Draw The Correct Organic Product For The Oxidation Reaction:

Ever looked at a molecule and wondered what happens when you give it a little energetic nudge? In the world of chemistry, a common and incredibly useful trick is to perform an oxidation reaction. Think of it like giving a molecule a mild electric shock – it changes things, often in predictable and exciting ways! This isn't just for lab coat-wearing scientists; understanding oxidation is key to grasping how many everyday processes work, from how our bodies digest food to how fuels burn. It’s a fundamental transformation that underpins a huge amount of chemistry, and today, we're going to dive into a specific kind of fun: predicting the correct organic product after an oxidation.
Why is this so fun and useful? Well, imagine you have a set of building blocks (organic molecules) and you want to create something new. Oxidation reactions are like special tools that let you modify those building blocks in a controlled way. The benefit is enormous: chemists can design and synthesize new materials, medicines, and countless other compounds by understanding these transformations. For students, mastering this skill is like unlocking a superpower in organic chemistry class. It allows you to see a reaction, understand its potential, and predict the outcome. It’s a problem-solving puzzle that’s both intellectually stimulating and incredibly practical.
The real magic lies in figuring out what the molecule becomes after the oxidation. It's a bit like a molecular makeover!
Let's get down to the exciting part: drawing the correct organic product. We'll focus on a common scenario where we're dealing with alcohols. Alcohols are organic molecules that contain a hydroxyl group (-OH) attached to a carbon atom. These are super versatile and are found everywhere, from the ethanol in your drinks to the glycerol in lotions.
The type of alcohol you start with dramatically influences the product you get after oxidation. We usually categorize alcohols into three types: primary, secondary, and tertiary. Each category behaves differently when exposed to oxidizing agents.

Primary alcohols are those where the carbon atom bonded to the -OH group is attached to only one other carbon atom. Think of them as being at the end of a chain. When you oxidize a primary alcohol, you can get a few different things depending on how vigorous the oxidation is. A mild oxidation will turn the alcohol into an aldehyde. Aldehydes are characterized by a carbonyl group (C=O) where the carbon atom is also bonded to at least one hydrogen atom. If you keep oxidizing, especially with stronger oxidizing agents or under harsher conditions, that aldehyde can be further oxidized to a carboxylic acid. Carboxylic acids have a -COOH group, meaning the carbonyl carbon is also bonded to a hydroxyl group.
Secondary alcohols are where the carbon atom attached to the -OH group is bonded to two other carbon atoms. These molecules are typically found in the middle of carbon chains. When a secondary alcohol is oxidized, it smoothly transitions into a ketone. Ketones have a carbonyl group (C=O) where the carbon atom is bonded to two other carbon atoms. Unlike aldehydes, ketones generally cannot be further oxidized under typical conditions without breaking carbon-carbon bonds, which is a much more difficult reaction. So, for secondary alcohols, the oxidation usually stops at the ketone stage.
Now, for the tricky ones: tertiary alcohols. These are alcohols where the carbon atom bonded to the -OH group is attached to three other carbon atoms. These are quite stable. When you try to oxidize a tertiary alcohol, you'll find it's much more resistant to the reaction. The carbon atom bearing the hydroxyl group doesn't have a hydrogen atom directly attached to it that can be easily removed, which is a key step in the oxidation of primary and secondary alcohols. Therefore, under normal oxidizing conditions, tertiary alcohols generally do not react. They just sit there, unbothered!
