Draw The Lewis Structure Of Sf4 Showing All Lone Pairs

Ever stared at a molecule and thought, "Wow, that looks like a tiny, energetic little critter!"? Well, get ready to meet one such character that's been making waves in the world of chemistry: SF4, or sulfur tetrafluoride. Now, drawing its Lewis structure might sound a bit like a science homework assignment, but trust me, it's way more fun than it sounds. Think of it as sketching a tiny, three-dimensional puzzle, and the end result is surprisingly... well, unique!
You see, when we talk about Lewis structures, we're basically looking at how atoms in a molecule decide to share their electrons. It's like a dance party where everyone's holding hands (or, in this case, electrons). For SF4, this dance gets a little jazzy. We've got a central sulfur atom, the party host, and it's invited four fluorine atoms to join the fun. Each fluorine atom brings one electron to the table, and sulfur, being a bit of a generous soul, has a few of its own to contribute.
The real magic happens when we start arranging these electrons. We draw little dots around each atom to represent these shared electrons, forming what we call chemical bonds. It's like putting stickers on a diagram. But here's where SF4 really stands out: it's not just about the bonds. Oh no, SF4 has a secret stash of electrons, and we call these lone pairs. These are electrons that belong solely to one atom and aren't participating in the sharing dance.
For SF4, that central sulfur atom ends up with not just the electrons it's sharing with the fluorines, but also a couple of extra, unattached electrons. This is where the shape gets interesting. Imagine trying to arrange four friends (the fluorines) around a central person, and then that central person has two extra toys they're holding onto. It's going to be a bit crowded and wonky, right? That's kind of what happens with SF4.
When you draw out the Lewis structure for SF4, you'll see the sulfur atom in the middle, connected to each of the four fluorine atoms by a single bond (that's two shared electrons). But then, nestled right there with the sulfur, you'll spot those two little dots representing the lone pair. This lone pair has a big personality and really influences how the whole molecule behaves. It pushes and shoves the fluorine atoms around a bit, giving SF4 a shape that’s not perfectly symmetrical. It's kind of like a seesaw, but with a few extra bits.

