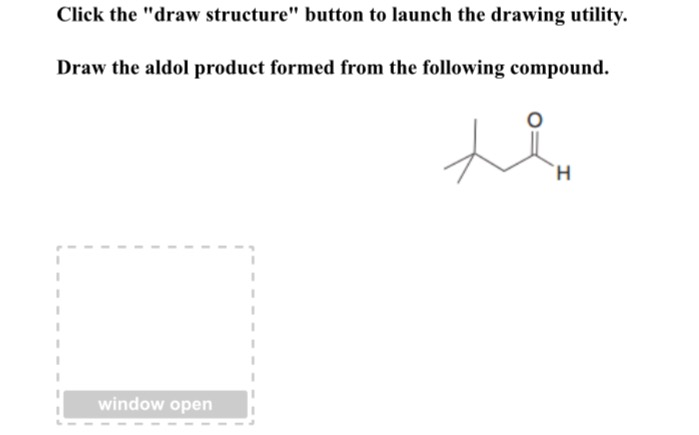
Draw The Aldol Product Formed From The Following Compound.
Ever looked at a complex chemical reaction and thought, "That looks like a puzzle, but way cooler"? Well, buckle up, because we're about to dive into the wonderfully wiggly world of the Aldol reaction, and specifically, how to predict its amazing products! It's like having a secret superpower to see what molecules can create when they decide to get together and form something new and exciting. Forget boring textbooks; this is chemistry with a side of delightful discovery!
Why is this particular bit of organic chemistry so special? Think of it as one of nature's fundamental building blocks. The Aldol reaction is responsible for forging the carbon-carbon bonds that form the backbone of so many molecules we encounter every day, from the flavors in our food to the pharmaceuticals that keep us healthy. Understanding it isn't just for the lab coat crowd; it's about appreciating the elegant dance of atoms that creates the very fabric of our material world. And the best part? With a little guidance, you can learn to predict the outcome of these transformations yourself. It’s incredibly satisfying to see a seemingly chaotic mix of ingredients coalesce into a predictable, structured product.
Unlocking the Secrets of Aldol Creations
So, what exactly is the goal when we talk about drawing the Aldol product? It's all about understanding how two molecules of the same compound (or sometimes two different ones, but we're keeping it simple today!) can join hands, or rather, bond together, under specific conditions. This joining process is more than just a random hug; it's a chemically directed event with predictable steps. The primary benefit of mastering this skill is gaining a powerful predictive tool. Instead of just reacting a compound and hoping for the best, you can anticipate what you'll get. This is crucial in designing synthetic pathways for new materials, optimizing existing processes, and even troubleshooting unexpected results in the lab.
The Aldol reaction, in its most common form, involves an aldehyde or a ketone. These are functional groups that have a carbon atom double-bonded to an oxygen atom (C=O). The magic happens because these compounds have special hydrogen atoms attached to the carbon atom next to the carbonyl group. These are called alpha-hydrogens, and they are surprisingly acidic. Imagine them as being a bit eager to leave their original spot, especially when a base is around to give them a nudge.
Here’s where the fun begins. When a base (let’s call it our helpful facilitator) comes along, it can pluck one of these precious alpha-hydrogens away. This leaves behind a negatively charged species called an enolate ion. Think of the enolate ion as being a highly reactive, nucleophilic character, ready to attack! It's like a tiny, charged magnet with a hankering to connect with something positive.
Now, in the Aldol reaction, another molecule of the same aldehyde or ketone is hanging around. This second molecule still has its intact carbonyl group (the C=O). The negatively charged enolate ion, with its newfound desire to form bonds, spots this electron-deficient carbonyl carbon and swings in for a perfect connection. This attack forms a new carbon-carbon bond, linking the two molecules together.
The initial product of this attack is called an aldol addition product. The name itself is a clue: "ald" for the aldehyde (or ketone) starting material and "ol" for the alcohol functional group that is also formed in this process. It’s a beautiful combination, showing the transformation within a single molecule.
"Understanding the Aldol reaction is like learning a secret handshake for molecules. Once you know the steps, you can predict who will join with whom and what they will become!"
So, when you are given a specific compound and asked to draw the Aldol product, here's the mental checklist you'll want to run through:
- Identify the alpha-carbons and alpha-hydrogens: Look for the carbon atoms directly adjacent to the carbonyl (C=O) group. Are there any hydrogen atoms attached to these carbons? These are your alpha-hydrogens, the key players in this reaction.
- Visualize the enolate formation: Imagine a base removing one of these alpha-hydrogens. This creates the enolate ion, where the negative charge is typically delocalized between the alpha-carbon and the oxygen of the original carbonyl.
- Locate the electrophilic site on a second molecule: Find the carbonyl carbon (the C=O) on a separate molecule of the same starting material. This carbon is electron-deficient and ready to be attacked.
- Connect the dots: Draw a bond between the carbon atom of the enolate ion (the one that lost the hydrogen) and the carbonyl carbon of the second molecule.
- Reconfigure and add the alcohol: The oxygen of the attacked carbonyl group will pick up a proton (usually from the solvent or the conjugate acid of the base), turning it into an alcohol (-OH) group. Make sure to place this new -OH group on the carbon that was originally the carbonyl carbon of the second molecule.
By following these steps, you can systematically construct the Aldol addition product. It’s a process of careful observation and logical connection, much like solving a visual puzzle. The beauty of this reaction lies in its predictability and its fundamental role in organic synthesis. So, the next time you see an aldehyde or ketone, remember the potential for Aldol magic! It’s a testament to how simple rules can lead to complex and useful molecular architectures.
