Draw The Correct Organic Product Of The Oxidation Reaction Shown:

Hey there, fellow curious minds! Ever looked at something and wondered what happens when it gets a little… poked? Or zapped? Today, we’re diving into the wonderfully weird world of organic chemistry, specifically, what happens when we give certain molecules a little push in the form of an oxidation reaction. Don’t worry, it’s not as scary as it sounds. Think of it like giving your ingredients a bit of a transformation in the kitchen, but with atoms and electrons!
So, we’re going to look at a specific reaction, a bit like a puzzle, and our mission, should we choose to accept it, is to figure out the correct organic product. Sounds like a detective story, right? We’re the chemists on the case, and our goal is to predict what our molecule will become after its little makeover.
What’s the Big Idea with Oxidation?
Before we get our hands dirty (metaphorically, of course!), let’s chat about what oxidation even means in the context of organic molecules. You might think of rusting iron, but it’s a bit different for carbon-based life. In organic chemistry, we often think about oxidation as a molecule losing electrons. Or, you can think of it as gaining oxygen atoms, or losing hydrogen atoms. It’s like a molecule is shedding some of its baggage.
Imagine a molecule is like a person. If they’re getting oxidized, it’s like they’re giving away some of their energy (electrons) or shedding some less desirable traits (hydrogen atoms). It’s a way for them to become something new, often more stable or ready for a different kind of interaction.
There are different degrees of oxidation, too. Think of it like going from being a little tired to really wiped out. A molecule can go through a few stages of this "getting poked" process.
