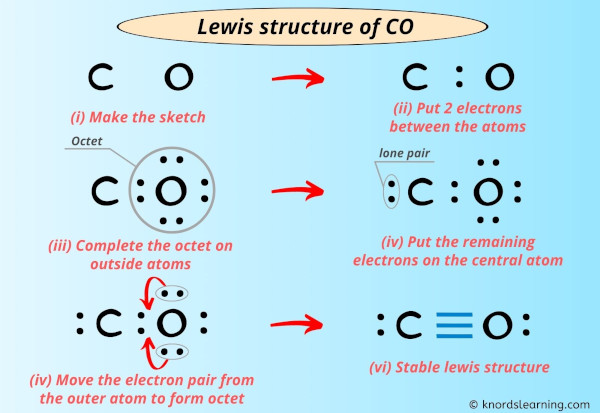
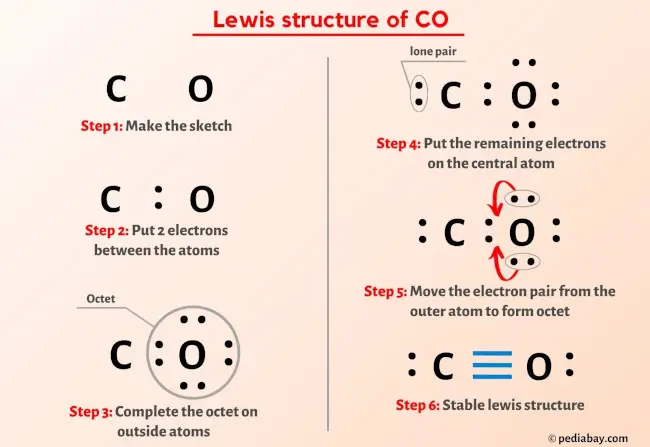
Draw The Lewis Structure Of Co Including Lone Pairs
Hey there, science curious pals! Ever looked at a tiny, everyday thing and wondered what's really going on under the hood? We’re not talking about your car here (though that’s cool too!), but something even smaller, something that makes up… well, everything! Today, we’re going on a little adventure into the electrifying world of atoms, and our star player is none other than Cobalt, or Co for short! Get ready to have your mind blown in the most delightfully simple way possible.
So, imagine atoms are like little Lego bricks that build the entire universe. Some of these bricks are super social and love to share their bits and bobs, while others are a little more… let’s say, possessive. These sharing or possessing habits are all about something called electrons. Think of electrons as tiny, zippy, energetic marbles that orbit the center of an atom like tiny race cars on a track. They’re the secret sauce that makes atoms stick together or do all sorts of cool chemical dance moves.
Now, Cobalt (Co). This is a metal, and metals are generally pretty good at sharing. They’re like the generous neighbors who always have extra cookies to pass around. When we talk about drawing a Lewis structure, we’re basically creating a super simplified, cartoon-like map of how these electron marbles are arranged around an atom, especially the ones on the very outside, which are the ones that get to play in the chemical playground.
For our pal Cobalt (Co), it’s a bit like looking at its outermost electron marbles. In the grand scheme of atoms, Cobalt usually likes to show off nine of these outermost electron marbles. Yep, nine! That might sound like a lot, but trust me, in the atomic world, it’s a manageable bunch. Think of it as Co having nine little bouncy balls it likes to keep close.
Now, the fun part! We represent these outermost electron marbles as dots around the symbol for the atom, which is Co. We're not just going to plonk them down anywhere, oh no. There's a bit of a method to this dot-tastic madness! We spread them out like you’re trying to give each one its own personal space. Imagine you’re decorating a very small, very important cake – you wouldn't just dump all the sprinkles in one corner, right? You’d spread them out evenly, giving each sprinkle its moment to shine.
![Lewis structure of CO [with video and free study guide]](https://biochemhelp.com/wp-content/uploads/2023/06/CO-lewis-structure.jpg)
So, for Cobalt (Co), we’d draw a big Co in the middle. Then, we’d start placing those nine electron dots around it. You start by giving each side a dot, almost like you’re putting a little guardian on each of the four sides. Then, you go back around and add a second dot to each side, because some of these marbles like to pair up! It’s like a few of them are best buddies who always hang out together. So, you’d have four pairs of dots, and then that lonely ninth dot that’s still looking for a friend, or maybe it’s just really independent!
But wait, there’s a special category of these electron marbles called lone pairs. These are the electron pairs that are chilling out, not actively looking to bond with another atom. They’re the ones saying, "Nah, I'm good right here, enjoying my own company." For Cobalt (Co), when we're just drawing its Lewis structure on its own, we have to show these little cliques of electron pairs. It’s like making sure every little buddy has a spot!
When you’re drawing the Lewis structure of Cobalt (Co), and you account for all those outermost electrons, you’ll see a beautiful arrangement. You’ll have pairs of dots, and then that single dot. It’s the atom’s way of saying, "Here are all my outermost marbles, nicely organized!"
The key takeaway here is that drawing a Lewis structure for an element like Cobalt is like making a simple, visual cheat sheet. It shows us the key players – the outermost electrons – and how they like to hang out. It's not rocket science; it’s more like playing a super-organized game of dot-to-dot with tiny, energetic particles. And the next time you hear about Cobalt, you can picture those little electron dots, buzzing around, ready for whatever chemical adventure awaits!
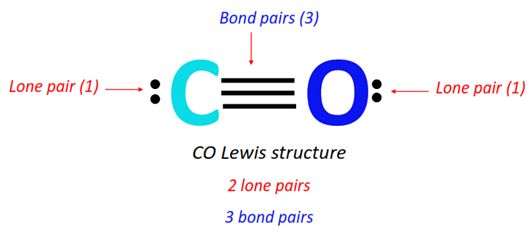
Think of it this way: if atoms were people at a party, the Lewis structure is like a quick sketch of who's standing where and who's holding hands (or electron pairs!). For Cobalt (Co), it’s showing us its outermost guests and how they’re grouped. It’s a peek into the atomic social scene!
And that, my friends, is the magic of drawing the Lewis structure of Cobalt (Co), including those important lone pairs. It’s a simple yet powerful way to visualize the building blocks of our universe. So go forth and draw those dots with joy and confidence! You’re basically an atom whisperer now!
