Draw The Lewis Structure Of Hcn Include Lone Pairs
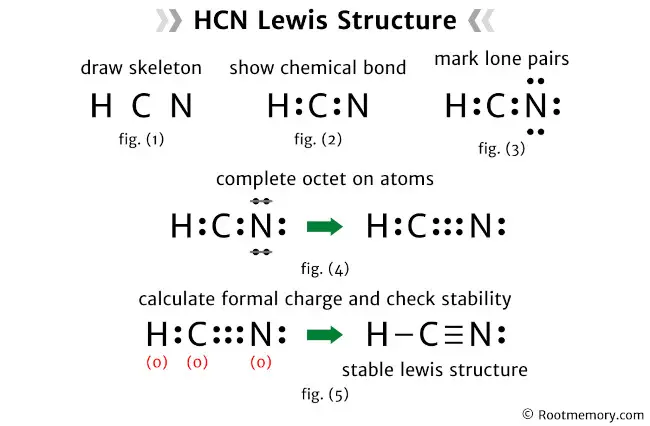
So, picture this: I’m in my first year of college, staring down a chemistry lecture that felt like it was delivered in ancient Sumerian. The professor, a man whose eyebrows seemed to have a life of their own, was explaining something called "Lewis structures." My brain immediately conjured images of tiny, grumpy atoms with little dot-people living in them. Honestly, I was pretty sure I’d be sticking to my major in… well, something that didn’t involve drawing little circles and lines that were supposed to represent… everything.
But then, he pulled up an example. It was HCN. Hydrogen Cyanide. Even the name sounded a bit… hazardous. And suddenly, something clicked. These aren’t just random dots and lines; they’re a peek into the secret lives of atoms, showing how they’re holding hands, or rather, sharing electrons. It’s like the atomic version of a relationship status update. And that, my friends, is what we’re diving into today. We're going to unravel the mystery of the Lewis structure for HCN, and trust me, it's less scary than it sounds. Probably.
Let's Get Down to Business: What's a Lewis Structure Anyway?
Alright, before we get all gaga over HCN, let's lay some groundwork. You can think of a Lewis structure as a sort of blueprint for how atoms bond together in a molecule. It’s all about showing the valence electrons – those are the electrons in the outermost shell of an atom, the ones that are ready to mingle and get into chemical shindigs. These valence electrons are the key players in forming chemical bonds.
G.N. Lewis, the chap who came up with this whole thing, decided that representing these electrons as little dots around an atomic symbol was the way to go. And when two atoms decide to get cozy and share a pair of electrons, we draw a line between them. A single line means a single bond (one shared pair), a double line is a double bond (two shared pairs), and you guessed it, a triple line is a triple bond (three shared pairs). It’s all very straightforward, like a secret handshake for atoms.
The ultimate goal? To satisfy something called the octet rule. Most atoms are happiest when they feel like they have eight valence electrons around them. It’s like they’re all aiming for that perfect, stable configuration. Except for hydrogen, of course. Hydrogen is a bit of a minimalist; it's perfectly content with just two valence electrons. So, it's like the "duet rule" for hydrogen. Keep that in mind, it's a crucial detail!
HCN: The Star of Our Show
Now, let’s talk about our main event: HCN. Hydrogen Cyanide. It's a molecule made up of three atoms: one hydrogen (H), one carbon (C), and one nitrogen (N). And like any good moleculer drama, we need to figure out how these three are going to get along and arrange their valence electrons.
First things first, we need to count up the total number of valence electrons we’re working with. This is like taking an inventory before you start building anything. * Hydrogen (H) is in Group 1, so it has 1 valence electron. Easy peasy. * Carbon (C) is in Group 14, so it has 4 valence electrons. Think of it as having four potential dance partners. * Nitrogen (N) is in Group 15, so it has 5 valence electrons. A bit more generous with its electron offerings.
So, total valence electrons for HCN = 1 (from H) + 4 (from C) + 5 (from N) = 10 valence electrons. That’s the magic number we need to distribute correctly.
Building the Structure: Step-by-Step
Okay, now for the fun part, or at least, the part where we actually draw something. We’re going to follow a general strategy that works for most molecules. Think of it as a recipe for Lewis structures.
