Draw The Electron Dot Formula For The Element Sulfur

Hey there, budding science enthusiasts and curious minds! Ever looked at a random element on the periodic table and thought, "What on earth is going on there?" Well, get ready to have your mind tickled, because today we're diving into the wonderfully whimsical world of sulfur and its electron dot formula. Yep, you heard that right. We're going to draw the electron dot formula for sulfur, and I promise, it's way more fun than it sounds. Think of it like giving an atom a little personality, a visual representation of its tiny, energetic self!
Now, before you start picturing tiny, grumpy dots, let's loosen up a bit. This isn't about complex equations or staring blankly at textbooks. This is about unlocking a little secret about how atoms behave, and trust me, understanding these atomic quirks can make the whole universe feel a little more knowable and a lot more exciting. It’s like learning a secret handshake for matter itself!
Sulfur: The Underappreciated Star
So, who is this sulfur character? You might know it as that distinct, sometimes… fragrant smell associated with volcanoes or even rotten eggs. (Don't worry, we'll get to why it smells like that later – it's all part of the fun!). But sulfur is so much more than just a stinky bystander. It's a crucial element found everywhere, from the proteins in your body to the fertilizers that help grow your food. It's a true workhorse of the universe, quietly doing its important jobs.
Sulfur’s symbol on the periodic table is a simple 'S'. Easy enough, right? But what really makes sulfur tick, and what we're going to visualize today, are its valence electrons. These are the outermost electrons, the ones that are always looking to mingle and form connections with other atoms. Think of them as the atom’s social butterflies, fluttering around and looking for dance partners.
The Magic of Valence Electrons
Why are these valence electrons so special? Because they're the key to chemical bonding. They're the reason why atoms decide to stick together and form molecules, creating everything from the water we drink to the air we breathe. Without them, the world as we know it simply wouldn't exist. Pretty mind-blowing for a few tiny particles, wouldn't you say?
Each element has a specific number of valence electrons, and this number dictates its chemical personality. It's like assigning personality traits to our atomic friends. Some atoms are happy to hang out alone, while others are desperately seeking a partner to complete their outer shell. It's all about achieving a state of happy stability, or as chemists like to call it, a full outer electron shell.
Unveiling Sulfur's Electron Dot Formula
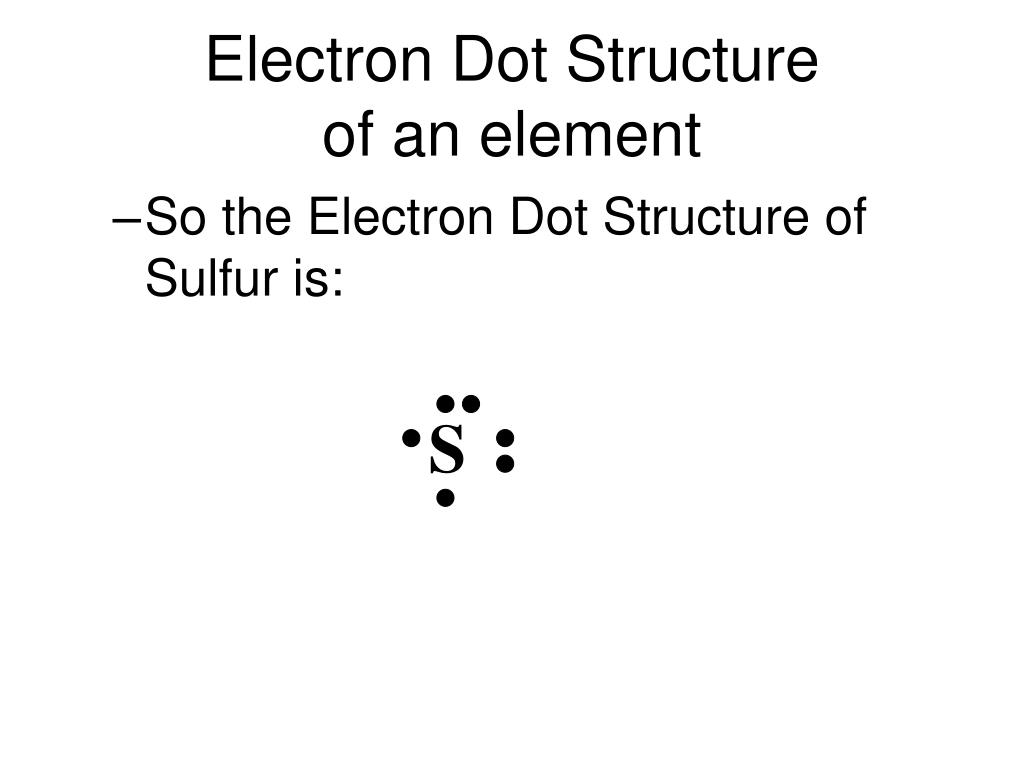
Alright, let's get down to business! To draw the electron dot formula for sulfur, we first need to know how many valence electrons it has. This is where our trusty friend, the periodic table, comes in. If you peek at sulfur (atomic number 16), you'll find that it’s in Group 16. For most elements, the group number tells you the number of valence electrons. So, sulfur has six valence electrons.

