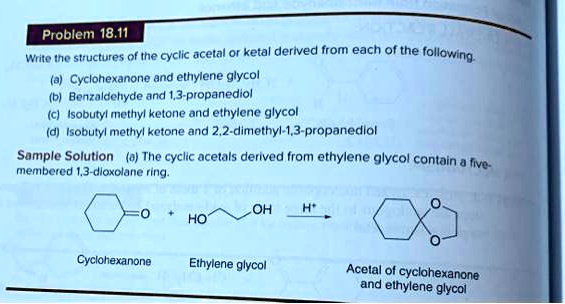
Draw The Ketal Derived From Ethylene Glycol And Cyclohexanone.

So, picture this: I’m fiddling around in my kitchen, trying to make this ridiculously complicated recipe for something I saw on a fancy cooking show. It involved, of all things, making my own vanilla extract. Yeah, I know, overkill. But the recipe called for steeping vanilla beans in high-proof alcohol for months. Months! And in my haste, I completely forgot to get the right kind of alcohol. My pantry was a wasteland of half-empty bottles of lukewarm gin and that questionable bottle of crème de menthe from my aunt’s last visit. Disaster averted? Not really. I ended up using some cheap vodka, which is fine, but it got me thinking… are there other ways to get these cool flavor compounds? Less waiting, more… chemical wizardry?
That’s when my mind, in its usual circuitous route, wandered into the fascinating world of organic chemistry. You see, sometimes the most elegant solutions aren’t about waiting for nature to do its thing, but about helping it along with a little bit of know-how. And today, we're going to do just that. We’re going to talk about a neat little reaction that’s pretty common in the lab and, believe it or not, has some pretty useful applications. We're going to draw the ketal derived from ethylene glycol and cyclohexanone. Sounds fancy, right? Don't worry, we'll break it down.
Think of it like this: you have two ingredients, ethylene glycol (which is basically just a fancy name for a type of alcohol with two hydroxyl groups, –OH, on adjacent carbons) and cyclohexanone (a ring-shaped molecule with a ketone group, C=O, right in the middle of the ring). Our mission, should we choose to accept it (and we totally are!), is to get them to hang out and form something new. Specifically, a ketal. Now, if you’re picturing them having a little cocktail party, you’re not far off. It’s a reaction where the oxygen atoms from the alcohol groups of ethylene glycol decide to get cozy with the carbon atom of the ketone group in cyclohexanone.
Before we dive headfirst into drawing, let’s get a feel for these players. Ethylene glycol, sometimes you might have seen it as antifreeze (don’t drink it, obviously!), is a simple molecule. It looks like this: HO-CH2-CH2-OH. See those two –OH groups? Those are our key players for this reaction. They’re the ones doing the heavy lifting, or rather, the heavy bonding.
And then there’s cyclohexanone. Imagine a hexagon, like the shape of a honeycomb cell, but made of carbon atoms. Now, instead of each carbon just having single bonds to its neighbors and hydrogens, one of those carbons is double-bonded to an oxygen atom. That’s your ketone group. So, it’s a six-membered ring with a C=O in it. Pretty neat, right?
Now, when you mix these two buddies together, usually with a little nudge from an acid catalyst (think of the acid as the super-enthusiastic matchmaker), something cool happens. The oxygen atoms from the ethylene glycol will attack the carbon of the carbonyl group (that’s the C=O part of cyclohexanone). And because it's a ketone, and we're using a diol (that’s an alcohol with two –OH groups), we’re going to form a ketal. Specifically, a cyclic ketal, because both –OH groups from the ethylene glycol are going to end up reacting with the same cyclohexanone molecule.
The cool part is how the structure rearranges. The double bond in the C=O of cyclohexanone breaks. One of the oxygen atoms from the ethylene glycol will bond to that central carbon, and the other oxygen from the ethylene glycol will also bond to that same carbon. And what happens to the original oxygen of the carbonyl group? It gets kicked out as a water molecule (H2O). Chemistry, folks, is all about atoms finding new and exciting relationships!
So, let's get to the drawing board, or in this case, the imaginary drawing board in your brain. We'll start with cyclohexanone. Remember that hexagon? Let’s draw it out. Make sure you’ve got that double bond to the oxygen at one of the carbons. We can represent that as C=O.

