Draw The Correct Structure For Acetaldehyde Or Ethanal.

Hey there, gorgeous! Ever found yourself staring at a chemical name and thinking, "What in the world is that?" Yeah, us too. Life’s too short for complicated jargon when we can break things down into something totally manageable, maybe even a little bit chic. Today, we're diving into the wonderfully simple world of acetaldehyde, also known as ethanal. Think of it as the friendly, approachable cousin in the aldehyde family. We're going to chat about what it looks like, why it matters, and how understanding its structure can be as satisfying as finding the perfect shade of lipstick. So grab your favorite mug, settle in, and let's get our molecule-drawing game on point.
First off, let's talk about the name. "Acetaldehyde" sounds a bit like something you'd hear in a retro sci-fi flick, right? But "ethanal" is actually a bit more telling if you're a chemistry nerd (and even if you're not, you can totally appreciate the logic). The "eth" part tells us we're dealing with a two-carbon chain. Think of it like having two little building blocks linked together. The "anal" ending? That's the giveaway for an aldehyde. Aldehydes are a class of organic compounds that have a specific functional group: a carbonyl group (that's a carbon atom double-bonded to an oxygen atom, C=O) at the end of a carbon chain. It’s like the signature flourish, the little detail that makes an outfit.
The Star of the Show: The Functional Group
So, let's zoom in on this aldehyde group, the hero of our story. This C=O is where all the action happens. It's polar, meaning the oxygen is a bit more electronegative and pulls the shared electrons closer, giving it a slightly negative charge. The carbon, on the other hand, gets a slightly positive charge. This makes it a prime spot for chemical reactions, like a magnetic attraction for other molecules. It's the secret ingredient that gives aldehydes their unique personality and chemical behavior. Think of it as the little black dress of functional groups – versatile, essential, and always in style.
Now, for acetaldehyde (ethanal), this carbonyl group is attached to a very simple carbon chain. Remember that "eth" from "ethanal"? That means we have two carbons in total. One of those carbons is part of the carbonyl group. The other carbon is simply attached to that carbonyl carbon. This second carbon also has three hydrogen atoms happily bonded to it. It's a straightforward setup, a clean and classic design. No fuss, no unnecessary frills. Just pure, functional elegance.
Sketching It Out: The Visual Appeal
Let's get down to the nitty-gritty of drawing it. We can do this in a few ways, each with its own charm. First, there's the Lewis structure. This is where we show all the atoms and all the valence electrons, represented by dots. You'll have your carbon with its double bond to oxygen and single bonds to the other carbon and a hydrogen. Then, you'll have the other carbon bonded to three hydrogens. It's like sketching out every single stitch in your favorite sweater – detailed, but incredibly accurate.
But let's be real, sometimes a full Lewis structure can look a bit cluttered. That's where the condensed structural formula comes in. This is like styling your hair in a way that shows off your features without getting bogged down in every single strand. For acetaldehyde, you'd write it as CH3CHO. See? The CH3 represents the methyl group (that’s the carbon with its three hydrogens), and the CHO represents the aldehyde group. The carbon in CHO is implicitly understood to be double-bonded to the oxygen and single-bonded to the preceding CH3 group. It’s concise, chic, and gets the point across instantly. Like a perfectly worded caption for your Insta post.