Our Mystery Molecule: What Are We Working With?
The image you’re seeing (if you had one, which you don’t, but imagine one with me!) shows us a starting molecule. Let’s call it our reactant. It’s got a certain structure, a specific arrangement of carbon, hydrogen, and maybe some other atoms. For this particular puzzle, we’re likely looking at an alcohol. Now, alcohols are a pretty common bunch of molecules. Think of things like ethanol (in your celebratory drinks!) or methanol (used as fuel, but please don’t drink that!).
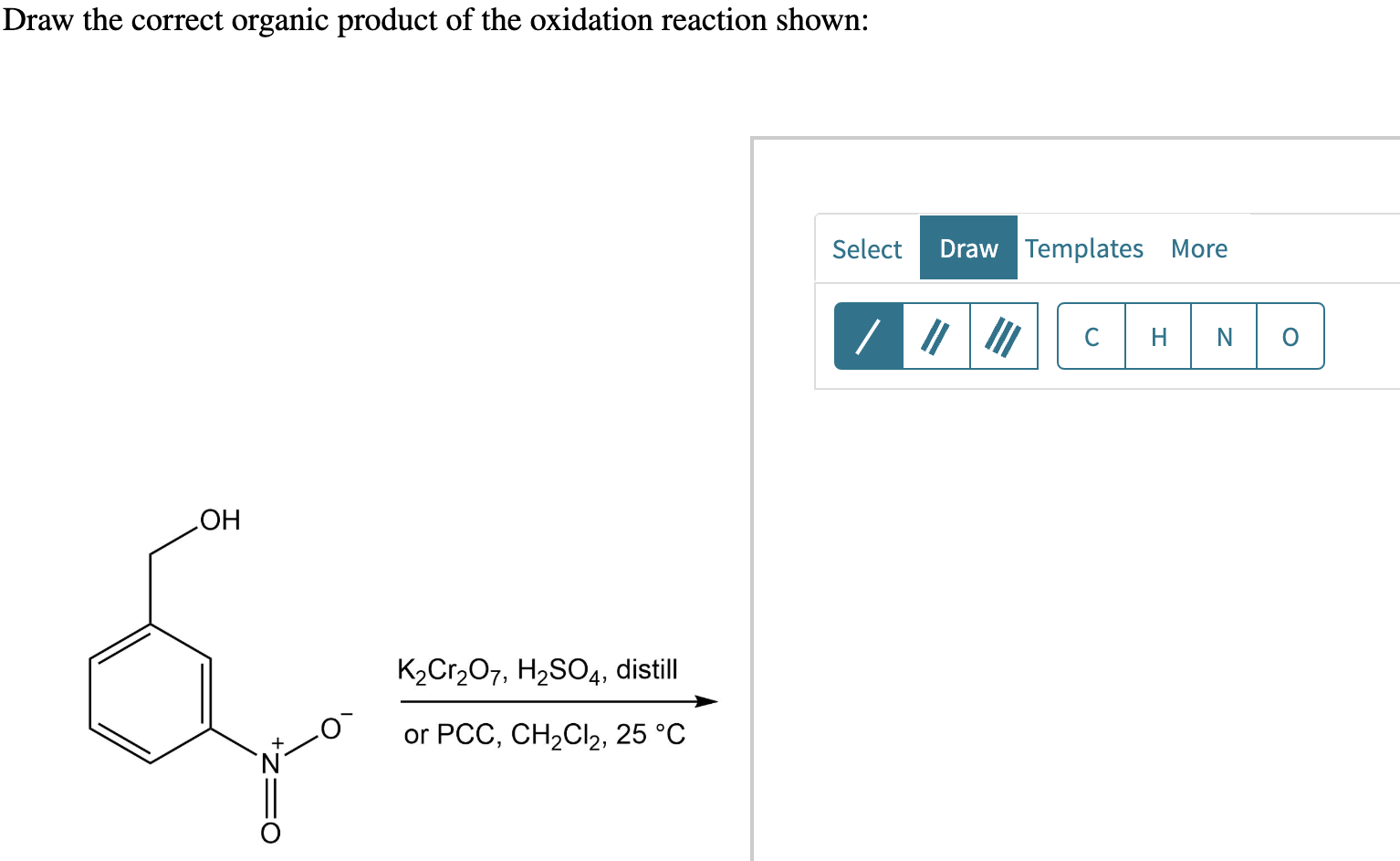
Alcohols have a very distinctive feature: a hydroxyl group, which is a fancy way of saying an oxygen atom bonded to a hydrogen atom (-OH). This -OH group is where all the action is going to happen. It’s like the target for our oxidation reaction.
The Oxidizing Agent: Who’s Doing the Poking?
Now, you can’t just will a molecule to oxidize. You need a little help. That help comes in the form of an oxidizing agent. These are like the tools or the catalysts in our chemical kitchen. They’re the things that cause the oxidation to happen. Think of them as the enthusiastic personal trainer for our molecules!
There are tons of oxidizing agents out there, each with a different level of intensity. Some are super gentle, like a light nudge, while others are like a full-on wrestling match. For our specific reaction, the choice of oxidizing agent is super important. It’s like choosing between a gentle facial peel and a more intensive chemical peel – the results will be very different!
Often, you’ll see common ones like potassium permanganate (KMnO₄), chromium trioxide (CrO₃), or even milder ones like pyridinium chlorochromate (PCC). The specific agent tells us how far our alcohol can be pushed.
