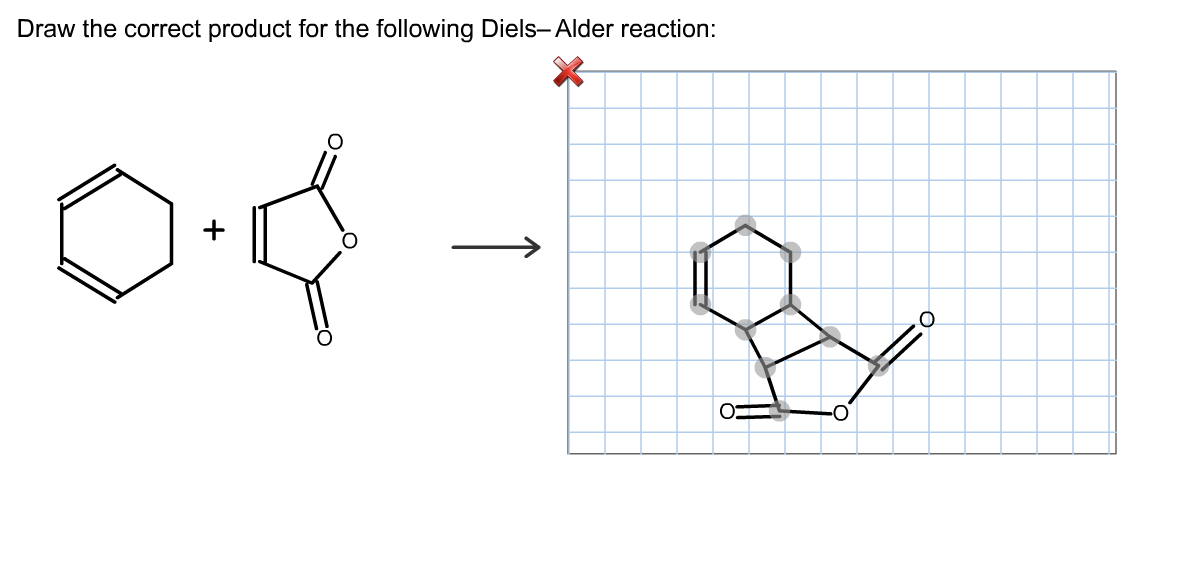
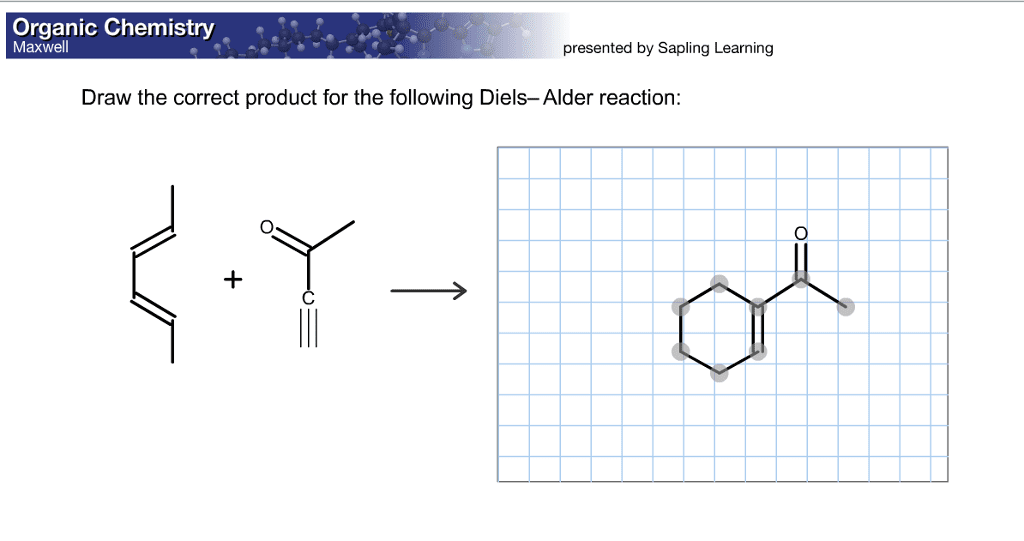
Draw The Correct Product For The Given Diels Alder Reaction

Ever feel like your kitchen is a chaotic wonderland of ingredients, just waiting to be mashed, mixed, and transformed into something delicious? Well, buckle up, because organic chemistry has its own version of a culinary adventure, and it's called the Diels-Alder reaction. Think of it as a fancy molecular recipe, where two specific ingredients – a diene and a dienophile – get together and, with a little heat (or sometimes just good vibes), whip up something new and exciting. It’s like making a cake, but instead of flour and sugar, we’re dealing with buzzing electrons and molecular bonds. And just like you can't just throw any old stuff into a cake batter and expect a masterpiece, you need the right diene and the right dienophile to get the correct product in a Diels-Alder reaction.