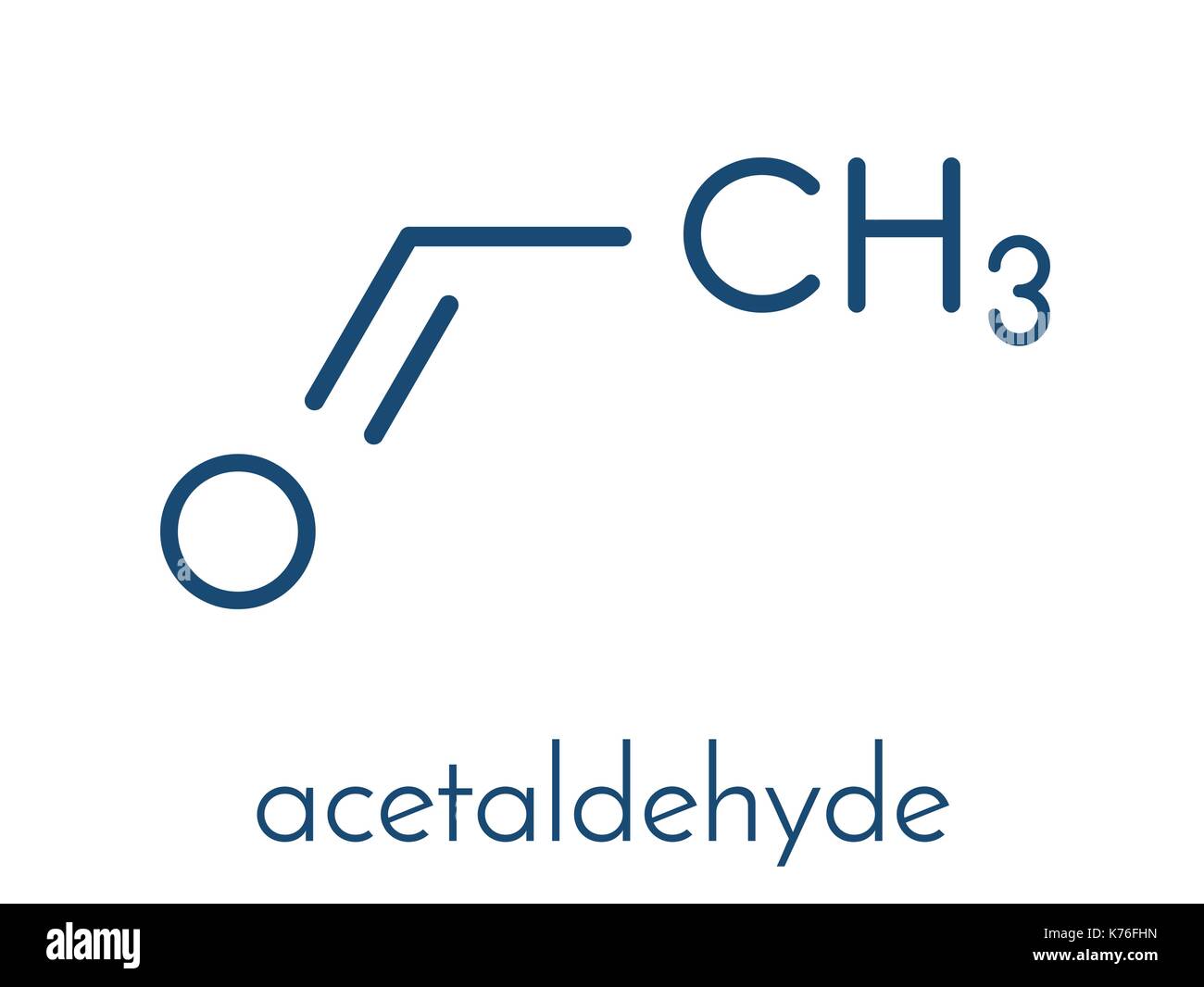
Then there's the skeletal structure, which is super popular in the organic chemistry world. It’s the minimalist approach. Imagine a clean, modern art piece. Lines represent carbon-carbon bonds, and the ends of lines and vertices represent carbon atoms. Hydrogens attached to carbons are usually not shown unless they are attached to a heteroatom (like oxygen or nitrogen). For acetaldehyde, it's incredibly simple. You’d have a line representing the C-C bond, and then a stylized representation of the aldehyde group. The double bond to oxygen is usually shown explicitly. It looks like a little zig-zag with an oxygen hanging off the end. So, you have a two-carbon chain, which is just two points connected by a line, and then the aldehyde group attached to one of those points. Clean, efficient, and oh-so-stylish.
Why Does This Simple Molecule Matter?
You might be thinking, "Okay, it's a pretty molecule, but what's the big deal?" Well, acetaldehyde is actually quite the multitasker. It's a key intermediate in many biological processes, meaning it’s involved in how our bodies work. For instance, it's formed when your body metabolizes alcohol. So, that glass of wine with dinner? Your body processes it, and acetaldehyde is part of that journey. It’s also a component of cigarette smoke, which is why it's something we want to keep away from our bodies.
Beyond biology, acetaldehyde is a workhorse in industry. It's used in the production of things like acetic acid (the main component of vinegar – hello, salad dressing!), which is a massive industry in itself. It's also used to make perfumes, dyes, and plastics. So, that lovely scent in your favorite candle or the vibrant color of your new scarf might have a little bit of acetaldehyde in its family tree. It’s like the unsung hero in the background of many of the things we use and enjoy every day. Pretty cool for something so structurally simple, right?

Fun Facts and Cultural Quirks
Did you know that acetaldehyde is responsible for some of the unpleasant symptoms of a hangover? When you drink alcohol, your body breaks it down into acetaldehyde. If your body can't process it quickly enough, it builds up, leading to headaches, nausea, and that general feeling of "oh no." It's like your body’s way of saying, "Hey, we need to slow down on this stuff!"