Okay, got it? It looks something like:
O
//
C
/ \
CH2 CH2
| |
CH2 CH2
\ /
CH2
Now, let's bring in ethylene glycol: HO-CH2-CH2-OH. In the presence of acid, the oxygen atoms with their lone pairs of electrons (those little electron clouds that are always looking for something to do) are going to be attracted to the positive-ish carbon atom of the carbonyl group in cyclohexanone. Remember, oxygen is more electronegative, so it pulls electrons towards itself, making the carbon it’s bonded to slightly positive.
The reaction proceeds in a few steps, but the end result is that both hydroxyl groups of ethylene glycol react with the carbonyl carbon of cyclohexanone. The oxygen from the C=O is removed as part of a water molecule. What’s left is a new ring structure formed by the cyclohexanone ring and the ethylene glycol molecule. This new structure is a cyclic ketal.
Let's visualize this. The carbon that used to have the double bond to oxygen will now be bonded to two oxygen atoms from the ethylene glycol molecule. And those two oxygen atoms will, in turn, be bonded to the carbons of the –CH2-CH2– chain from the ethylene glycol. This creates a five-membered ring fused to the original six-membered cyclohexanone ring. It’s a bit like putting a little crown on the cyclohexanone.
So, the cyclohexanone ring is still there, still a hexagon. But instead of a C=O group, that carbon is now bonded to two oxygen atoms. Each of those oxygen atoms is then bonded to a CH2 group, and those two CH2 groups are bonded to each other, forming the five-membered ring. Let's try to sketch this out. It's going to look a bit complex at first, but we'll get there.
Start with your cyclohexanone ring. Replace the C=O with a carbon atom that has two single bonds to oxygen atoms. Now, draw a chain of two carbons (CH2-CH2) connecting those two oxygen atoms. That’s your ethylene glycol part, now incorporated into a ring.
The structure will look something like this:
O - CH2
/ \
C CH2
/ \ /
CH2 CH2 O
| | |
CH2 CH2 - CH2
Wait, that’s not quite right. Let’s redraw. It’s a bit easier to visualize if you think about the original cyclohexanone ring and then how the ethylene glycol fits in. The carbon that was part of the C=O is now the center of a new ring with two oxygens and two carbons from the ethylene glycol.
Let’s try again. The cyclohexanone ring remains. The carbonyl carbon (the one that was double-bonded to oxygen) is now bonded to two oxygens. Each of those oxygens is bonded to a CH2 group. And those two CH2 groups are bonded to each other. This forms a 1,3-dioxolane ring (that’s the name for a five-membered ring with two oxygen atoms and three carbon atoms) attached to the cyclohexanone ring.
So, the cyclohexanone ring is your base. Let's say you have a carbon in that ring that was C=O. Now, that carbon is bonded to two oxygens. These oxygens are part of the ethylene glycol molecule. The structure of ethylene glycol is HO-CH2-CH2-OH. So, one oxygen connects to the carbonyl carbon, and the other oxygen connects to the same carbonyl carbon. And these two oxygens are connected to each other via the -CH2-CH2- part of the ethylene glycol.
This creates a rather stable cyclic structure. The two oxygen atoms from ethylene glycol are now bridging the carbonyl carbon. It’s like the ethylene glycol has hugged the carbonyl carbon. This is the ketal linkage.
Let's break down the drawing process more systematically. Imagine the cyclohexanone molecule. Pick any carbon in the ring. Let's call it C1. This is where our carbonyl group was. So, C1 was double-bonded to O. Now, C1 will be single-bonded to two oxygen atoms. Let's call them O_A and O_B.
These O_A and O_B are part of the ethylene glycol. So, we have O_A – CH2 – CH2 – O_B. And O_A is bonded to C1, and O_B is also bonded to C1. This forms a five-membered ring (C1, O_A, CH2, CH2, O_B). This five-membered ring is fused to the original cyclohexanone ring.
The remaining carbons of the cyclohexanone ring (let's call them C2, C3, C4, C5, C6) are still connected to each other and to C1. The carbons C2 and C6 were adjacent to C1 in the original cyclohexanone. They will remain so. The structure can be a bit tricky to draw in 2D, as we’re trying to represent a 3D molecule on a flat surface. But the key is the formation of that new five-membered ring.
Here’s a more standard way to visualize it. The cyclohexanone ring is drawn. The C=O group is replaced by a carbon atom bonded to two oxygen atoms. These two oxygen atoms are then connected by a two-carbon chain (from ethylene glycol). This essentially creates a fused ring system. The carbon atom that was the carbonyl carbon now becomes part of a dioxolane ring.
