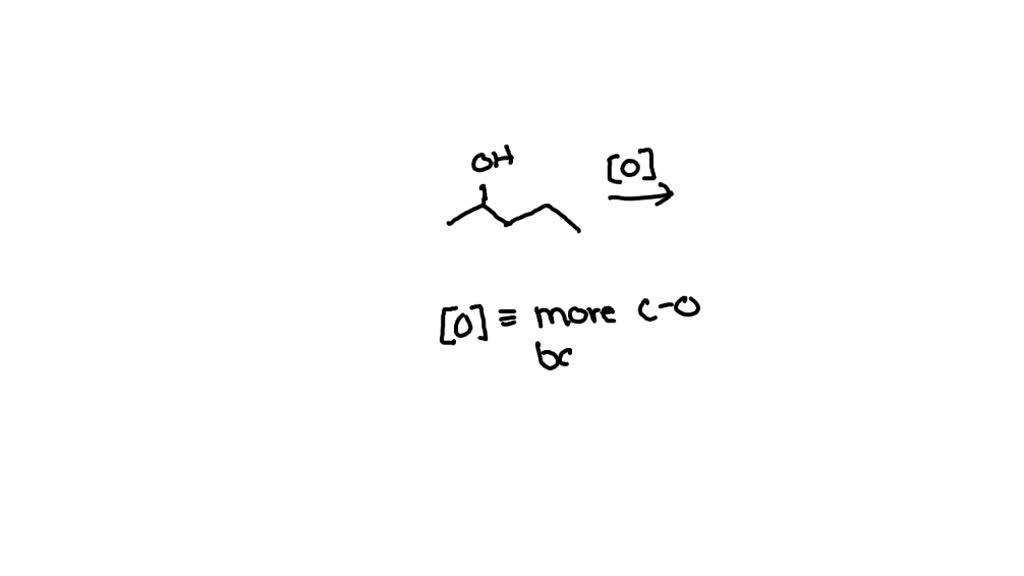
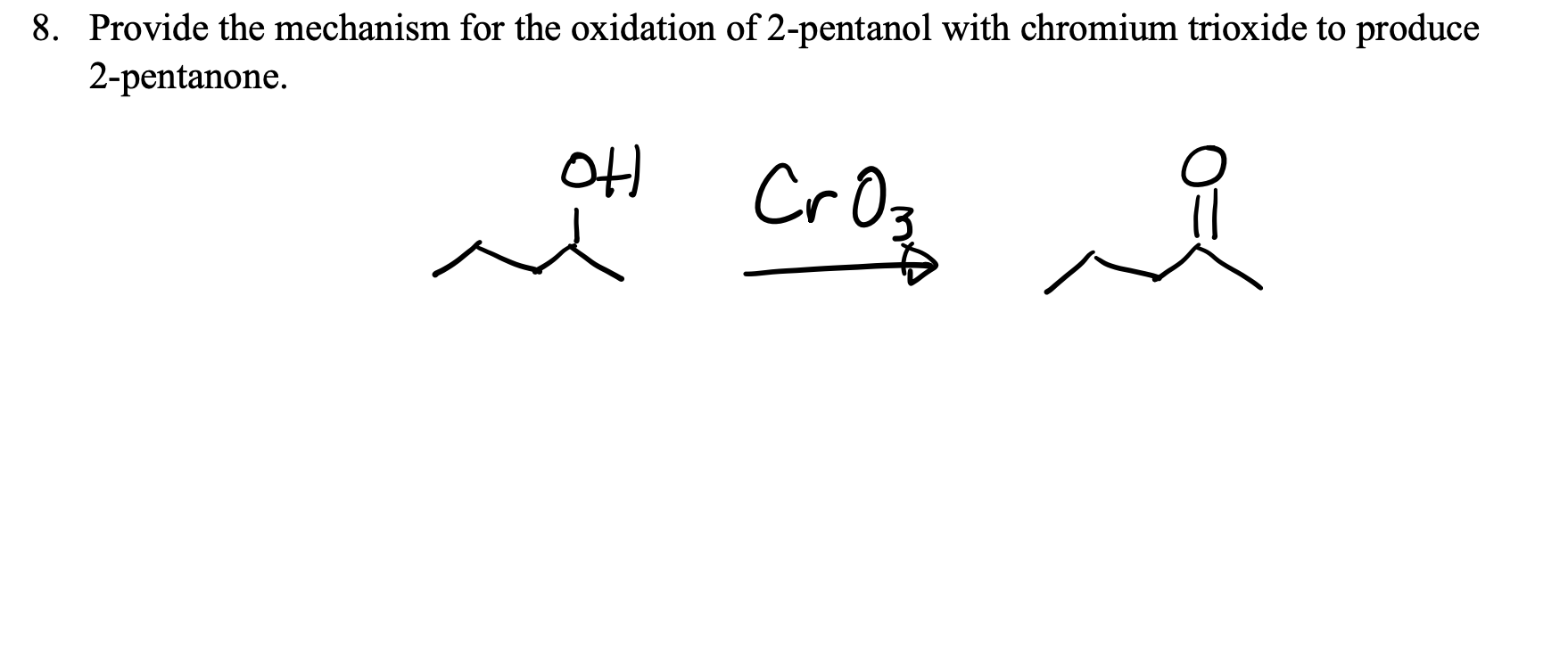
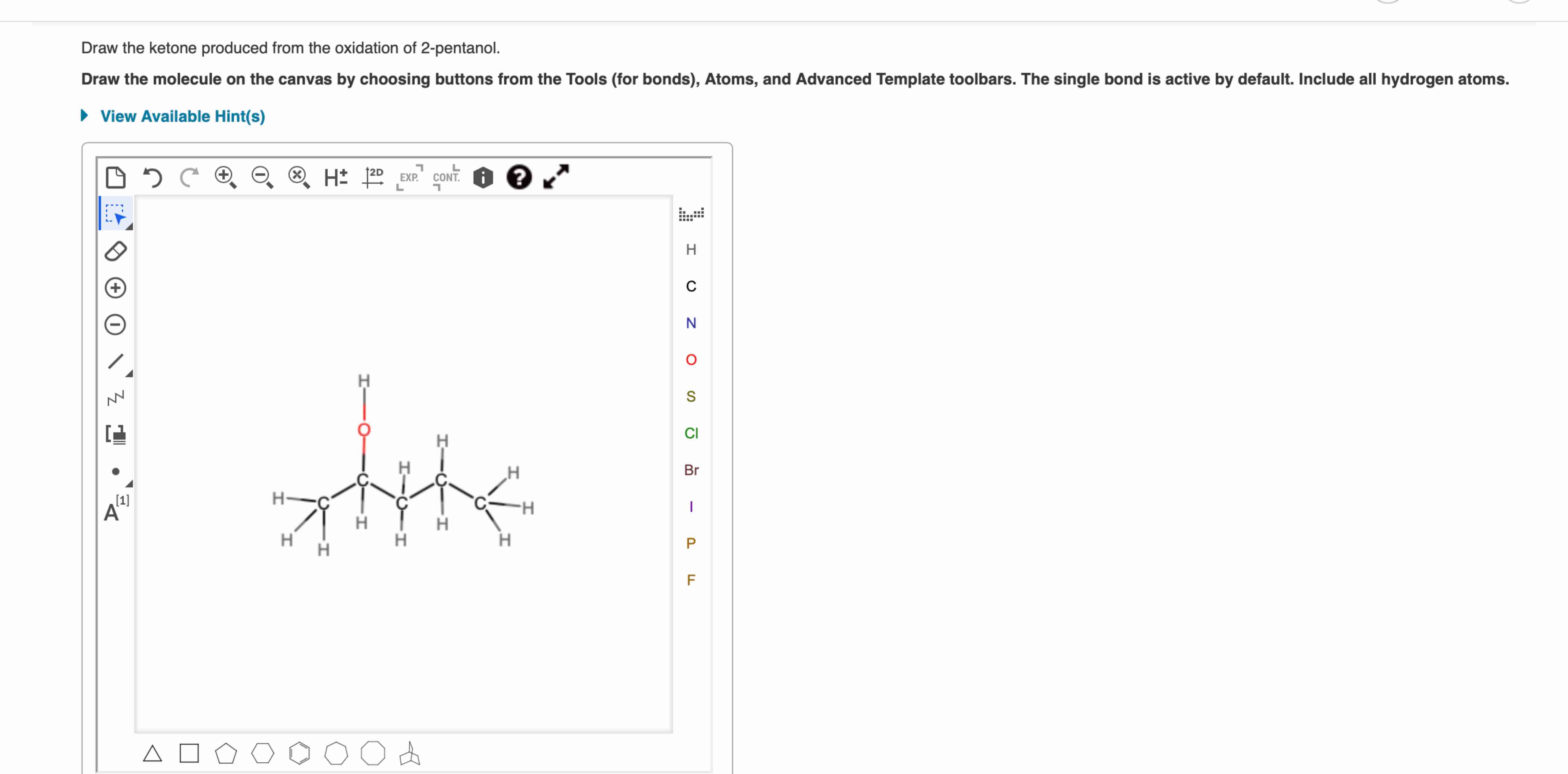
Draw The Ketone Produced From The Oxidation Of 2 Pentanol

Hey there, science curious folks! Ever wondered what happens when you give a little nudge to a simple alcohol molecule? It's like a tiny chemical party, and today we're going to peek at the guest list for one particular shindig. We're talking about 2-pentanol. Now, this isn't some fancy, complicated potion. It's a straightforward little guy, and when we put it through a process called oxidation, it undergoes a fun transformation.