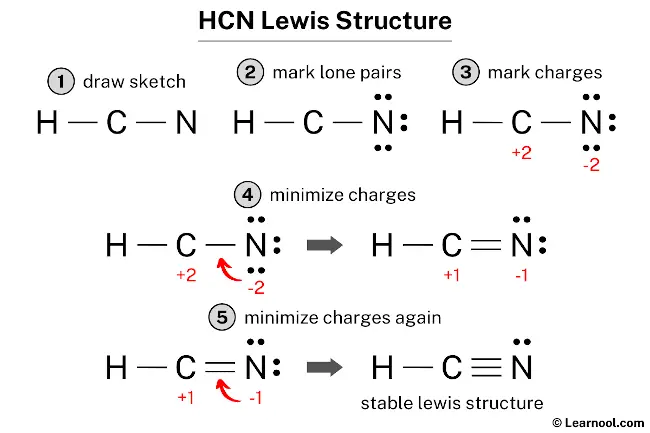
Step 1: Skeletal Structure
We need to figure out which atom is in the middle. Generally, the least electronegative atom goes in the center. Carbon is usually a good candidate for the central atom because it can form multiple bonds. Hydrogen is always on the outside, never in the middle. So, in HCN, carbon is our central atom. We’ll arrange them linearly for now, as that's often the case for simple diatomic and triatomic molecules like this.
So, our initial guess for the skeletal structure looks like this: H – C – N. See those lines? Those represent single bonds, meaning we've used up 2 electrons for the H-C bond and 2 electrons for the C-N bond. That’s a total of 4 electrons used. We still have 10 - 4 = 6 valence electrons left to place.
Step 2: Distribute Remaining Electrons
Now, we need to place the remaining 6 electrons around the atoms to satisfy the octet rule (and the duet rule for hydrogen). We usually start by giving electrons to the outer atoms first, then move to the central atom. Let's try putting them on nitrogen and hydrogen.
If we put all 6 electrons on nitrogen, it would look like this: H – C – N with 6 dots around N. Let’s check octets:
- Hydrogen: Has 2 electrons from the single bond. Happy (duet rule satisfied).
- Carbon: Has 2 electrons from the H-C bond and 2 from the C-N bond, totaling 4. Not happy yet. It needs more!
- Nitrogen: Has 2 electrons from the C-N bond and 6 lone pair electrons around it, totaling 8. Happy (octet rule satisfied).
So, this arrangement is H – C – N:, where the ":" represents the 6 lone pair electrons. But our carbon is still feeling a bit lonely with only 4 electrons. That’s not going to cut it in the molecular world.
Step 3: Form Multiple Bonds
When an atom in the center doesn't have enough electrons to reach its octet, we often have to move some electrons from surrounding atoms to form multiple bonds (double or triple bonds). We can do this by taking a lone pair from an adjacent atom and turning it into a shared pair between the two atoms.
Let’s go back to our structure: H – C – N: (with 6 lone pairs on N). Carbon only has 4 electrons. Nitrogen has 8. Hydrogen has 2. Carbon is the one in need. We can take a lone pair from nitrogen and form a double bond between carbon and nitrogen.

So, we move one pair of electrons from the lone pairs on nitrogen to form a double bond with carbon:
H – C = N: (with 4 lone pairs on N and a double bond between C and N).
Let’s re-check the octets:
- Hydrogen: Still has 2 electrons from the single bond. Happy.
- Carbon: Now has 2 electrons from the H-C bond + 4 electrons from the C=N double bond = 6 electrons. Still not quite there. Needs one more!
- Nitrogen: Now has 4 electrons from the C=N double bond + 4 lone pair electrons = 8 electrons. Happy.
We’re getting closer! Carbon still needs to boost its electron count. Since nitrogen is already participating in a double bond, and hydrogen only wants two, the only place to get more electrons for carbon is from nitrogen again. Let’s move another lone pair from nitrogen to form a triple bond.
So, we take another lone pair from nitrogen and form a triple bond with carbon:
H – C ≡ N: (with 2 lone pairs on N and a triple bond between C and N).
Let’s do the final octet check:
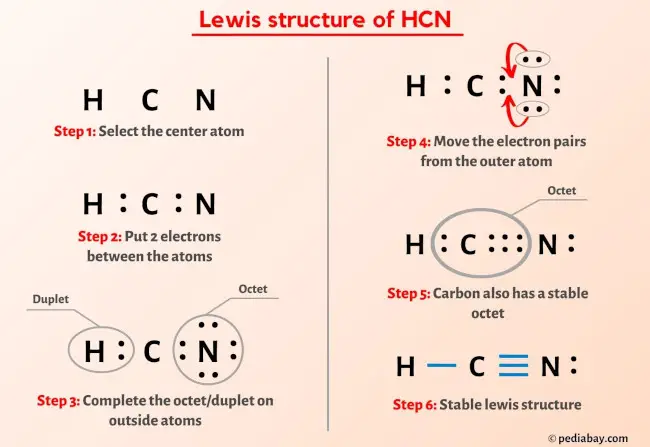
- Hydrogen: Has 2 electrons from the single bond. Duet satisfied. Phew.