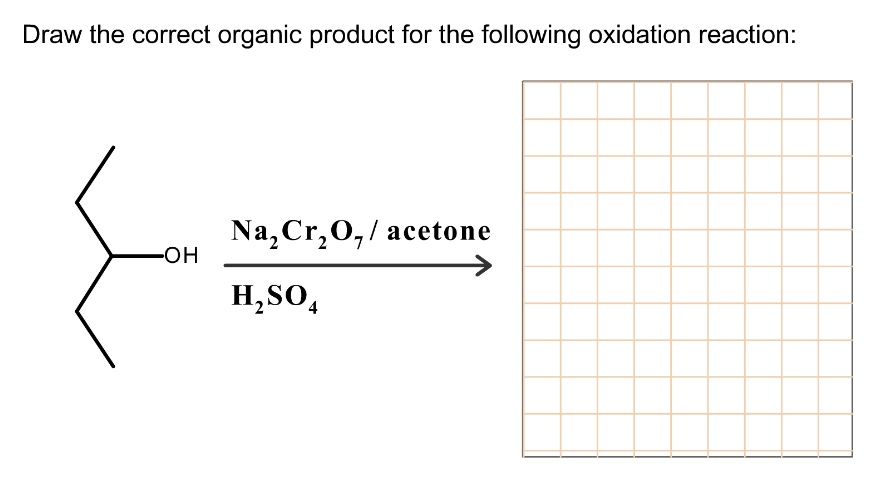
So, how do we determine the correct product? It all comes down to identifying the type of alcohol and the strength of the oxidizing agent. For many introductory problems, you might be given a specific oxidizing agent, like potassium permanganate (KMnO4) or chromic acid (H2CrO4), which are known to be strong oxidizers. If you see one of these and you have a primary alcohol, you're likely aiming for the carboxylic acid. If it's a secondary alcohol, it's the ketone. If it's a tertiary alcohol, well, congratulations, you draw the same molecule back!
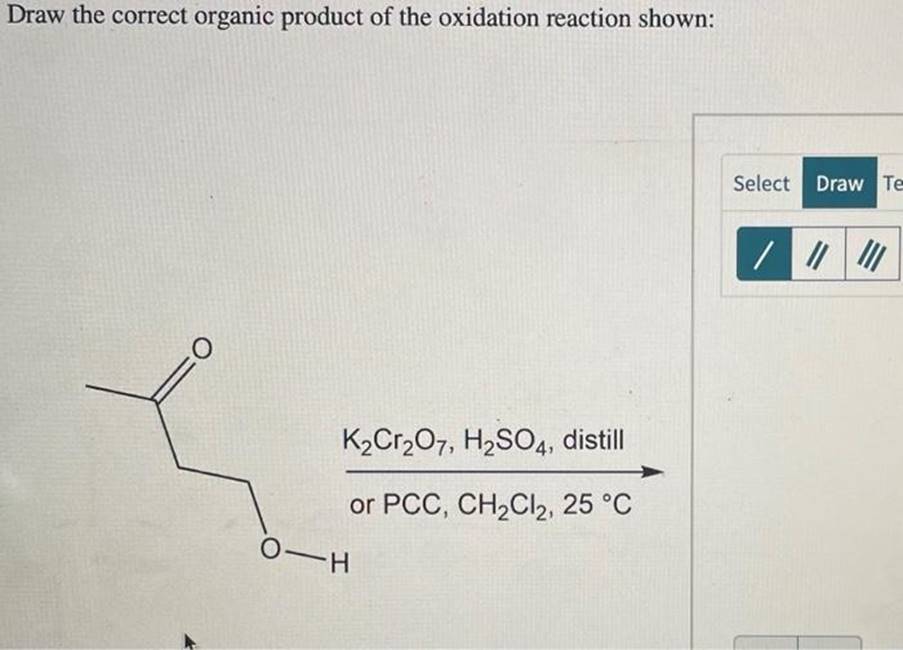
Sometimes, you might see milder oxidizing agents like pyridinium chlorochromate (PCC). PCC is special because it's a gentler oxidizer. When used with a primary alcohol, PCC will stop the reaction at the aldehyde stage. This is a fantastic tool for chemists who specifically want to make aldehydes, as it prevents them from over-oxidizing to carboxylic acids. For secondary alcohols, PCC still produces ketones, just like stronger oxidizers.
To practice, try this: If you see 1-propanol (a primary alcohol) reacting with chromic acid, what do you get? You start with a primary alcohol, and a strong oxidizer like chromic acid is present. This means the reaction will go all the way to the carboxylic acid. The product would be propanoic acid. If you saw 2-propanol (a secondary alcohol) reacting with the same chromic acid, you would get a ketone: acetone (also known as propanone).
The beauty of organic chemistry is its logic. Once you understand the rules for these fundamental reactions like oxidation, you can apply them to a vast array of molecules. It’s like learning the alphabet; once you know your letters, you can start forming words and sentences. So, the next time you encounter an oxidation reaction, don't just see a jumble of atoms. See the potential for transformation, identify the alcohol's type, consider the oxidizing agent, and with a bit of practice, you’ll be drawing the correct organic product with confidence and, dare we say, a little bit of fun!