In a more positive light, acetaldehyde plays a role in the aroma of some beloved foods and drinks. It contributes to the characteristic fruity notes in ripe apples, bananas, and even some wines and beers. So, the next time you savor the bouquet of a fine Cabernet or enjoy the sweetness of a perfectly ripe pear, you can appreciate the subtle influence of this molecule. It's like the secret ingredient that makes your favorite dishes sing.
Interestingly, in some cultures, fermented foods and beverages containing low levels of acetaldehyde are enjoyed for their unique flavors. It’s a reminder that even chemicals we often associate with negative effects can have a place in our culinary traditions, provided they are present in safe amounts. It’s all about balance and context, isn't it?
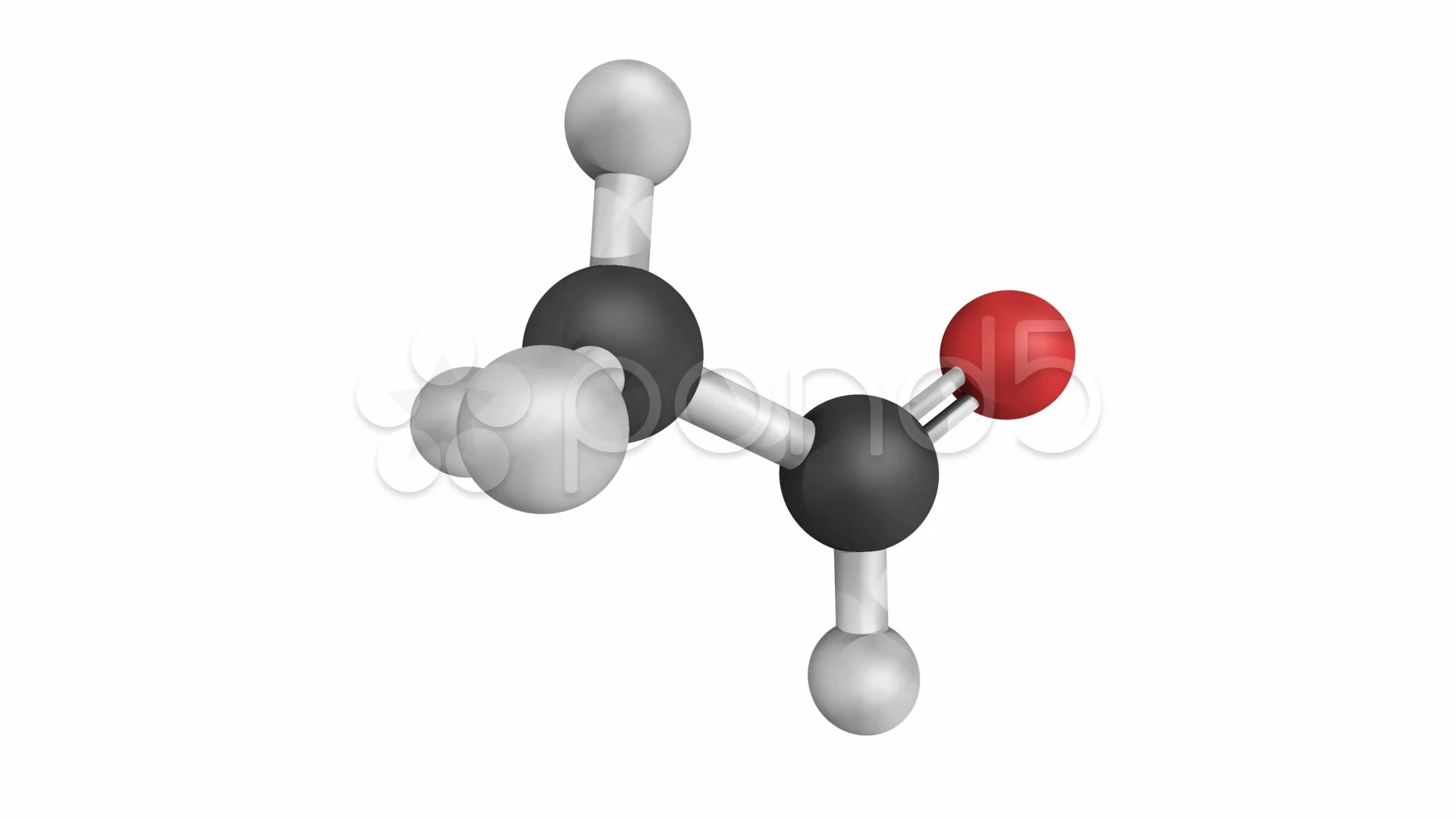
Bringing It All Together: Structure and Function
The beauty of acetaldehyde's structure lies in its simplicity. That CH3 group is a relatively stable, non-reactive part of the molecule. But that CHO group? That's where the magic happens. The polarity of the carbonyl double bond makes it susceptible to attack by nucleophiles (molecules that are electron-rich and looking for a positive spot). This reactivity is what makes acetaldehyde such a valuable building block in organic synthesis. It's like having a perfectly designed tool that can be used for a multitude of tasks. The structure dictates the function, and in acetaldehyde's case, its structure lends itself to a whole lot of useful chemistry.
