Draw The Lewis Structure Of Sf2 Showing All Lone Pairs

Ever wondered about the secret life of molecules? You know, those tiny building blocks that make up everything from the air we breathe to the tasty snacks we enjoy? Well, buckle up, because we're about to dive into the fascinating world of Lewis structures, and specifically, we're going to draw the Lewis structure of SF2, also known as sulfur difluoride. It might sound a bit technical, but trust us, it's surprisingly fun and incredibly useful for understanding how atoms bond together!
Think of a Lewis structure as a molecular blueprint. It's a visual representation that shows us exactly how atoms in a molecule are connected and where their electrons are hanging out. Why is this so cool? Because understanding these electron arrangements helps us predict all sorts of things about a molecule's behavior, like its shape, its reactivity, and even its physical properties. It's like having a secret decoder ring for the chemical world!
The beauty of Lewis structures lies in their simplicity. They use symbols for atoms and dots to represent valence electrons – those outermost electrons that are involved in bonding. By following a few simple rules, we can construct these structures and unlock the secrets of how atoms share or transfer electrons to achieve a stable arrangement. It’s a bit like a puzzle, where you’re trying to fit the pieces (electrons) in the right places around the atoms.
Now, let's get down to business with our star of the show, SF2. This molecule is made up of one sulfur atom (S) and two fluorine atoms (F). Our mission, should we choose to accept it, is to draw its Lewis structure, making sure we show all the lone pairs. Lone pairs are pairs of valence electrons that aren't involved in bonding; they just kind of chill out on an atom. They play a crucial role in determining the molecule's shape, so we definitely don't want to forget them!
So, how do we start this molecular detective work? First, we need to determine the total number of valence electrons we have to work with. Sulfur is in Group 16 of the periodic table, so it has 6 valence electrons. Fluorine, on the other hand, is in Group 17, meaning each fluorine atom brings 7 valence electrons to the party. Since we have two fluorine atoms, that's 2 * 7 = 14 valence electrons from fluorine. Add that to the sulfur's 6, and we've got a grand total of 6 + 14 = 20 valence electrons to distribute in our Lewis structure for SF2.

Next, we need to figure out which atom is going to be the central atom. Generally, the least electronegative atom is the central one, and it's also the one that appears only once in the formula. In SF2, sulfur is less electronegative than fluorine, and it's the single atom, so sulfur will be our central atom. We'll place the two fluorine atoms around it, like satellites orbiting a planet.
Now for the fun part: drawing the bonds! We connect the central atom to the surrounding atoms with a single bond, which represents a pair of shared electrons. So, we'll draw a line between sulfur and each of the fluorine atoms. Each line uses up 2 electrons, so we've used 2 * 2 = 4 electrons so far.

We still have 20 - 4 = 16 valence electrons left to place. Our goal is to satisfy the octet rule for each atom, meaning we want each atom to have 8 electrons around it (except for hydrogen, which is happy with 2). Fluorine atoms are notorious for wanting a full octet. So, we'll start by adding lone pairs to the fluorine atoms until they are surrounded by 8 electrons. Each fluorine atom already has 2 electrons from the single bond, so we need to add 6 more electrons (3 lone pairs) to each fluorine atom.
Let's do that: we add 3 lone pairs to the first fluorine, using 6 electrons. Then we add 3 lone pairs to the second fluorine, using another 6 electrons. In total, we've used 6 + 6 = 12 electrons to complete the octets on the fluorine atoms. We started with 16 electrons to place after forming the bonds, and we've used 12, leaving us with 16 - 12 = 4 valence electrons remaining.
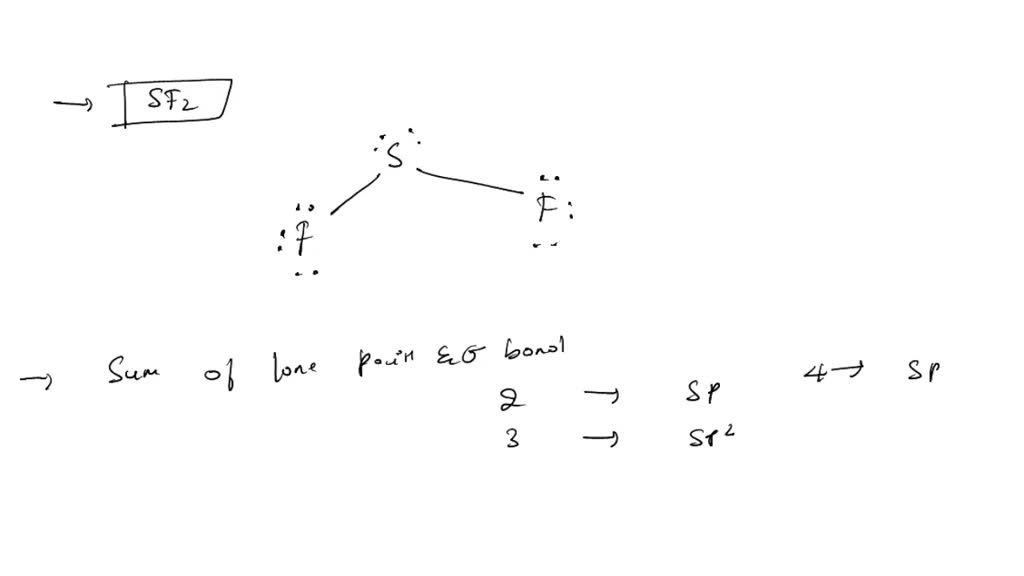
Where do these last 4 electrons go? They must go onto the central atom, sulfur. Since we have 4 electrons left, we'll place them as 2 lone pairs on the sulfur atom. Now, let's check our work. Each fluorine atom has 2 electrons in the bond plus 6 electrons in lone pairs, totaling 8 electrons. That's a happy fluorine! The sulfur atom has 2 electrons from the bond to the first fluorine, 2 electrons from the bond to the second fluorine, and then the 4 electrons from the 2 lone pairs we just added. That gives sulfur 2 + 2 + 4 = 8 electrons. Excellent! Everyone has a full octet.
So, the final Lewis structure for SF2 shows a central sulfur atom with two single bonds connecting it to two fluorine atoms. Each fluorine atom has three lone pairs of electrons around it. And importantly, the sulfur atom has two lone pairs of electrons on it. This visual representation, with all the dots clearly showing the lone pairs, is the Lewis structure of SF2. Isn't it neat how we can represent the complex dance of electrons in such a simple diagram? This is just one example, but it demonstrates the power and elegance of Lewis structures in understanding the fundamental nature of chemical compounds.