Seriously, it’s all about matching. You wouldn’t put anchovies in your chocolate chip cookies, would you? (Unless you’re going for a… unique experience). The Diels-Alder reaction is super picky about its partners. The diene, which is basically a molecule with two double bonds that are just chilling next to each other (like a molecular conga line!), needs to be ready to party. And the dienophile? That’s the molecule that’s eager to join the dance, usually sporting a double or triple bond. When they meet, it’s a beautiful thing. They lock arms (or rather, electrons) and form a brand new ring structure. It’s like a molecular handshake that solidifies into a friendship bracelet.
So, the big question is: how do we figure out what that new ring structure is going to be? This is where our inner detective comes out. We're looking at the starting ingredients and predicting the final dish. It's like looking at a recipe card that says "Bake at 350°F for 30 minutes" and knowing exactly what’s going to come out of the oven. No surprises, just good old-fashioned prediction. And the coolest part? It’s all predictable, based on a few simple rules. It’s not some mystical alchemy; it’s just following the molecular breadcrumbs.
The Two Stars of the Show: Diene and Dienophile
Let’s break down our main players. The diene is our enthusiastic host. It’s got this cool, flexible structure with two double bonds separated by a single bond. Think of it like a molecule that’s always ready to swing dance. It needs to be able to twist and bend so that those two double bonds can get close enough to interact with its dance partner. If the diene is too rigid, like a statue, it just won't be able to get its groove on.
Now, the dienophile. This is the molecule that’s a bit more… electrophilic. That’s a fancy word for saying it’s really attracted to electron-rich areas. Think of it as a magnet, or maybe someone who’s always drawn to the life of the party. It’s got a double or triple bond that’s just begging to be part of a ring. The more electron-withdrawing groups it has attached (like fancy chemical decorations), the more appealing it is to our diene. It’s like adding extra sprinkles to an already delicious cupcake – makes it even more irresistible!
The most common dienes you’ll see are things like 1,3-butadiene, which is basically just a four-carbon chain with double bonds at positions 1 and 3. Simple, right? And for dienophiles, you’ve got things like acrylates (think of these as fancy vinyl groups with a little something extra attached) or even alkynes (which are like even more super-charged double bonds). The key is that these double bonds need to be in the right places and have the right electronic personalities to click.
Matching Made Easy: The Electron Shuffle
So, how do these two actually decide to form a bond? It’s all about electron flow. Imagine the electrons in the double bonds are like little messengers. In the diene, these messengers are a bit spread out, but they’re ready to move. The dienophile has its own messengers, and they're often a bit more concentrated or eager to jump into action. When they get close, the electrons from the diene’s double bonds essentially migrate over to the dienophile, and in return, new bonds are formed, creating a six-membered ring. It’s a collaborative effort, a beautiful exchange.
Think of it like a game of musical chairs, but instead of chairs, we have electron pairs, and instead of music, we have the inherent reactivity of the molecules. The diene has electrons that are a bit more "free-range," ready to be shared. The dienophile is often "electron-deficient" in its double bond region, meaning it's looking for those electron-rich partners. When they get together under the right conditions, bam! A new ring is formed. It’s like they found their perfect match in the molecular dating pool.
