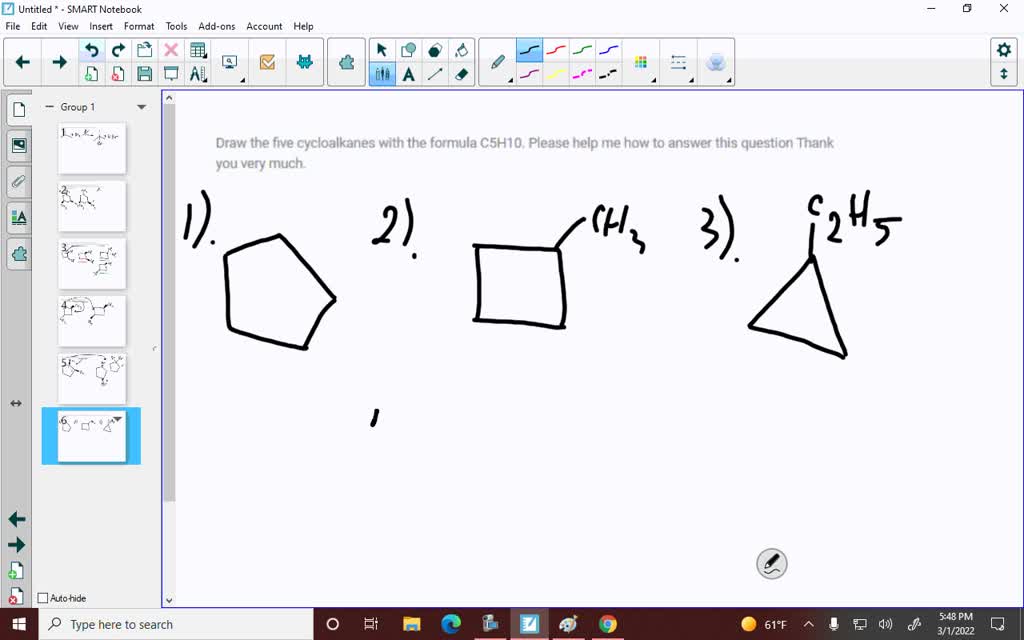
Draw The Five Cycloalkanes With The Formula C5h10

Hey there, science explorers! Ever feel like the world of chemistry is a bit… well, intimidating? All those fancy formulas and complex names can make you want to run for the hills, right? But what if I told you that even something as seemingly dry as drawing some molecules can actually be a blast? Yep, you heard me! Today, we're diving into the wonderfully weird world of cycloalkanes, and specifically, the five different ways you can arrange five carbon atoms and ten hydrogen atoms into a ring. Intrigued? You should be!
So, what exactly are cycloalkanes? Think of them as carbon atoms forming a cozy little circle. Instead of a long chain, they decide to hold hands and form a loop. And the formula we're playing with today is C5H10. This means we've got five carbon buddies and ten hydrogen sidekicks. Our mission, should we choose to accept it (and trust me, you totally should!), is to find all the distinct shapes these five carbons can make when they link up in a ring.
This isn't just about memorizing boring diagrams, oh no! This is about unleashing your inner artist and your problem-solving superpowers. It's like a molecular puzzle, a geometric challenge, and a tiny peek into the incredible diversity of the universe, all rolled into one. Think of it as a molecular LEGO set – you've got your basic bricks (the carbon atoms) and you've got to figure out all the cool things you can build with them, keeping the hydrogen count just right.
The Grand Unveiling: Our Five Ring Buddies!
Alright, drumroll please! We're about to meet the five unique cycloalkanes with the formula C5H10. Each one is a distinct structural isomer, meaning they have the same atoms but are connected in a different order. It's like having five different breeds of dogs, all with the same number of legs, but looking and acting completely unique!