- Carbon: Has 2 electrons from the H-C single bond + 6 electrons from the C≡N triple bond = 8 electrons. Octet satisfied!
- Nitrogen: Has 6 electrons from the C≡N triple bond + 2 lone pair electrons = 8 electrons. Octet satisfied!
Success! This looks like the correct Lewis structure for HCN. We’ve used all 10 valence electrons, and every atom (except hydrogen) has a full octet. And hydrogen has its perfect duet.
The Final Lewis Structure: Let's See It!
So, to be super clear, the Lewis structure for Hydrogen Cyanide (HCN) is:
H – C ≡ N:
Let's break it down visually:
- The single line between H and C represents a single bond, meaning they share one pair of electrons.
- The triple line between C and N represents a triple bond, meaning they share three pairs of electrons. That's a lot of sharing! It makes this bond very strong.
- The two dots next to Nitrogen (:) represent its lone pair of electrons. These electrons aren't involved in bonding but are still part of nitrogen's valence shell and contribute to its octet.
And that’s it! You’ve drawn the Lewis structure of HCN. See? Not so terrifying after all. It’s a bit like solving a little puzzle, isn’t it? You’ve got your pieces (valence electrons) and your rules (octet/duet rule), and you’re just trying to fit them together in the most stable arrangement.
Why is this important? (Besides passing chemistry class)
You might be thinking, "Okay, so I drew some dots and lines. Big deal." But these Lewis structures are actually super important! They give us a visual clue about a molecule's shape and its reactivity.
For example, the presence of that triple bond between carbon and nitrogen in HCN tells us it's a very stable and strong molecule. This has implications for how it behaves in chemical reactions. Also, knowing the arrangement of atoms and electrons can help predict if a molecule is polar or nonpolar, which affects how it interacts with other molecules. It's like getting the basic personality profile of a molecule!
And hey, even if you don't plan on becoming a chemist, understanding these fundamental concepts can really demystify a lot of the science you encounter in everyday life. From how medicines work to how our bodies function, it all comes back to these tiny building blocks and how they interact.
A Little Note on Formal Charges (Optional, but good to know!)
Sometimes, when you're drawing Lewis structures, especially with more complex molecules, you might end up with a structure where atoms seem to have a "charge" even though the molecule as a whole is neutral. This is where the concept of formal charge comes in. It's a way of assigning a hypothetical charge to each atom in a Lewis structure to help determine the most likely and stable structure.
The formula is: Formal Charge = (Valence Electrons of the atom) – (Non-bonding electrons) – (1/2 * Bonding electrons).
For our HCN structure (H–C≡N:):
- Hydrogen: 1 (valence) - 0 (non-bonding) - 1/2 * 2 (bonding) = 0. It has a formal charge of 0. Perfect.
- Carbon: 4 (valence) - 0 (non-bonding) - 1/2 * 8 (bonding) = 0. It also has a formal charge of 0. Even better!
- Nitrogen: 5 (valence) - 2 (non-bonding) - 1/2 * 6 (bonding) = 5 - 2 - 3 = 0. Nitrogen also has a formal charge of 0.
When all atoms in a Lewis structure have a formal charge of 0, it's a really good sign that you've got the best representation! The less formal charge on atoms, the more stable the molecule tends to be. So, our HCN structure is looking even more legit now. Pretty neat, right?
So, there you have it. The Lewis structure of HCN, complete with lone pairs and a triple bond that makes carbon and nitrogen practically inseparable. It’s a reminder that even in the seemingly chaotic world of atoms and molecules, there's an underlying order and logic. And sometimes, all it takes is a few dots and lines to reveal it.
Next time you’re presented with a molecule, remember these steps. Count your valence electrons, draw a skeleton, fill in the octets, and don’t be afraid to create multiple bonds. You've got this! And who knows, maybe you’ll even start seeing the world a little differently, one Lewis structure at a time. wink wink
