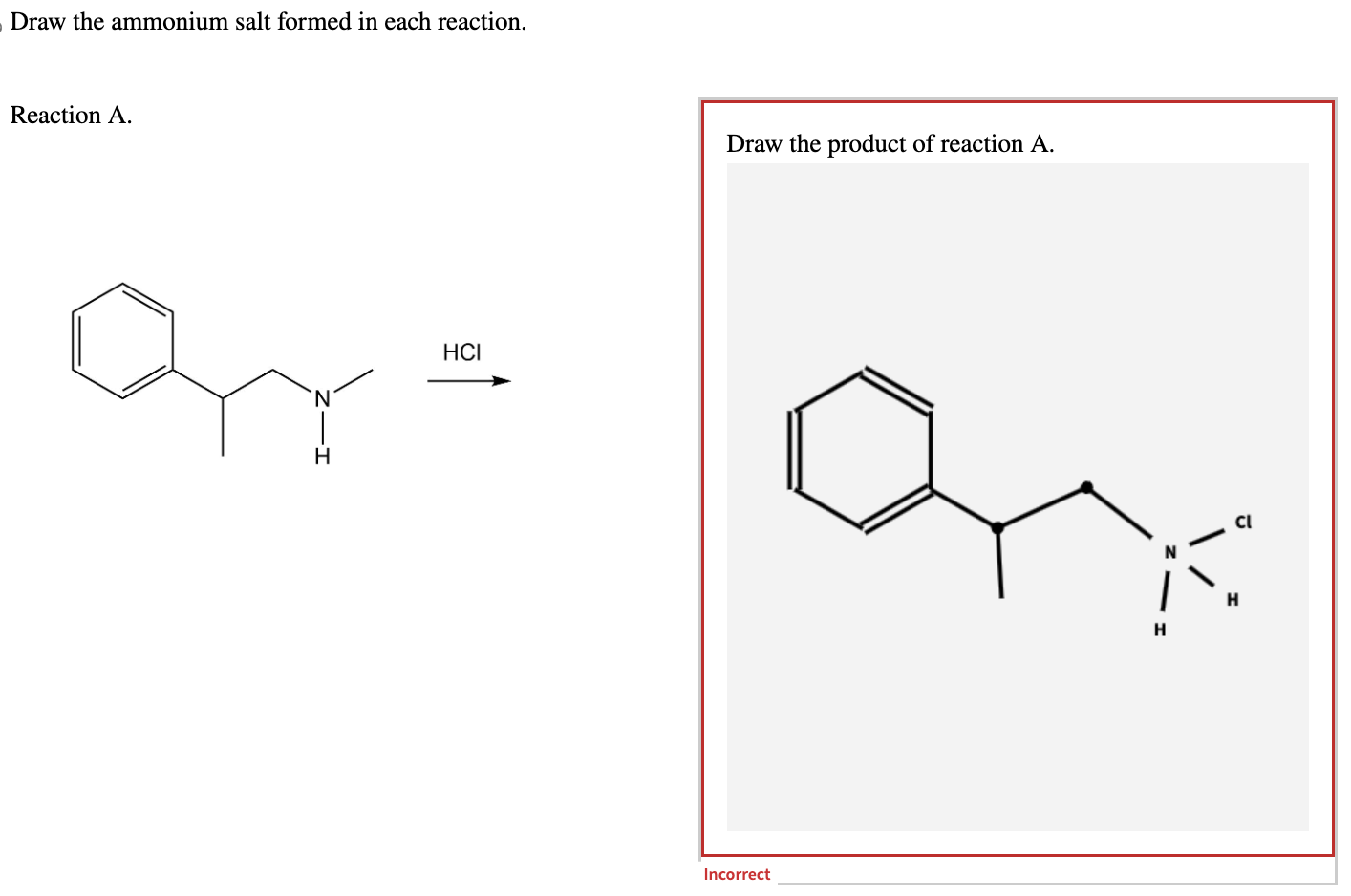
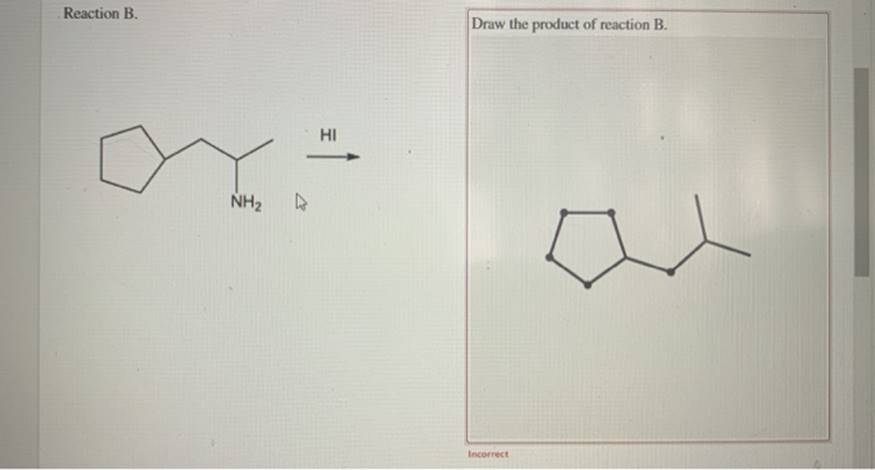
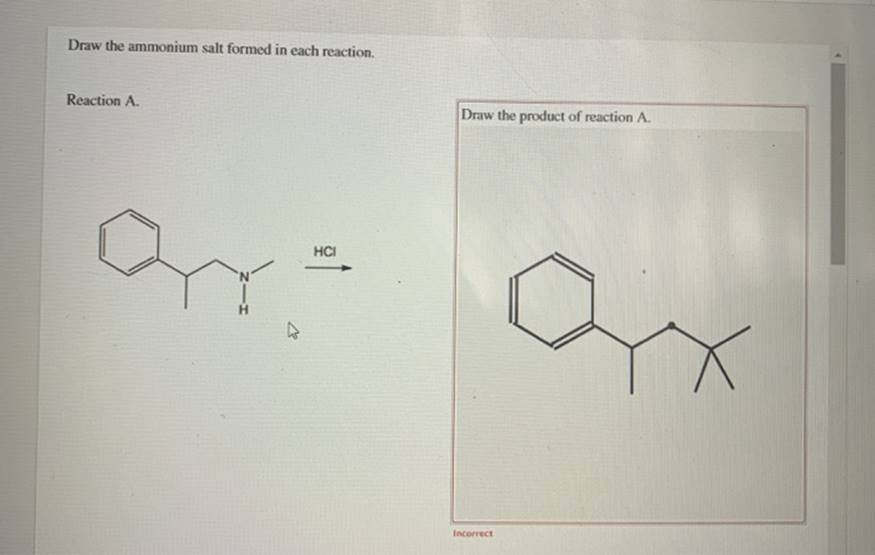
Draw The Ammonium Salt Formed In Each Reaction
Ever feel like you're staring at a chemical equation and thinking, "Where's the fun in that?" Well, get ready to have your mind changed! Today, we're diving into a corner of chemistry that's surprisingly engaging and incredibly useful: drawing the ammonium salt formed in each reaction. It might sound a bit niche, but trust us, understanding this is like unlocking a secret code for how many everyday substances behave. Think about it – from the cleaning power of ammonia-based products to the fertilizers that help our food grow, ammonium salts are everywhere! So, let's ditch the dry textbook approach and explore the cool visual aspect of chemistry.
Why is This So Cool?
The beauty of drawing these salts lies in their simplicity and the way they visually represent a fundamental chemical interaction. It's not about memorizing complex structures; it's about understanding how a positively charged ammonium ion (NH₄⁺) partners up with a negatively charged anion to create a stable, neutral compound. Think of it like a chemical handshake! When you can visualize this pairing, you start to grasp the essence of ionic bonding. Plus, there's a certain satisfaction in sketching out these molecules, seeing the atoms arrange themselves in a neat and predictable way. It’s like solving a mini-puzzle every time!
