Draw The Ether With The Common Name Phenyl Propyl Ether
Hey there, science curious friends! Ever feel like some chemical names sound like they belong in a sci-fi movie or a wizard's spellbook? You know, like something you'd whisper dramatically before brewing a potion? Well, today, we're going to demystify one of those names and, believe it or not, it's actually pretty cool and, dare I say, fun to talk about. We're diving into the wonderfully (and perhaps slightly mysteriously) named: Phenyl Propyl Ether. Sounds fancy, right? Like it might be made of stardust and dreams. But hold onto your lab coats, because it's a lot more down-to-earth than it seems!
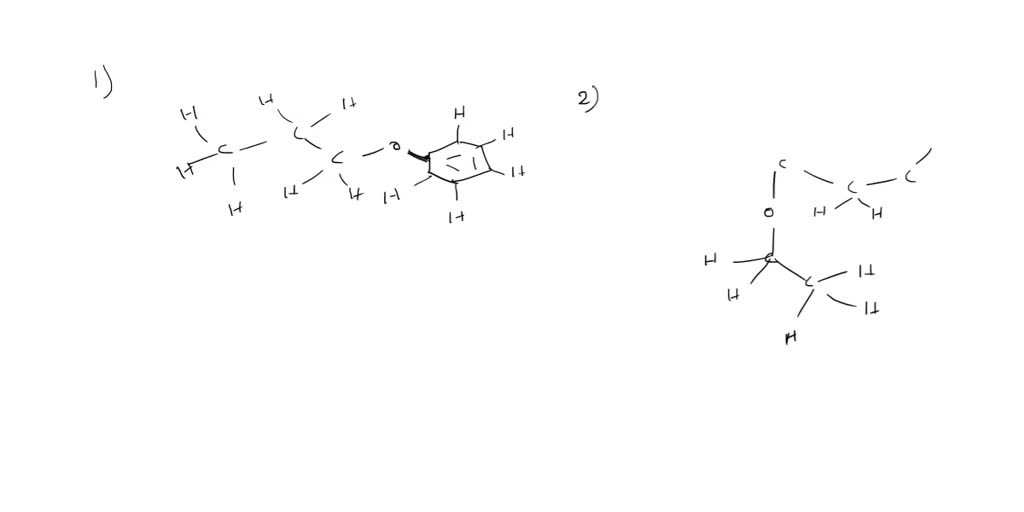
So, what exactly is this "phenyl propyl ether" business? Think of it like this: we're talking about a molecule, a tiny, tiny building block of stuff. And this particular molecule has a couple of key parts. We've got the "phenyl" bit, which comes from something called benzene. Benzene is a ring of six carbon atoms, and it's a really common and important structure in organic chemistry. Think of it as the sophisticated, slightly aloof cousin in the molecular family. It's got this neat, flat, six-sided shape. You'll find phenyl groups in all sorts of things, from drugs to dyes to… well, our friend here!
Then we have the "propyl" part. This refers to a chain of three carbon atoms. It's like the friendly, energetic younger sibling of the carbon chains. Three carbons in a row, nothing too complicated. It's just a straight line of them. Think of it as a little molecular caterpillar. So, we have our distinguished phenyl ring and our little propyl caterpillar. They sound like they're about to go on an adventure, don't they?
And the "ether" part? Ah, this is where the magic (or rather, the chemistry!) happens. In chemistry, an ether is a functional group that has an oxygen atom bonded to two carbon atoms. So, picture our phenyl ring and our propyl chain, and smack dab in the middle, connecting them, is an oxygen atom. It's like the diplomat who brings two different worlds together. So, we have a phenyl group attached to one side of the oxygen, and a propyl group attached to the other. And voilà! You've got yourself a phenyl propyl ether. See? Not so scary after all. It’s just a matter of putting the right pieces together.
Let's Break It Down a Smidge More (Without Getting Too Wiggly)
Okay, so we know the parts. But what does it do? Well, phenyl propyl ether is, at its core, an organic compound. That just means it's made primarily of carbon and hydrogen, with some other bits and bobs thrown in. In this case, our "bit" is that crucial oxygen atom.
The "phenyl" part (from benzene) gives it some interesting properties. Benzene itself is quite stable, and when you attach it to other things, it tends to lend them a certain… je ne sais quoi. It can influence how the molecule interacts with other molecules, how soluble it is (can it dissolve in water or oil?), and even its smell. Speaking of smell…