Think of it like this: The cyclohexanone is the host. Ethylene glycol is the guest. In the presence of acid, the guest checks into the host and they form a very stable partnership. The ketal linkage is particularly interesting because it’s often used as a way to protect a carbonyl group during other chemical reactions. If you have a molecule with both a ketone and other functional groups that might react under certain conditions, you can temporarily turn the ketone into a ketal. Then, once you're done with the other reactions, you can easily convert it back to the ketone.
The name of this specific ketal is 1,4-dioxaspiro[4.5]decane. Let’s break that down. "Dioxa" means two oxygens. "Spiro" indicates a spiro compound, meaning two rings share a single common atom. In this case, that common atom is the carbon that was originally the carbonyl carbon of cyclohexanone. The "[4.5]" tells you about the rings. There's a 4-membered ring (excluding the spiro atom) and a 5-membered ring (excluding the spiro atom). In our case, the dioxolane ring has 3 carbons and 2 oxygens, which, if you count the spiro atom, gives you 5 atoms in that side. The cyclohexanone ring has 6 carbons. So, the numbers refer to the number of atoms in each ring other than the shared spiro atom. The dioxolane ring has 3 carbons and 2 oxygens that aren't the spiro atom (total 5 atoms in the ring). The cyclohexanone ring has 5 carbons and the spiro atom. Wait, that interpretation of spiro notation can be tricky! Let’s simplify. The "[4.5]" refers to the number of atoms in each path from one side of the spiro atom to the other. So, one path goes through the two carbons of ethylene glycol and the two oxygens (4 atoms). The other path goes through the five carbons of the cyclohexanone ring (5 atoms).
And "decane" means there are a total of 10 carbon atoms in the structure if it were all saturated hydrocarbons. In our case, we have the cyclohexanone ring (6 carbons) and the ethylene glycol unit (2 carbons). Plus the original carbonyl carbon that is now part of the bridge. That makes 6 + 2 + 1 = 9 atoms in the core structure if you consider the spiro atom as part of both. Actually, it's more about the total number of atoms involved in the rings. The cyclohexanone has 6 carbons. The dioxolane part has 2 carbons and 2 oxygens. The spiro carbon is shared. So, if we count atoms: 6 carbons from cyclohexanone + 2 carbons from ethylene glycol = 8 carbons. Plus 2 oxygens. The "decane" part might be a bit misleading if you think of it as only carbons. It often refers to the parent hydrocarbon system.
The more important takeaway is that we've formed a ketal. And it's a cyclic ketal because the ethylene glycol molecule has created a ring structure with the cyclohexanone. This particular ketal is quite stable under neutral or basic conditions, but it can be readily hydrolyzed back to the starting materials (cyclohexanone and ethylene glycol) in the presence of aqueous acid. This reversibility is what makes ketals so useful in organic synthesis.
Let’s draw it one more time, focusing on the connectivity. Start with the cyclohexanone ring. Let's label the carbons 1 through 6, with carbon 1 being the carbonyl carbon. C1 = O (original ketone) Now, C1 is bonded to two oxygens, let’s call them Oa and Ob. Oa is bonded to C1. Ob is bonded to C1. Oa is also bonded to a CH2 group (let's call it C7). Ob is also bonded to a CH2 group (let's call it C8). C7 and C8 are bonded to each other. So, the sequence is C1 – Oa – C7 – C8 – Ob – C1. This forms the five-membered dioxolane ring. The original cyclohexanone carbons C2, C3, C4, C5, C6 are still bonded in sequence, and C2 is bonded to C1 and C3, C6 is bonded to C1 and C5. A common way to represent this is to draw the cyclohexanone ring, and at one carbon, replace the double bond to oxygen with two single bonds to oxygen atoms. These two oxygen atoms are then connected by a -CH2-CH2- chain, forming the five-membered ring. The overall shape is like the cyclohexanone ring with a little dioxolane ring "stitched" onto one of its carbons.
It’s pretty cool, isn't it? From simple starting materials, you get this new, more complex molecule. And the process is quite general. You can make ketals from ketones and various diols, or from aldehydes and diols (which would be called acetals, but the principle is very similar). The key is the reaction of the carbonyl group with alcohol functionalities. So, the next time you’re thinking about complex chemical structures, remember that sometimes, it’s just about bringing the right molecules together for a little chemical embrace. And in this case, that embrace results in a rather elegant cyclic ketal!