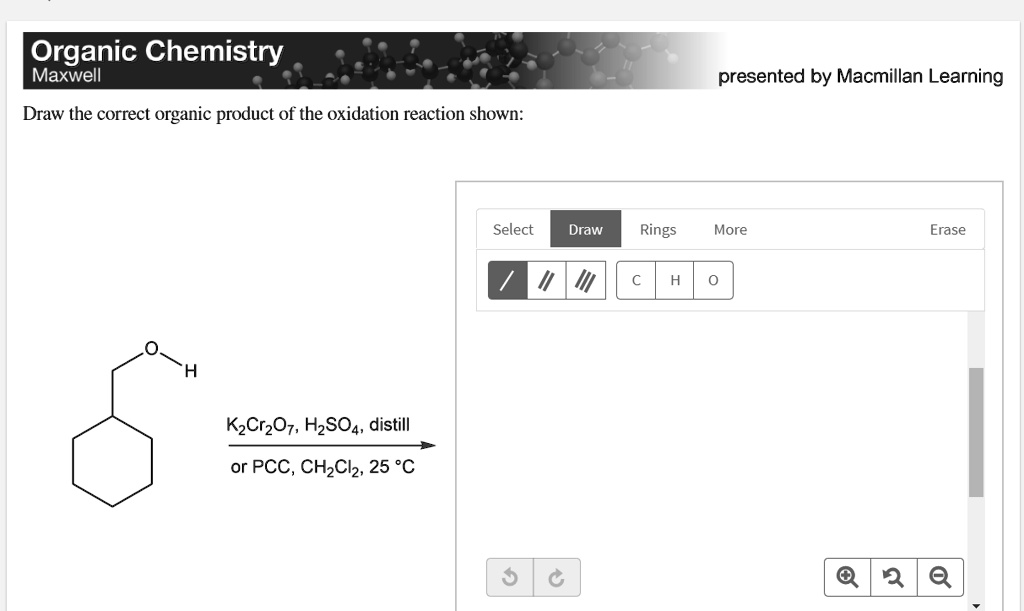
The Big Transformation: What Happens to the Alcohol?
So, we have our alcohol reactant and our oxidizing agent ready to go. What’s the most likely outcome? This is where the structure of the alcohol really matters. Alcohols can be classified into three types: primary, secondary, and tertiary.
Imagine the carbon atom that’s attached to the -OH group. If that carbon is attached to only one other carbon atom, it’s a primary alcohol. If it’s attached to two other carbon atoms, it’s a secondary alcohol. And if it’s attached to three other carbon atoms, it’s a tertiary alcohol. This little detail is key to predicting the product!
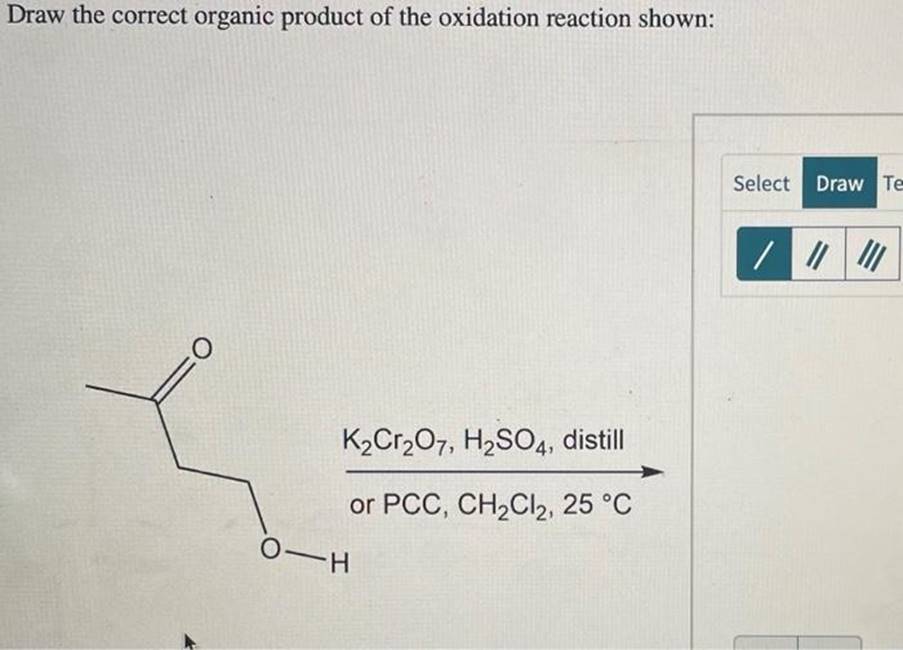
Primary Alcohols: The First Step Up
Primary alcohols are the most eager to get oxidized. When they encounter a decent oxidizing agent, they usually get transformed into aldehydes. Think of aldehydes as having a carbon atom double-bonded to an oxygen atom and also bonded to a hydrogen atom (R-CHO). It’s like the alcohol took off one hydrogen from the -OH group and one hydrogen from the carbon it’s attached to, and that carbon is now forming a double bond with oxygen.