The trickiest part, and where we sometimes scratch our heads, is figuring out the stereochemistry – basically, how the atoms are arranged in 3D space. It’s not just what bonds are formed, but how they are oriented. This is where we bring in concepts like the endo and exo rule. Imagine you’re building a little LEGO house. The diene is the base, and the dienophile is like the roof you’re adding. The endo product is when the dienophile tucks itself underneath the diene, almost like it’s hiding. The exo product is when it sticks out more, like it’s waving hello.
Predicting the Winner: End o’ the Line
The endo rule is our secret weapon for predicting which orientation is preferred. Generally, when you have a dienophile with electron-withdrawing groups (those fancy decorations we talked about), these groups will end up in the "endo" position. Why? It’s all about secondary orbital interactions. Think of it as a little extra electrostatic attraction, a subtle pull that guides the dienophile into that tucked-under position. It’s like the dienophile saying, “Hey, I can get a little closer to this part of the diene over here, which makes me feel extra stable!”
So, when you’re given a diene and a dienophile and asked to draw the correct product, your first step is to identify them. Is it a molecule with two conjugated double bonds? That’s your diene. Is it a molecule with a double or triple bond that’s electron-poor? That’s your dienophile. Once you’ve got them, visualize them getting ready to dance.
Now, draw the diene in its s-cis conformation. This is crucial! The diene needs to be able to bend so that the ends of its double bonds can connect to the dienophile simultaneously. If it's stuck in an "s-trans" shape (all stretched out), the reaction won't happen. Imagine trying to hug someone while you’re both on opposite sides of a room – not very effective! So, the diene needs to curl up a bit.
Then, bring in the dienophile. If the dienophile has any electron-withdrawing groups (like carbonyls, nitriles, or esters – think of these as the "flavor enhancers" of the molecular world), these groups will tend to point downward towards the diene in the final product. This is the endo preference in action. It’s like the flavor enhancers are trying to get as close as possible to the core of the molecule for maximum impact.
Let’s say you’re given butadiene (that simple four-carbon diene) and methyl acrylate (a common dienophile with a carbonyl group). Butadiene is our flexible dancer. Methyl acrylate has that carbonyl group, which is an electron-withdrawing group. When they get together, you’ll draw a six-membered ring. The butadiene will form the base of the ring. The methyl acrylate will add two carbons to the ring. And that carbonyl group on the methyl acrylate? It will end up pointing underneath the ring, tucked towards the other double bond from the original butadiene. That’s your endo product!
Beyond the Basics: When Things Get Spicy
Sometimes, the reaction conditions can also influence the outcome. High temperatures can sometimes favor the exo product because it's often kinetically more stable (meaning it forms faster) at higher energies. It's like when you're in a hurry and you just grab the first thing you can; sometimes that leads to a slightly less "perfect" arrangement, but it gets the job done quicker.
Also, the substituents on the diene itself can play a role. If the diene has bulky groups attached, it might force the dienophile into a particular orientation. Imagine trying to put on a bulky sweater – it might dictate how you move your arms. So, always look at all the pieces of the puzzle, not just the main actors.
The regiochemistry (where the substituents end up on the new ring) is another important aspect, especially when the diene or dienophile are unsymmetrical. Think of it like this: if you're adding a topping to a pizza that already has toppings on one side, where does the new topping naturally fall? There are rules for this too, often related to matching electron-rich areas with electron-poor areas to form the most stable intermediate. But for predicting the correct product in terms of its basic structure and stereochemistry, focusing on the diene, dienophile, and the endo rule is usually your strongest bet.
It’s a bit like being a master chef. You know your ingredients, you know your techniques, and you can predict the delicious outcome. The Diels-Alder reaction, while sounding super scientific, is fundamentally about these molecules finding their perfect partner and coming together to create something new and, often, quite useful. Whether it's for making plastics, medicines, or all sorts of other cool stuff, this reaction is a fundamental building block in the world of chemistry. So next time you see a Diels-Alder problem, don't panic! Just channel your inner molecular matchmaker, identify your diene and dienophile, and predict that beautiful ring formation. And remember, usually, that dienophile with the extra goodies likes to snuggle up underneath. Happy predicting!
