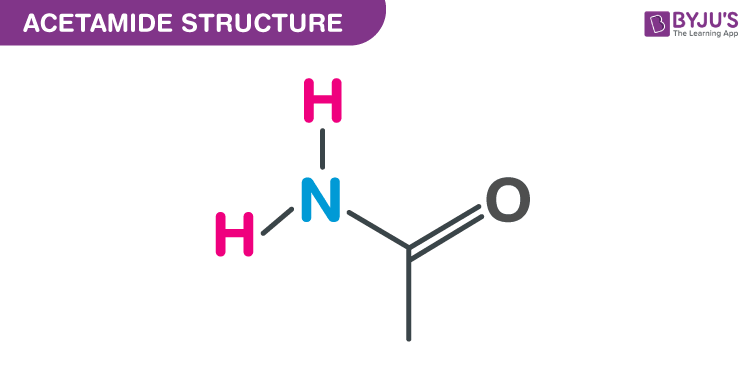
Okay, confession time. I have a little secret. It’s something that might make a few chemistry folks wrinkle their noses, but I’m just going to put it out there. I actually enjoy drawing Lewis structures. There, I said it. It’s like a little puzzle, a doodle with rules. And when it comes to the Lewis structures for resonance forms of acetamide, well, that’s where things get particularly… sparkly.
Acetamide. Sounds fancy, right? Like something a butler would polish. But at its core, it’s just a humble little molecule. It’s got a carbon, a nitrogen, some hydrogens, and an oxygen. Pretty standard stuff. But the way these atoms arrange themselves, the way their electrons dance around, that’s the real magic. And when we talk about resonance, we’re talking about a molecule that can’t quite make up its mind. It’s like a teenager with too many outfit options. It presents a few different looks, and we, the observers, have to appreciate them all.
So, let’s dive into this. Imagine you’ve got your trusty pen and paper, or maybe a fancy digital drawing tablet. We’re going to sketch out these different possibilities for acetamide. It’s not about finding the one perfect picture. Oh no, that would be far too simple, wouldn’t it? It’s about seeing the different ways the electrons can spread out, creating a more stable, a more interesting molecule.
Think of it like this: you’ve got a really good cookie recipe. But maybe sometimes you add a little extra chocolate chip, and sometimes you add a sprinkle of sea salt on top. Both are amazing cookies, right? They’re variations on a delicious theme. Acetamide’s resonance forms are kind of like that. The molecule is fundamentally the same, but the electron distribution gives it slightly different personalities.
First up, we’ve got the structure that probably feels most “normal.” The carbon is double-bonded to the oxygen. This one often feels like the default setting. It’s the comfy sweater of acetamide’s wardrobe. Everyone’s seen it, everyone’s comfortable with it. The nitrogen is happily attached to the carbon with a single bond, and it’s got those lovely lone pairs of electrons. They’re like little fluffy clouds just hanging out, ready to participate in something exciting.
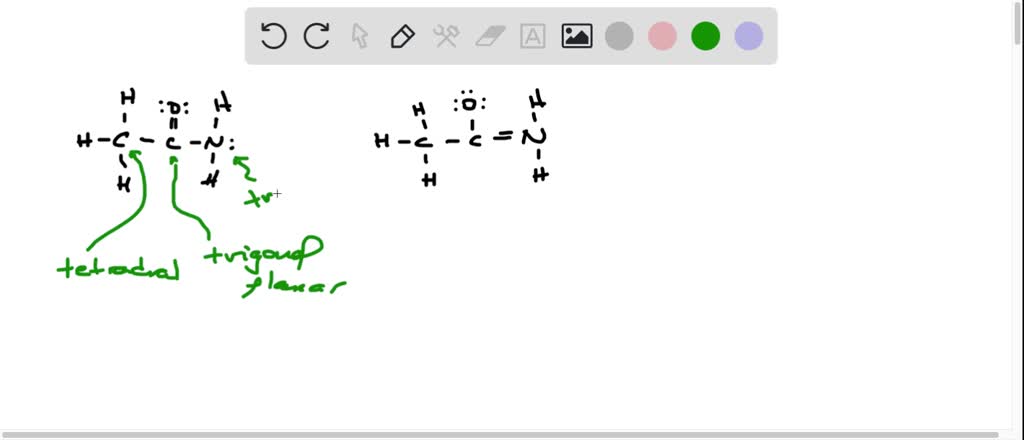
But then, the electrons get restless. They’re like, “You know what? Let’s try something different!” And poof! The double bond between the carbon and oxygen decides to go on vacation. Where does it go, you ask? It gets redistributed. A pair of electrons from the oxygen, which was chilling in a lone pair, decides to form a double bond with the carbon. This is where things get a little dramatic.
Suddenly, the oxygen is still double-bonded to the carbon, but now it has a negative charge. It’s like it’s wearing a slightly grumpy hat. And the nitrogen? Oh, the nitrogen becomes the star of the show! It forms a double bond with the carbon. This is a big deal. And because it’s now sharing more electrons, it picks up a positive charge. It’s strutting its stuff, feeling all important. This is the slightly edgy, punk-rock version of acetamide.