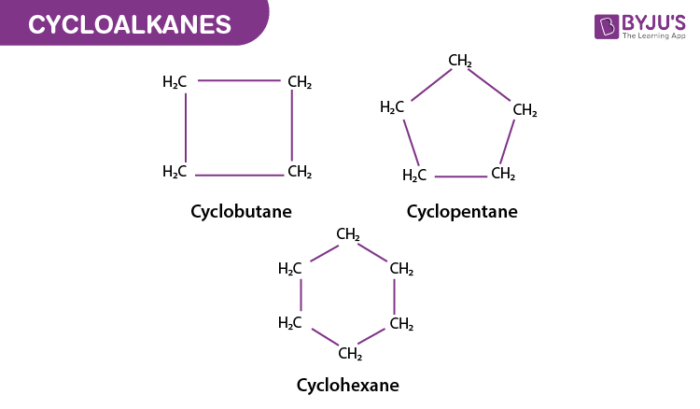
1. The Classic: Cyclopentane
Let's start with the most straightforward one. Imagine five carbon atoms, all linked together in a perfect, undisturbed pentagon. No fancy branches, no unusual connections, just a simple, happy ring. This is our star player, cyclopentane. Each carbon atom in this ring is bonded to two other carbons and two hydrogen atoms. It's the foundational shape, the classic car of our C5H10 family. Easy peasy, right? You can practically draw this one with your eyes closed!
Why is it so cool? Well, it's the simplest way to form a five-membered ring. It's like learning to ride a bike with training wheels – it gets the job done and sets you up for more advanced cycling (or molecular building!). Plus, cyclopentane is found in nature, contributing to the scent of things like coffee. So, next time you enjoy a good brew, you can give a little nod to our friendly pentagonal friend!

2. The Not-So-Little Ring: Cyclopropane and Cyclobutane (Wait, What?)
Now, before you get too comfortable, you might be thinking, "But what about smaller rings?" And you'd be right to wonder! With C5H10, we're focusing on the cycloalkanes, which means the carbons are in the ring. So, while we can make smaller rings with fewer carbons, we'd need extra carbon atoms somewhere else to reach our C5H10 total. For instance, you could have a cyclopropane ring (3 carbons) with two extra carbon atoms attached as a side chain. Or a cyclobutane ring (4 carbons) with one extra carbon atom hanging off. These are also valid isomers of C5H10!
Let's break these down, because they add a whole new dimension to our drawing adventure. Think of it as adding accessories to your basic ring. For cyclopropane with side chains, you could have the two extra carbons together as an ethyl group, or one on each of two different carbons. For cyclobutane, you have one extra carbon to play with. These arrangements might be a bit trickier to visualize at first, but they're just as important!
3. The Branched Beauty: Methylcyclobutane
Okay, let's focus on the cyclobutane ring (that's four carbons making a square). Now, where do we put that fifth carbon to get C5H10? We can attach it as a single carbon group, called a methyl group, to one of the carbons in the cyclobutane ring. This gives us methylcyclobutane. Imagine a little flag sticking out from one corner of your square! It’s still a ring, but with a little extra flair.
This isomer is super interesting because it shows how adding just one atom in a different spot can create a whole new molecule. It’s like changing one ingredient in a recipe – suddenly, you have a different dish! And isn't that exciting? The subtle differences can lead to vastly different properties. So, while it might look like a simple addition, it’s a crucial piece of our C5H10 puzzle.
4. The Other Branched Beauty: Ethylcyclopropane
Now, let's think about our cyclopropane ring (three carbons in a triangle). We've got two more carbons to play with to reach C5H10. We can link these two carbons together and attach them as a group to one of the carbons in the cyclopropane ring. This group of two carbons is called an ethyl group. So, we have ethylcyclopropane! Picture a tiny triangle with a little two-carbon tail wagging from one of its points.
This is where things get really fun. You can even attach those two carbons individually to different carbons on the cyclopropane ring! This gives us another isomer called dimethylcyclopropane. Imagine our little triangle with two tiny flags. And here's the kicker: those flags can be on the same carbon, or on different carbons, and if they're on different carbons, they can be next to each other or on opposite carbons. This is where the "five" isomers really start to show their magic!