Why is this so cool? Because it's not just a pretty picture. This arrangement of atoms and electrons, the whole dance with bonds and those sneaky lone pairs, is what gives SF4 its unique chemical properties. It's what makes it react in certain ways and not others. It's like understanding a character's quirks – once you know them, you can predict what they're going to do.
And the drawing itself? It’s a little bit like solving a mystery. You start with the atoms, you count up the electrons, and then you start fitting them together, trying to satisfy everyone. Sometimes, it takes a bit of shuffling. You might draw bonds one way, and then realize, "Wait a minute, this isn't quite right. Where do those extra electrons go?" And then, aha! You discover the lone pairs, the hidden stars of the show.
So, when you're looking at the Lewis structure of SF4, you're not just seeing dots and lines. You're seeing a story. You're seeing how atoms, with their electrons, come together to form something new and interesting. You're seeing the importance of those lone pairs, which are often overlooked but play a crucial role in the molecular drama. It’s a simplified peek into the complex world of chemistry, presented in a way that’s surprisingly accessible and, dare I say, fun.
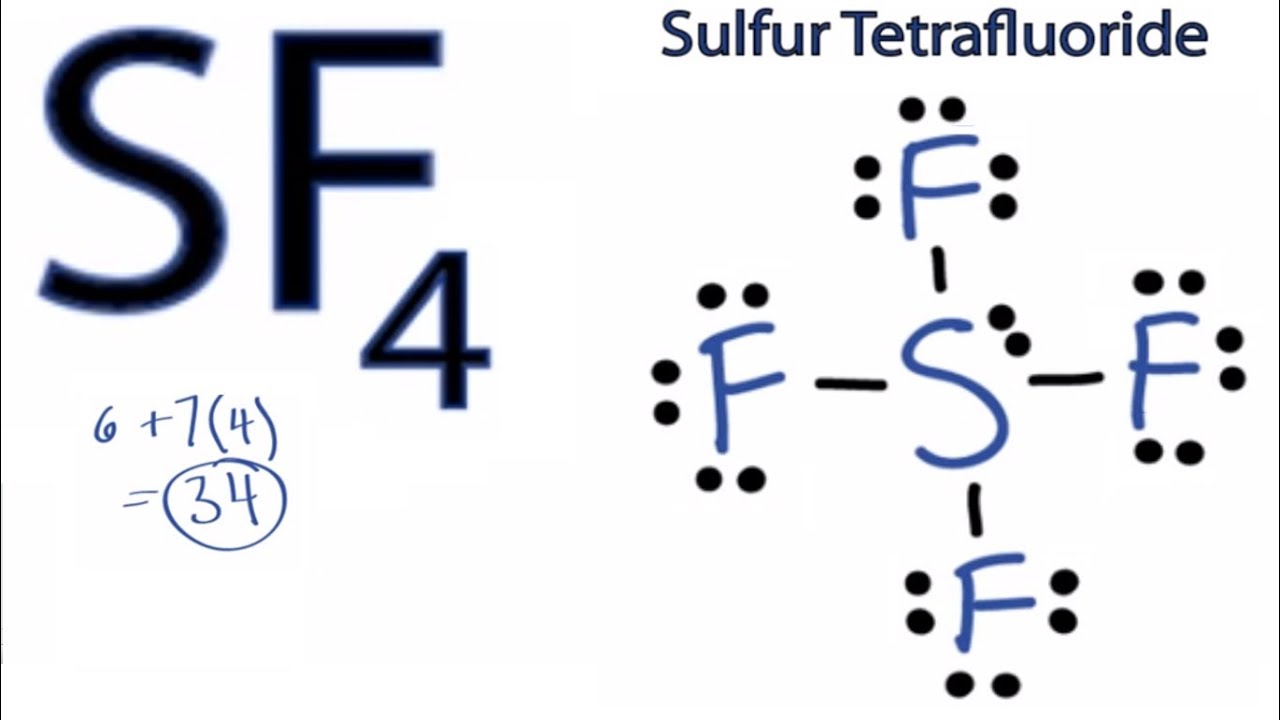
Think of it as a visual puzzle that unlocks understanding. The sulfur atom, with its two lone pairs, creates a bit of a lopsided arrangement. It’s this specific arrangement, often described as a seesaw or a distorted trigonal bipyramidal shape, that makes SF4 so interesting to chemists. It's not your typical, neat-and-tidy molecule. It has character!
And drawing it? It’s a step-by-step process that builds confidence. You start with the central atom, usually the least electronegative (and sulfur fits that bill here). Then you surround it with the more electronegative atoms – in this case, the four fluorines. You draw single bonds between them, which uses up some of your electron budget. Then comes the crucial part: accounting for all the remaining electrons. This is where you discover the beauty of those lone pairs on the central atom. It's like finding missing pieces that make the whole picture make sense.

The act of drawing it, of meticulously placing those dots and lines, is incredibly satisfying. It's a tangible way to grasp abstract concepts. You're not just reading about it; you're actively creating it. You see the bonds form, you see the electrons pair up, and you see those solitary lone pairs adding their influence. It’s a moment of clarity when you finally get the structure right, and you can visualize the molecule in its full, slightly quirky glory.
So, the next time you hear about Lewis structures, especially for a molecule like SF4, don't shy away. Embrace the challenge! Think of it as an artistic endeavor, a scientific doodle that reveals the inner workings of matter. It’s a little peek into the microscopic world, a world where electrons play musical chairs and lone pairs add a touch of delightful chaos. Give it a try, and you might just find yourself surprisingly entertained by the intricate dance of atoms. Who knew chemistry could be this engaging? It’s all about seeing the story unfold, one dot and line at a time, with those crucial lone pairs leading the way.
So, the next time you’re looking at a molecule like SF4, remember it’s more than just symbols and lines. It’s a tiny, fascinating entity with a unique shape and personality, all thanks to the way its electrons are arranged, including those important lone pairs!