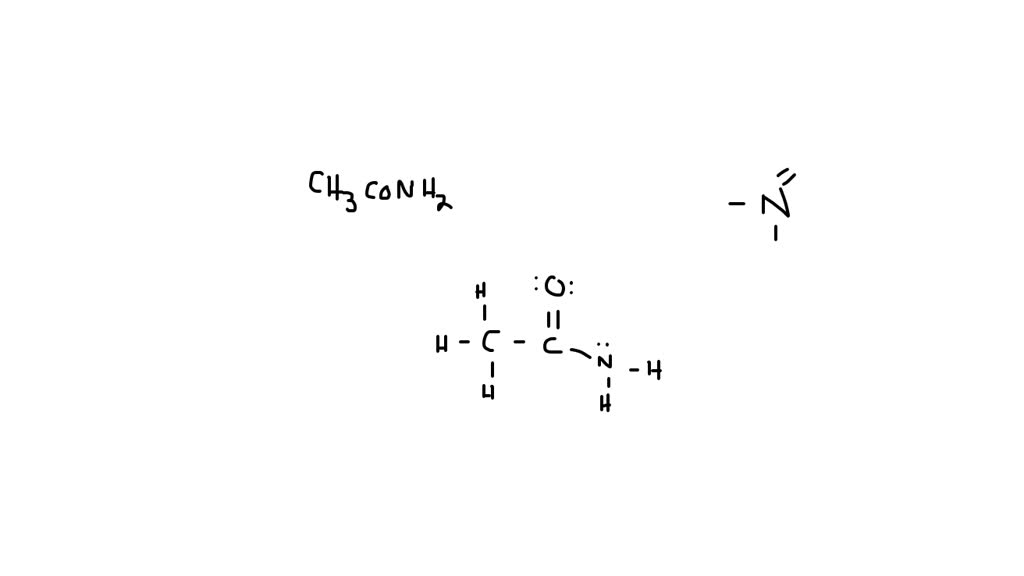
So, you’ve got these two main scenes playing out. The “comfy sweater” version where the oxygen is neutral, and the “punk-rock” version where the nitrogen is positively charged and the oxygen is negatively charged. And the real beauty of it is that the actual acetamide molecule is like a blend of these two. It’s not just one or the other. It’s like a smoothie, a perfect combination of all the best ingredients.
It’s important to remember that these aren’t two separate molecules existing side-by-side. They’re just different ways we, with our limited human perspective, can draw the electron distribution. The molecule itself is just being acetamide, doing its molecular thing. It’s like trying to describe a feeling. You can use different words, different metaphors, but the feeling itself is a whole, complex experience.
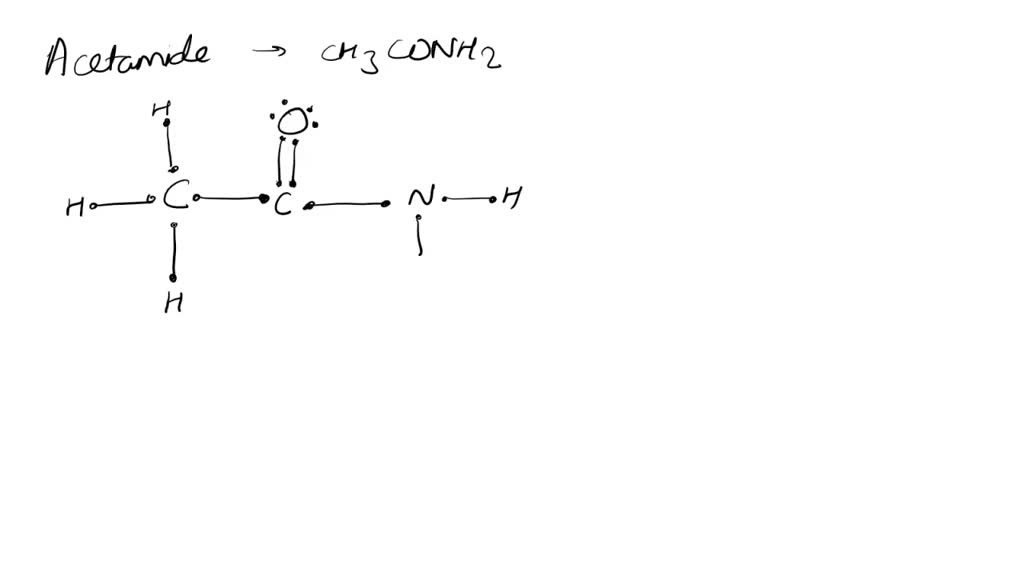
Drawing these resonance forms is like giving the molecule a chance to show off its different angles. It helps us understand how the electrons are distributed, where the molecule is a little more electron-rich, and where it might be a little electron-deficient. This, in turn, tells us a lot about how acetamide will behave in reactions. It's like getting a sneak peek at its personality before you even introduce yourselves.
And honestly, isn't that kind of cool? That something so seemingly simple can have these internal dialogues, these electron-driven debates? It's a reminder that even in the smallest corners of the universe, there's complexity, there's dynamism, and there's a whole lot of electron shuffling going on. So, next time you’re faced with drawing some Lewis structures, especially for something like acetamide’s resonance forms, embrace the puzzle. Enjoy the different possibilities. It’s not just chemistry; it’s a little bit of molecular art. And who doesn't love a good doodle that actually means something?