Think about it in terms of fashion. A well-structured garment is key. You can have the most luxurious fabric, but if the cut is off, it won't look right. Similarly, the specific arrangement of atoms in acetaldehyde, particularly the aldehyde functional group, is what gives it its characteristic properties and enables it to participate in so many important chemical reactions. It’s a masterclass in efficient design.
Practical Tips for Understanding Molecules
So, how can you get better at visualizing these molecules in your daily life? Start by noticing patterns. The "eth" for two carbons, "meth" for one, "prop" for three – these prefixes are your best friends. And remember that "-al" ending for aldehydes, "-one" for ketones, "-ol" for alcohols. It's like learning a new language, and once you get the basic vocabulary, you can start to understand the sentences.
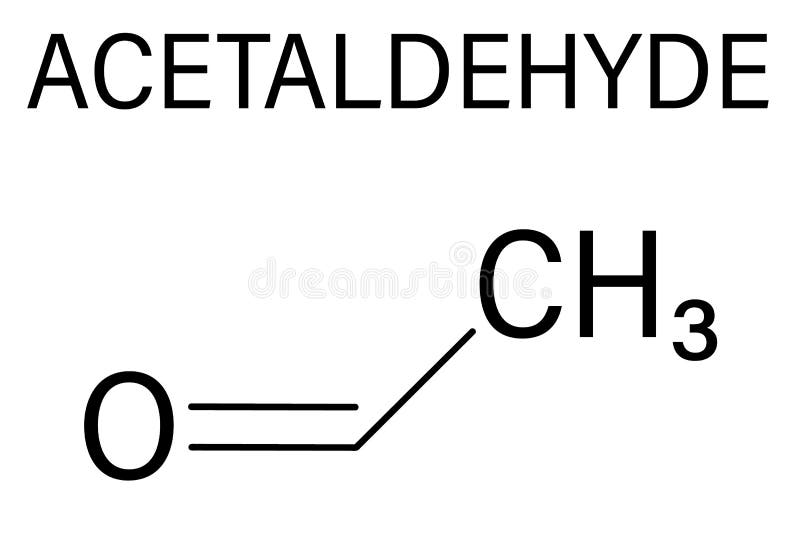
When you see a chemical formula, try to mentally sketch out the structure. Even if you don't draw it perfectly every time, the act of visualizing helps solidify the concepts. Think of it like practicing a dance routine. The more you rehearse, the more fluid and natural it becomes. Don't be afraid to use online resources or apps that show 3D models of molecules. It’s like having a virtual fashion mannequin to see how a design looks from all angles.
And when you encounter a new chemical name, break it down. What are the root parts? What does the suffix tell you? This analytical approach will serve you well not just in chemistry, but in many areas of life where understanding complex information is key. It’s the principle of breaking down a big task into smaller, more manageable steps, a strategy that’s useful for anything from organizing your closet to planning your next big adventure.
A Little Reflection on Simplicity
It’s fascinating how something as seemingly simple as acetaldehyde, with its two carbons and an aldehyde group, can have such a profound impact on our lives, from the biological processes within us to the products we use every day. It’s a reminder that often, the most elegant solutions and the most important components are found in their simplicity. Like a perfectly brewed cup of coffee, the complex flavors arise from a few fundamental elements, expertly combined. Or the timeless appeal of a well-tailored blazer – it’s the clean lines and functional design that make it a staple.
In our often chaotic and over-stimulated world, taking a moment to appreciate these fundamental building blocks, whether they’re molecules or concepts, can be incredibly grounding. Understanding the structure of acetaldehyde, in its neat CH3CHO form, brings a sense of order and clarity. It’s a small, satisfying victory in the grand scheme of things. And that, my friends, is a beautiful thing.