If we use a really strong oxidizing agent and plenty of it, or if the aldehyde is left to hang out for too long, it can get oxidized even further. The aldehyde then becomes a carboxylic acid. Carboxylic acids are a bit more complex, with a carbon atom double-bonded to an oxygen and also bonded to a hydroxyl group (-COOH). So, a primary alcohol can become an aldehyde, and then potentially a carboxylic acid. It’s a two-stage process, like leveling up in a game!
Secondary Alcohols: A Stable Intermediate
Secondary alcohols are a bit more laid-back. When they get oxidized, they typically become ketones. Ketones have a carbon atom double-bonded to an oxygen atom, and this carbon is also bonded to two other carbon atoms (R-CO-R'). Notice that a ketone doesn't have any hydrogen atoms directly attached to that carbonyl carbon (the C=O part). This makes ketones a bit more stable than aldehydes in terms of further oxidation.
Once a secondary alcohol becomes a ketone, the reaction usually stops there. It’s like reaching a comfortable plateau. Ketones are generally resistant to further oxidation under normal conditions. So, if you start with a secondary alcohol, your product is most likely a ketone.
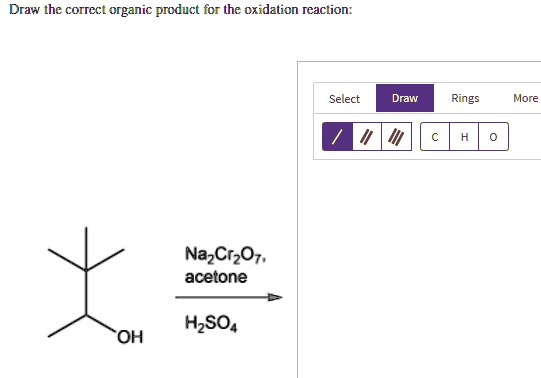
Tertiary Alcohols: The Unreactive Ones
Tertiary alcohols are the rebels of the group. They’re pretty resistant to oxidation. Why? Because the carbon atom attached to the -OH group is already bonded to three other carbon atoms. This means it doesn’t have any spare hydrogen atoms to easily lose, which is often a requirement for oxidation. Think of them as already being quite "full up" and not wanting to change much.
To oxidize a tertiary alcohol, you’d need some seriously harsh conditions that would likely break down the whole molecule, not just a simple oxidation. So, for most typical oxidation reactions we encounter, tertiary alcohols often remain unchanged. They just sit there, cool as ever.
Putting It All Together: The Puzzle Solved!
So, when you’re presented with a reaction like this, your job is to:
- Identify the starting alcohol: Is it primary, secondary, or tertiary? Look at the carbon attached to the -OH group and count how many other carbons it's connected to.
- Consider the oxidizing agent: Is it a strong one that can push things further, or a milder one that stops at an intermediate step? For this specific "draw the correct product" question, the agent usually implies the intended outcome.
Let’s say, for example, the image showed a secondary alcohol. Then, knowing that secondary alcohols turn into ketones upon oxidation, you would draw a ketone as your product. You’d be looking for that C=O group where the original -OH group was, with the same carbon skeleton around it.
It’s like being a master chef who knows exactly how to transform a simple ingredient into a gourmet dish. You know that this type of vegetable, with this cooking method, will result in this delicious outcome. In chemistry, we’re using our knowledge of how atoms behave to predict the delicious (or at least, scientifically accurate) outcome!
It’s this predictive power that makes organic chemistry so fascinating. We can look at a starting point, understand the rules of engagement, and accurately guess what the final result will be. It’s a beautiful dance of electrons and atoms, and figuring out the steps is half the fun!
