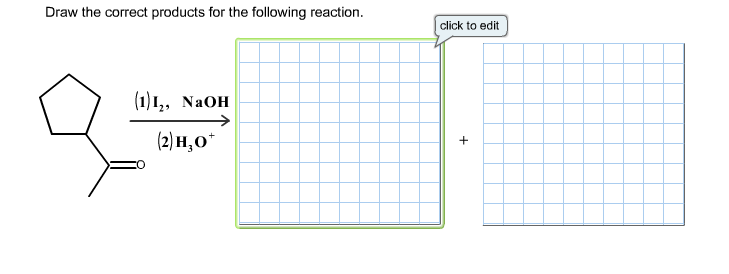
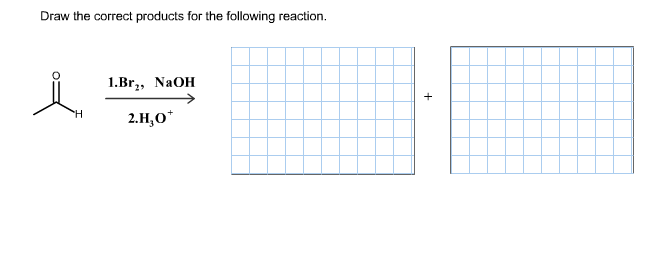
Draw The Correct Products For The Given Reaction.

Ever looked at a chemical reaction and felt a spark of curiosity, like you're about to solve a puzzle? That's the magic of predicting chemical products! It's not just for scientists in lab coats; it's a fantastic mental workout that taps into your inner detective. Think of it as a visual riddle where the reactants are your clues, and the products are the solution you get to uncover. It’s incredibly satisfying to see how different molecules can rearrange and transform into something entirely new, and being able to predict that outcome is like having a secret superpower in the world of chemistry.
This skill, often simply called "drawing the correct products for a given reaction," is a cornerstone of understanding how the chemical world works. It’s less about rote memorization and more about understanding the fundamental rules of engagement between different atoms and molecules. When you learn to predict products, you're essentially learning to read the 'language' of chemistry. You begin to understand why certain reactions happen and why others don't. It’s a gateway to appreciating the intricate dance of electrons and bonds that make up everything around us, from the air we breathe to the food we eat and the medicines that heal us.
The Joy of Prediction: Unlocking Chemical Transformations
The purpose of learning to draw the correct products for a given reaction is beautifully straightforward: to understand and anticipate chemical change. Imagine you're given a set of ingredients for a recipe. You know the ingredients, but what will the final dish taste and look like? Predicting chemical products is the chemistry equivalent of that. You're given the starting materials – the reactants – and by applying a set of well-established principles, you can determine what will be formed – the products.
The benefits of mastering this skill are numerous and far-reaching. Firstly, it fosters a deeper and more intuitive understanding of chemical principles. Instead of just memorizing equations, you start to grasp the 'why' behind them. You learn about concepts like electron movement, bond breaking and formation, and the influence of factors like acids, bases, and catalysts. This understanding is crucial for anyone pursuing further studies in chemistry, biology, medicine, engineering, or environmental science.
Beyond academics, this skill is incredibly practical. In research and development, chemists use this knowledge to design new materials, synthesize life-saving drugs, and develop cleaner industrial processes. For example, understanding how to predict the products of a combustion reaction helps engineers design more efficient engines, while predicting the outcome of biochemical reactions is vital for understanding diseases and developing treatments. Even in everyday life, a basic understanding of chemical reactions can help you make informed decisions, whether it's about how to store food safely or understand the ingredients in cleaning products.
Learning to predict reaction products is like acquiring a new lens through which to view the world. It transforms abstract chemical formulas into tangible processes and potential outcomes.
The process itself can be surprisingly engaging. It often involves looking for specific functional groups within the reactant molecules, identifying the type of reaction occurring (like an addition reaction, a substitution reaction, or an elimination reaction), and then applying the relevant rules to see how those groups interact and rearrange. It’s a bit like playing a strategic board game, where each piece (atom or molecule) has its own rules of movement and interaction.
Turning Clues into Creations
So, how does one actually go about drawing the correct products? It starts with recognizing the key players. You’ll often be presented with one or more reactants – the starting molecules. Then, you'll see an arrow, indicating the direction of the transformation, and on the other side, you'll be expected to draw the products. Sometimes, there might be extra information above or below the arrow, like the name of a reagent (a substance that causes a chemical reaction) or a condition like heat (Δ) or light (hν).
The core principle is that atoms are conserved in chemical reactions; they are simply rearranged. So, the total number and type of atoms you start with must be the same as the total number and type of atoms you end up with. Your task is to figure out how they will bond together in the new arrangements.
Let’s take a very simple example. Imagine you have methane (CH₄) and oxygen (O₂). If you know this is a combustion reaction (often indicated by the presence of oxygen and sometimes heat), you can predict the products. Methane consists of one carbon atom bonded to four hydrogen atoms. Oxygen exists as diatomic molecules (O₂). In combustion, the carbon atom typically ends up bonded to oxygen to form carbon dioxide (CO₂), and the hydrogen atoms combine with oxygen to form water (H₂O). So, the reactants are CH₄ and O₂, and the products are CO₂ and H₂O. The challenge, and the fun, lies in figuring out these specific rearrangements and balancing the equation to ensure atom conservation.
Another common scenario involves acid-base reactions. If you have an acid (which tends to donate a proton, H⁺) and a base (which tends to accept a proton), you can predict a transfer of that proton. For instance, hydrochloric acid (HCl) reacting with sodium hydroxide (NaOH) will result in the formation of water (H₂O) and sodium chloride (NaCl), common table salt. Here, the H⁺ from HCl is accepted by the OH⁻ from NaOH to form water, leaving Na⁺ and Cl⁻ to form the salt.
The beauty of this process is that it builds upon itself. As you encounter more types of reactions – from simple acid-base neutralizations to complex organic synthesis steps – you start to recognize patterns. You learn about the reactivity of different functional groups, such as alkenes (compounds with carbon-carbon double bonds), alcohols (compounds with -OH groups), and carbonyls (compounds with C=O groups). Each functional group has its own set of preferred reactions, like a distinct personality guiding its interactions.
When you're faced with a new reaction, you can break it down. First, identify the functional groups present in the reactants. Then, consider the reagents and conditions. Do they suggest a particular type of reaction? Is there a strong acid or base present? Is there a double or triple bond that could be attacked? By asking these questions, you can start to piece together the likely outcome. It’s a skill that requires practice, but the reward is immense. You’re not just memorizing; you’re understanding and predicting, actively participating in the unfolding of chemical events.