"Understanding ammonium salts is like gaining a superpower for deciphering common chemical interactions."
This skill isn't just for aspiring chemists. For anyone interested in science, it provides a tangible way to understand abstract chemical concepts. It’s a stepping stone to understanding more complex reactions and the properties of various compounds. You'll be able to look at a reaction and predict, with a bit of practice, what the resulting salt will look like. This predictive power is incredibly empowering and makes chemistry feel less like a foreign language and more like a solvable riddle.
The Purpose and Benefits
So, what's the real point of learning to draw these ammonium salts? The primary purpose is to solidify your understanding of ionic compounds. When an acid reacts with ammonia (or an amine, which is similar), the ammonia molecule gains a proton (H⁺) to become the positively charged ammonium ion. This ion then electrostatically attracts a negatively charged ion (the anion) from the acid. Drawing this process helps you visualize:
- The formation of the ammonium ion: Notice that characteristic tetrahedral shape, with a central nitrogen atom bonded to four hydrogen atoms, carrying a +1 charge.
- The identity of the anion: This can be anything from a simple halide like chloride (Cl⁻) or bromide (Br⁻), to a more complex oxyanion like sulfate (SO₄²⁻) or nitrate (NO₃⁻).
- The overall neutral salt: The final drawing shows the ammonium cation and the anion in their correct stoichiometric ratio, resulting in a neutral compound.
The benefits are numerous. Firstly, it significantly aids in memorization. Visualizing a concept is often far more effective than simply reading it. When you can draw ammonium chloride (NH₄Cl) by pairing the NH₄⁺ and Cl⁻ ions, it sticks in your memory much better. Secondly, it enhances your ability to predict reaction products. Once you recognize that an acid is reacting with ammonia, you can immediately anticipate the formation of an ammonium salt. Your drawing becomes your proof and your understanding.
Furthermore, this skill is directly applicable in many fields. In agriculture, understanding ammonium nitrate (NH₄NO₃) as a fertilizer is crucial. In household products, recognizing the presence of ammonium chloride in certain cleaners helps explain their effectiveness. Even in organic chemistry, the formation of ammonium salts is a key step in many reactions involving amines. Being able to visualize these compounds means you can better understand their properties, such as solubility, melting point, and reactivity.
Let’s consider a simple example. When hydrochloric acid (HCl) reacts with ammonia (NH₃), the HCl donates a proton to the NH₃. This results in the formation of the ammonium ion (NH₄⁺) and the chloride ion (Cl⁻). To draw the ammonium salt formed, you would draw the NH₄⁺ ion, which looks like a nitrogen atom at the center with four hydrogen atoms bonded to it, and a '+' sign indicating its positive charge. Then, you would draw the chloride ion (Cl⁻), which is simply a chlorine atom with a '-' sign. These two ions are then brought together to form ammonium chloride (NH₄Cl). The drawing shows the NH₄⁺ and Cl⁻ ions side-by-side, demonstrating their ionic attraction.
Another example: what happens when sulfuric acid (H₂SO₄) reacts with ammonia? Sulfuric acid is a diprotic acid, meaning it can donate two protons. It reacts with ammonia to form ammonium sulfate ((NH₄)₂SO₄). Here, two ammonium ions are needed to balance the charge of one sulfate ion (SO₄²⁻). So, your drawing would show two NH₄⁺ ions and one SO₄²⁻ ion. The sulfate ion itself has a specific structure with a central sulfur atom bonded to four oxygen atoms, carrying a 2- charge. The drawing would visually represent these units interacting to form the neutral salt.
The process is quite straightforward once you get the hang of it. You identify the acid and the base (ammonia or an amine). The acid provides the anion, and the ammonia provides the ammonium cation (or a substituted ammonium cation if it's an amine). Then, you simply draw these ions and combine them in a way that makes the overall compound electrically neutral. It’s a fantastic way to connect the abstract world of chemical formulas to something you can sketch and understand visually. So, next time you encounter a reaction involving ammonia or an acid, try drawing the resulting ammonium salt. You might just find yourself enjoying chemistry a lot more!