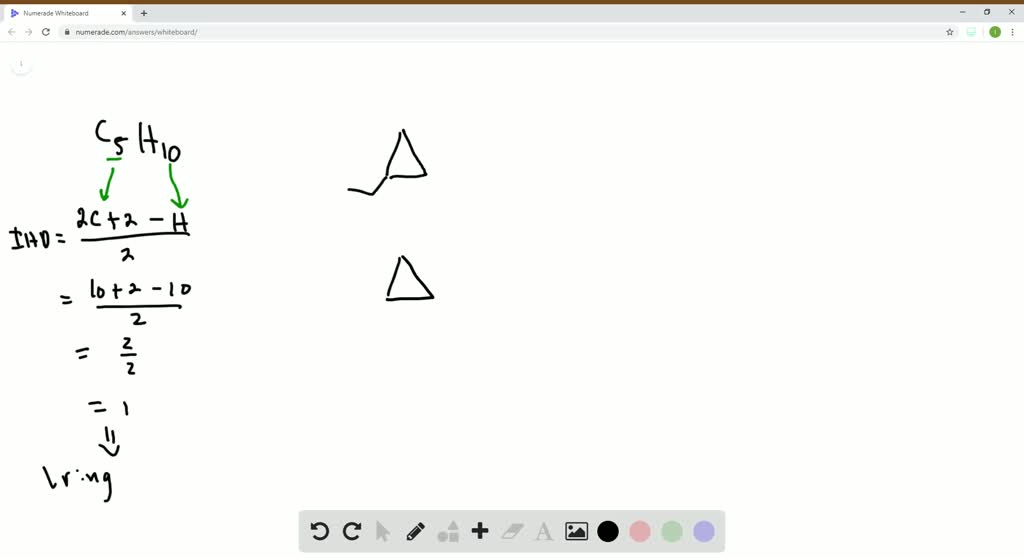
5. The Double Trouble Duo: Dimethylcyclopropanes (Yes, Plural!)
Ah, this is where the true diversity of C5H10 shines! Remember our cyclopropane ring? We have two carbons left to play with. We can attach these two carbons as methyl groups (single carbon units) to the cyclopropane ring. Now, this is where it gets exciting because there are three different ways to do this!
- 1,1-Dimethylcyclopropane: Both methyl groups are attached to the same carbon atom on the cyclopropane ring. Imagine our little triangle with two little flags sprouting from the same corner.
- 1,2-Dimethylcyclopropane (cis): The two methyl groups are attached to adjacent carbon atoms on the ring, and they are on the same side of the ring. Think of our triangle with two flags, one on each of two connected corners, both pointing upwards.
- 1,2-Dimethylcyclopropane (trans): The two methyl groups are attached to adjacent carbon atoms on the ring, but they are on opposite sides of the ring. Now, imagine our triangle with two flags, one on each of two connected corners, but one points up and the other points down.
See? Even with the same basic ring and the same number of extra atoms, the positioning can change everything! This is the essence of isomerism, and it’s a fundamental concept in chemistry that explains the vast complexity and variety of organic molecules. It’s like different arrangements of the same musical notes creating entirely different melodies!
Why Draw Molecules Anyway?
You might be asking yourself, "Why bother drawing these things?" Well, my friends, drawing molecules is like learning the alphabet before you can read a book. It helps you visualize what's actually happening at the molecular level. It's how chemists understand how molecules will behave, how they'll react with other molecules, and what their properties will be.
And honestly? It’s a fantastic way to engage your brain. It's a mix of logic, spatial reasoning, and a dash of creativity. It’s a mental workout that can be incredibly satisfying. Plus, when you can draw these structures accurately, you're unlocking a whole new language – the language of chemistry! Think of it as gaining a secret code that allows you to understand the building blocks of everything around you.
Learning to draw these cycloalkanes is just the tip of the iceberg. It's an invitation to explore further, to understand the principles behind molecular structure and bonding, and to appreciate the elegance and complexity of the chemical world. So, don't shy away from these diagrams. Embrace them! See them as little puzzles waiting to be solved, and as beautiful representations of the tiny, invisible world that makes up our reality.
Go Forth and Draw!
So, there you have it! The five distinct cycloalkanes with the formula C5H10. From the simple pentagon of cyclopentane to the slightly more complex branched structures, each one is a testament to the infinite possibilities that arise from just a few atoms. Isn't that incredible? The universe is full of these amazing arrangements, and understanding them, even in a small way, is a superpower!
The next time you encounter a chemical formula, don't feel overwhelmed. Think of it as an invitation to draw, to explore, and to discover. You've got the tools, you've got the imagination. So, grab a pen and paper, or fire up some molecular modeling software, and start drawing! You might just find that chemistry is a lot more fun and inspiring than you ever imagined. Keep that curiosity alive, and you'll be amazed at what you can learn and create!
