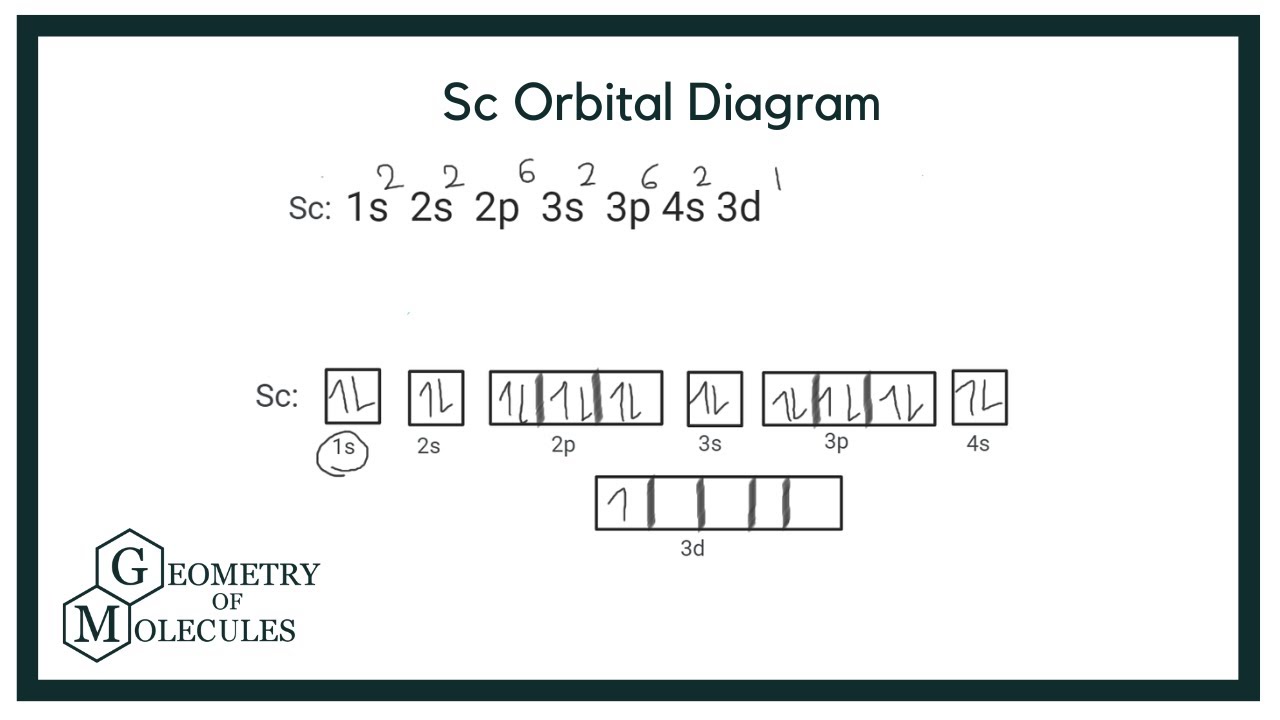
Draw The Electron Configuration For A Neutral Atom Of Scandium
Imagine atoms as tiny, bustling cities, each with its own unique blueprint. Today, we're going to peek inside the city limits of a rather special atom called Scandium. Now, Scandium might not be a household name like Gold or Iron, but it's got a bit of a quirky personality and a surprisingly important job in the grand scheme of things. Think of it as the unsung hero of the periodic table, always working diligently behind the scenes.
Our mission, should we choose to accept it (and we absolutely should, because it's fun!), is to draw its electron configuration. Don't let that fancy term scare you. Think of it like mapping out the neighborhoods where the tiny, energetic residents – the electrons – live within the atom. These electrons aren't just randomly scattered; they have specific addresses, organized into different shells and subshells, much like apartments in a skyscraper or houses on a street.
So, let's start sketching our Scandium city. We know Scandium is a neutral atom, which means it's perfectly balanced, like a happy little kid who has the same number of toys as their sibling. This means the number of positive charges in its center (the nucleus) is exactly matched by the number of negative electrons buzzing around it. For Scandium, this magic number is 21. That's right, 21 electrons are ready to find their homes!
We fill these "homes" in a specific order, like filling up a concert hall from the front row to the back. The first shell, closest to the nucleus, is like the VIP section. It can only hold a couple of electrons, and they go into a spot called the 1s subshell. So, we've got 1s2. Two electrons have found their cozy spots. Easy peasy!
Next, we move to the second shell. This one's a bit bigger. It has two types of "neighborhoods": the 2s subshell and the 2p subshell. The 2s can take another two electrons, so we add 2s2 to our map. Now we have 2 + 2 = 4 electrons settled. We still have plenty of room and lots more electrons to place. The 2p subshell is a bit more spacious, like a whole block of apartments. It can hold up to six electrons. So, we fill it up: 2p6. Now we have 4 + 6 = 10 electrons comfortably housed.

We're halfway there with our Scandium city! We’ve used up the first two shells. Time to move onto the third shell. This shell is like the sprawling suburbs of our atom. It's got a 3s subshell, a 3p subshell, and – here’s where it gets interesting for Scandium – a 3d subshell! We'll start filling these just like we did before.
First, the 3s subshell takes its two electrons: 3s2. Now we have 10 + 2 = 12 electrons placed. Then, the 3p subshell gets its maximum of six electrons: 3p6. We're up to 12 + 6 = 18 electrons. Wow, the third shell is really filling up!

We’ve now accounted for 18 electrons, and we still have 21 - 18 = 3 electrons left. These last few electrons are the adventurous ones, the explorers of our Scandium atom. They’re going to move into that new neighborhood we mentioned, the 3d subshell. The 3d subshell is a bit special; it can hold up to ten electrons. Since we only have three left, they’ll take up residence there. So, we add 3d3.
And there you have it! The complete electron configuration for a neutral Scandium atom, all mapped out. It looks like this:

1s2 2s2 2p6 3s2 3p6 3d3
But wait, there's a little twist, a humorous quirk that makes Scandium even more endearing. You see, the electrons in the 3d subshell are a bit of a late bloomer. They actually start filling up before the 4s subshell, even though the 4s is technically in the next "shell" outwards. It's like the mailman delivering packages to the 3d neighborhood first, even though the 4s houses are technically closer to the street. It’s one of those little eccentricities of the atomic world that makes it so fascinating. It’s like nature is saying, “Just a little detour here, folks!”
So, while our initial drawing might show the 3d subshell last, a slightly more accurate representation of how they fill would involve the 4s subshell. But for a simple drawing, the configuration above is perfectly understandable and shows where all 21 electrons call home. It's this kind of unexpected behavior that keeps chemists and physicists on their toes, discovering new things about the tiny worlds around us. Scandium, with its slightly mischievous electron arrangement, is a perfect example of how even the smallest parts of the universe can have their own unique stories and charm. It's a reminder that even in the most ordered systems, there's always room for a little delightful surprise.
