Draw The Enone Product Of Aldol Self-condensation Of 3-pentanone.

Alright, settle in, grab your imaginary latte, because we're about to dive headfirst into a culinary adventure of the chemical kind. Today's special isn't a croissant, it's the aldol self-condensation of 3-pentanone. Sounds fancy, right? Like something you'd order at a Michelin-starred molecular gastronomy place. But trust me, it's way more fun than watching a chef meticulously arrange microgreens.
So, what in the name of all that is holy is 3-pentanone? Think of it as a little ketone buddy, a five-carbon chain with an oxygen hanging out in the middle, doing its ketone thing. It's got a bit of a fruity, acetone-like aroma, so if your science lab smells faintly of nail polish remover, you're probably in the right neighborhood. Now, 3-pentanone is a bit of a loner, but it's also secretly a social butterfly. It likes to hang out with other 3-pentanone molecules, and today, it's throwing a party. A very specific kind of party.
This party is called an aldol self-condensation. Don't let the big words scare you. "Aldol" is a portmanteau, like "brunch" or "motel," but way cooler. It's a mashup of aldehyde and alcohol. And "self-condensation"? That just means our little 3-pentanone is getting cozy with itself. It's like a molecule doing a solo karaoke session and somehow forming a duet.
To get this party started, we need a catalyst. Think of it as the DJ for our molecular rave. Usually, it's either a strong base (like sodium hydroxide, which is basically a chemical diva) or a strong acid (which is just a grumpy proton with a mission). Let's imagine we're using a base, a friendly little hydroxide ion, OH⁻. This tiny powerhouse is the spark that ignites the whole shebang.
The first step is like our 3-pentanone doing a little stretch. The base comes along and plucks off a hydrogen atom from a carbon that's next to the carbonyl group (that's the C=O bit). This carbon, with its missing hydrogen, now has a negative charge and becomes what we call an enolate ion. It's like the molecule suddenly develops a fabulous, slightly rebellious attitude. This enolate ion is the star of our show, the prima donna ready to steal the spotlight.
Now, here's where the "self" part of self-condensation really kicks in. Our newly formed, very enthusiastic enolate ion, with its negative charge practically screaming "pick me!", goes looking for a party favor. And what's more convenient than another unsuspecting 3-pentanone molecule nearby? It's like finding a willing dance partner at a party where you thought you'd be going stag.
The enolate ion, being the electrophilic party-crasher it is, attacks the carbonyl carbon of another 3-pentanone molecule. Remember that carbonyl carbon? It's got a slight positive charge, making it a tasty target for our electron-rich enolate. This is the chemical equivalent of a dramatic embrace. The two molecules, once separate, are now joined. It's a beautiful, albeit temporary, union.
This initial joining creates a new molecule, which, after a bit of proton shuffling (think of it as a molecule getting its hair done), forms a beta-hydroxy ketone. It's got both an alcohol group (-OH) and a ketone group. It's like our 3-pentanone decided to go to the party and came back with a plus-one and a new hobby. This beta-hydroxy ketone is the result of the aldol addition reaction, the first act of our chemical play.

But the party isn't over yet! The condensation part of this whole shebang comes next. Beta-hydroxy ketones are a bit like teenagers – they get bored easily and love drama. Under the influence of heat or the same catalyst, they tend to lose a molecule of water. Yes, water! The stuff we drink! It's being ejected from our happy couple like a grumpy ex from a VIP lounge.
The loss of water is a crucial step. The hydrogen atom from the alcohol group and a hydrogen atom from the alpha-carbon (that's the carbon next to the carbonyl, the one that was so eager to lose its hydrogens earlier) are yanked away, forming H₂O. And where that water molecule used to be, a beautiful double bond springs up between the two carbons. It's like a fresh tattoo, permanently marking their union, but in a much more chemically stable way.
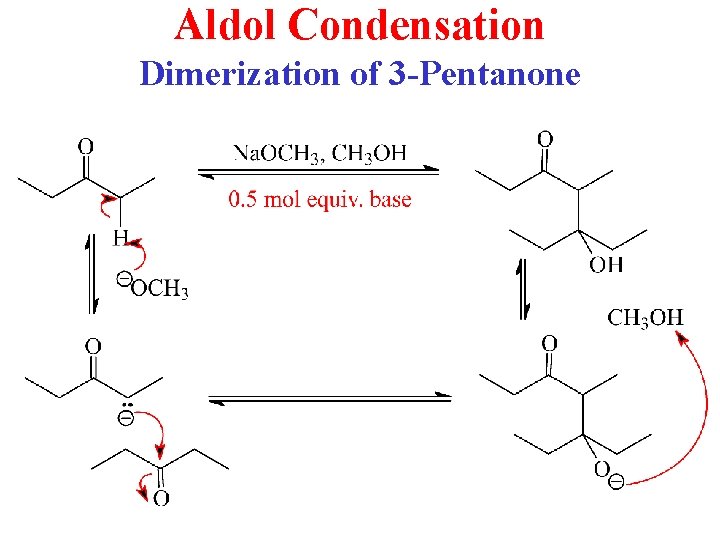
And voilà! We have our enone product. An enone is a molecule that has both a carbon-carbon double bond (the "ene") and a carbonyl group (the "one"). In our case, because we started with 3-pentanone, we're going to get something like 3,5-nonadien-2-one. (Okay, technically, if we're being super precise, and who wants that when there's drama to be had? It's more likely to be 3-propyl-4-nonen-3-one or something similar if we're talking about the product of self-condensation. The naming can get a bit hairy, like trying to find your car keys in a messy room. But the key takeaway is we've created a molecule with a double bond conjugated to a carbonyl group.)
This enone product is pretty neat. It's a more stable molecule, and that double bond is itching to react with all sorts of other things. It's like our 3-pentanone went from being a wallflower to the life of the party, not just once, but twice! It's a master of disguise, a chemical shapeshifter, all thanks to a little nudge from a base and its own inherent molecular urges.
So, next time you're smelling acetone, remember the epic romance, the dramatic attack, and the water-shedding breakup that could be happening in your environment. It’s a reminder that even the simplest molecules can have the most exciting social lives. It's a chemical soap opera, playing out right under our noses. And the enone product? That's just the happily-ever-after, or at least, the chemically-stable-for-now ending to this thrilling tale of 3-pentanone's self-love.