Think of oxidation like giving something a bit of a zap, a chemical spark plug. It usually involves adding oxygen or taking away hydrogen. With alcohols, this process can lead to some really cool new molecules. And for 2-pentanol, the star of our show, the result is a brand new ketone. Let's dive into what that means and why it's kind of a big deal in the world of chemistry, without getting bogged down in all the heavy-duty jargon.
So, we start with 2-pentanol. Imagine a chain of five carbon atoms, like little links in a bracelet. On the second link, we've got a hydroxyl group, which is just a fancy way of saying an -OH. That's what makes it an alcohol. It's like a little handle that gives the molecule its alcoholic personality.
Now, when we oxidize 2-pentanol, something neat happens. That -OH group, along with a hydrogen atom attached to the same carbon, gets a bit of a makeover. They leave the party, so to speak, and in their place, the carbon atom forms a double bond with an oxygen atom. Ta-da! We've just created a ketone. This specific ketone is called 2-pentanone. It's like the alcohol molecule swapped its dancing shoes for a more sophisticated look.
What makes this transformation so entertaining? Well, for starters, it's a clear example of how structure dictates function. The simple act of rearranging atoms and bonds can completely change the properties and potential uses of a molecule. 2-pentanol might be a liquid with certain characteristics, but 2-pentanone, our ketone friend, can behave quite differently. It's like going from a comfortable pair of sneakers to stylish heels – they both get you around, but they do it with a different flair.
The formation of a ketone from a secondary alcohol like 2-pentanol is a fundamental reaction in organic chemistry. It's a building block for understanding more complex reactions and for synthesizing all sorts of interesting compounds. Think of it as learning your ABCs before you can write a novel. This is one of those essential ABCs.
What's so special about 2-pentanone, then? Well, ketones in general are a super versatile class of organic compounds. They're often found in natural products and are used in a huge variety of applications. Many ketones have distinct, sometimes pleasant, odors. They can be used as solvents, in perfumes, and as intermediates in the production of plastics and pharmaceuticals. So, that little molecule we drew? It's got some serious potential!
Let's talk about drawing it, because that's where the fun really begins for some of us. When you're asked to draw the ketone produced from the oxidation of 2-pentanol, you're essentially being asked to visualize that chemical transformation. You start with the structure of 2-pentanol. Remember our five-carbon chain? You draw that out, placing the -OH group on the second carbon.
Then, you imagine the oxidation happening. That -OH group on the second carbon transforms. The oxygen from the -OH group stays, but instead of being attached to a hydrogen, it now forms a double bond with the carbon. The hydrogen that was part of the -OH group? It's gone, and so is the hydrogen that was also attached to that same second carbon atom. It’s a neat little swap. The result is a carbon atom with a double bond to an oxygen atom, sitting right there on the second carbon of our five-carbon chain. That’s your ketone: 2-pentanone.
It’s like solving a tiny molecular puzzle!
The beauty of this is its simplicity and elegance. It's a predictable outcome from a specific starting material. It’s a testament to the ordered world of chemistry, where reactions follow rules and produce consistent results. And when you're the one doing the drawing, you're not just copying something; you're actively participating in the chemical process, albeit on paper.
So, why is this "entertaining" or "special"? Because it’s a gateway. It’s the moment you realize you can predict and depict chemical changes. It’s the satisfaction of understanding how one thing can become another through a controlled process. It’s a tiny victory in comprehending the molecular world around us.
Imagine you’re a detective, and the clue is "oxidation of 2-pentanol." Your job is to figure out what the culprit is. And with a little knowledge of chemical reactions, you can draw it out, identify it, and understand its potential. That’s pretty cool, right?
The actual drawing is straightforward once you get the hang of it. You have your five carbons in a row. On the second carbon, you draw a double bond to an oxygen atom. Make sure all the other carbons have their usual number of bonds filled in with hydrogens. It’s a visually clear representation of the change. You see the functional group shift, and you understand the transformation. It's not just abstract knowledge; it becomes something you can see and sketch.
This is the kind of thing that can spark curiosity. It’s a glimpse into a world where molecules have personalities and undergo transformations that are both predictable and useful. The next time you hear about oxidation or ketones, you might just think of our little friend, 2-pentanol, and its exciting journey to becoming 2-pentanone. It’s a simple reaction, but it unlocks a whole universe of chemical possibilities. So, why not grab a pen and paper and give it a try? You might be surprised at how much fun you have drawing out this little chemical adventure!