Many ethers, especially those with aromatic rings like our phenyl group, have a distinct aroma. Now, "distinct" can mean anything from "mildly pleasant" to "might clear your sinuses." Phenyl propyl ether is often described as having a sweet, somewhat floral, or even fruity scent. Imagine a subtle hint of a garden after a light rain, mixed with a whisper of something a little more exotic. It’s not going to be as overpowering as, say, your grandma’s potent perfume, but it can add a pleasant nuance. This is why some of these types of compounds find their way into the fragrance industry, making things smell… well, nice.
So, Where Does This Little Guy Hang Out?
Phenyl propyl ether isn't exactly something you'll find lying around on your kitchen counter, unless you happen to be a mad scientist with a penchant for fine fragrances. It's more likely to be found in a laboratory, or perhaps as a component in a more complex mixture. It’s a building block, a cog in a larger molecular machine.
You might encounter it, or compounds very similar to it, in a few different places:

- Flavors and Fragrances: As we touched upon, its scent makes it a candidate for perfumes, colognes, and even some artificial flavorings. Think of those little scent strips in magazines – some of those molecules might be distant cousins of our phenyl propyl ether. It's all about creating those sensory experiences!
- Chemical Synthesis: In the world of organic chemistry, it can be used as a solvent or as an intermediate in the creation of other, more complex molecules. It’s like a helpful tool for chemists, a way to get from point A to point B in a chemical reaction. Sometimes you need a specific ingredient to help another ingredient behave the way you want it to.
- Research and Development: Scientists are always tinkering, trying to create new materials, new medicines, new ways to do things. Compounds like phenyl propyl ether are part of that vast exploration. Who knows what amazing discoveries are waiting to be made involving molecules like this?
It’s important to remember that while it has a pleasant name and can contribute to lovely scents, like all chemicals, it should be handled with care. Safety first, always! But that doesn't diminish its interesting nature.
The Chemistry Nerds' Secret Handshake: Naming Conventions
Let’s have a quick peek at the name itself again: Phenyl Propyl Ether. It's a classic example of how chemists name things based on their structure. It’s like a little chemical ID card!
The "phenyl" comes first because it's often considered the more complex or important substituent in this type of ether. Then comes "propyl," indicating the three-carbon chain. And finally, "ether," telling us about that oxygen bridge. It's a logical system, even if it takes a bit of getting used to.

Sometimes, you might see it written with some Greek letters or numbers attached, like n-propyl phenyl ether. The 'n-' just means it's a normal or straight-chain propyl group. If it were a branched chain, it might have a different prefix. It’s all about precision! Imagine trying to give directions without being specific – you’d end up with lost friends and confused delivery drivers. Chemistry names are just highly specific directions for molecules.
There are also isomers of phenyl propyl ether. Now, an isomer is like a molecule that has the same ingredients (the same atoms in the same proportions) but arranged differently. Think of Lego bricks – you can build a car or a house with the same set of bricks. So, you could have the oxygen attached differently, or the propyl group could be branched. These different arrangements would give the molecules slightly different properties. It’s a whole world of subtle variations, and it’s what makes chemistry so endlessly fascinating!
Why Should You Care About This Ether-y Thing?
You might be thinking, "Okay, it smells nice and chemists use it. Big deal." But here's the beautiful thing about chemistry: everything is made of molecules. From the air you breathe to the coffee you drink to the device you’re reading this on, it’s all molecular magic. Understanding even a seemingly obscure molecule like phenyl propyl ether gives you a tiny glimpse into the incredible complexity and interconnectedness of the world around us.
It’s a reminder that even the most mundane objects have fascinating chemical stories behind them. And when you hear a name like "phenyl propyl ether," instead of feeling intimidated, you can think, "Ah, that’s the one with the sophisticated phenyl cousin and the friendly propyl caterpillar, linked by a wise old oxygen!"
It’s like learning a few words in a new language. Suddenly, you can understand a little bit more of the conversation happening all around you. Every chemical name, every reaction, is a piece of a much larger, incredibly intricate puzzle that makes up our universe. And by learning about one piece, you’re getting closer to appreciating the whole magnificent picture.
The Takeaway: A Whiff of Wonderful
So, there you have it! Phenyl propyl ether, demystified. It’s not a mythical beast, but a rather neat organic compound with a pleasant aroma and a useful role in the world of chemistry. It’s a perfect example of how seemingly complex scientific terms can be broken down into understandable parts, revealing the elegant logic behind nature’s building blocks.
Next time you catch a whiff of something sweetly floral or subtly fruity in a perfume, or if you ever find yourself reading a chemistry textbook (perhaps with a cup of tea and a biscuit!), take a moment to think about molecules like phenyl propyl ether. They are the unsung heroes, the quiet contributors to our sensory experiences and the progress of science. They remind us that even in the smallest structures, there's a world of wonder waiting to be discovered. So go forth, my friends, and embrace the delightful, the intriguing, and the ever-so-slightly ether-y world of chemistry! Keep exploring, keep learning, and always keep that smile of curiosity shining bright!