Now for the drawing part! It's super simple. We write the element's symbol, which is 'S' for sulfur, and then we represent each valence electron with a dot. We usually place these dots around the symbol, one on each of the four sides (top, bottom, left, right) before we start doubling up. It’s like placing guests around a dinner table, ensuring everyone gets their own spot first.
So, for sulfur, we'll have our 'S' in the middle. Then, we'll place one dot on the top, one on the bottom, one on the left, and one on the right. That’s four dots placed. We still have two more valence electrons to go. Now, we start pairing them up. So, we can add a second dot to any of the sides that already has one. Let's say we add the fifth dot next to the first dot on the top, and the sixth dot next to the dot on the bottom. And voilà!
What Does It Mean?
You’ve just drawn the electron dot formula for sulfur! It looks something like this (imagine it, because I can’t actually draw here, but you can visualize it!):

Imagine the 'S' symbol. Then, imagine four dots surrounding it, one on each side. Now, imagine two of those sides have a second dot next to the first one. So you have two sides with single dots, and two sides with double dots.
So, why is this significant? Well, those six valence electrons are sulfur's way of interacting with the world. It needs two more electrons to achieve a full outer shell of eight (that magic number for stability!). This means sulfur is eager to gain, share, or even borrow those missing electrons. This eagerness is what drives chemical reactions and allows sulfur to form all sorts of amazing compounds. It's like a puzzle piece that's almost complete, always looking for the right fit.
Making Chemistry Fun
See? Not so scary, right? In fact, it’s quite elegant! This little visual representation tells us so much about sulfur's behavior. It's the secret language of atoms, and once you learn a bit of it, the world around you starts to make a whole lot more sense. Think about it: the next time you encounter a sulfurous smell, you can casually nod to yourself and think, "Ah, yes, that’s sulfur and its six social butterflies looking for a couple more friends to feel complete!"
This is just the tip of the iceberg, of course. Every element has its own electron dot formula, its own unique way of interacting. Learning these formulas is like collecting a set of cosmic trading cards, each one revealing a little more about the building blocks of our universe. It’s a journey of discovery that can make everyday observations suddenly much more fascinating. Who knew a few dots could be so powerful?
Embark on Your Atomic Adventure!
So, I hope this little dive into sulfur's electron dot formula has sparked your curiosity. It's a small step, but it opens the door to understanding so much more. The world of chemistry is full of these delightful little insights, waiting for you to uncover them. Don't be afraid to pick up a periodic table, try drawing a few more electron dot formulas, and see what secrets you can unlock. Embrace the wonder, have fun with it, and remember that even the smallest things can hold the most amazing stories. Happy dot-drawing, and may your atomic adventures be ever inspiring!
