Draw The Acetal Produced When Ethanol Adds To Propanone.
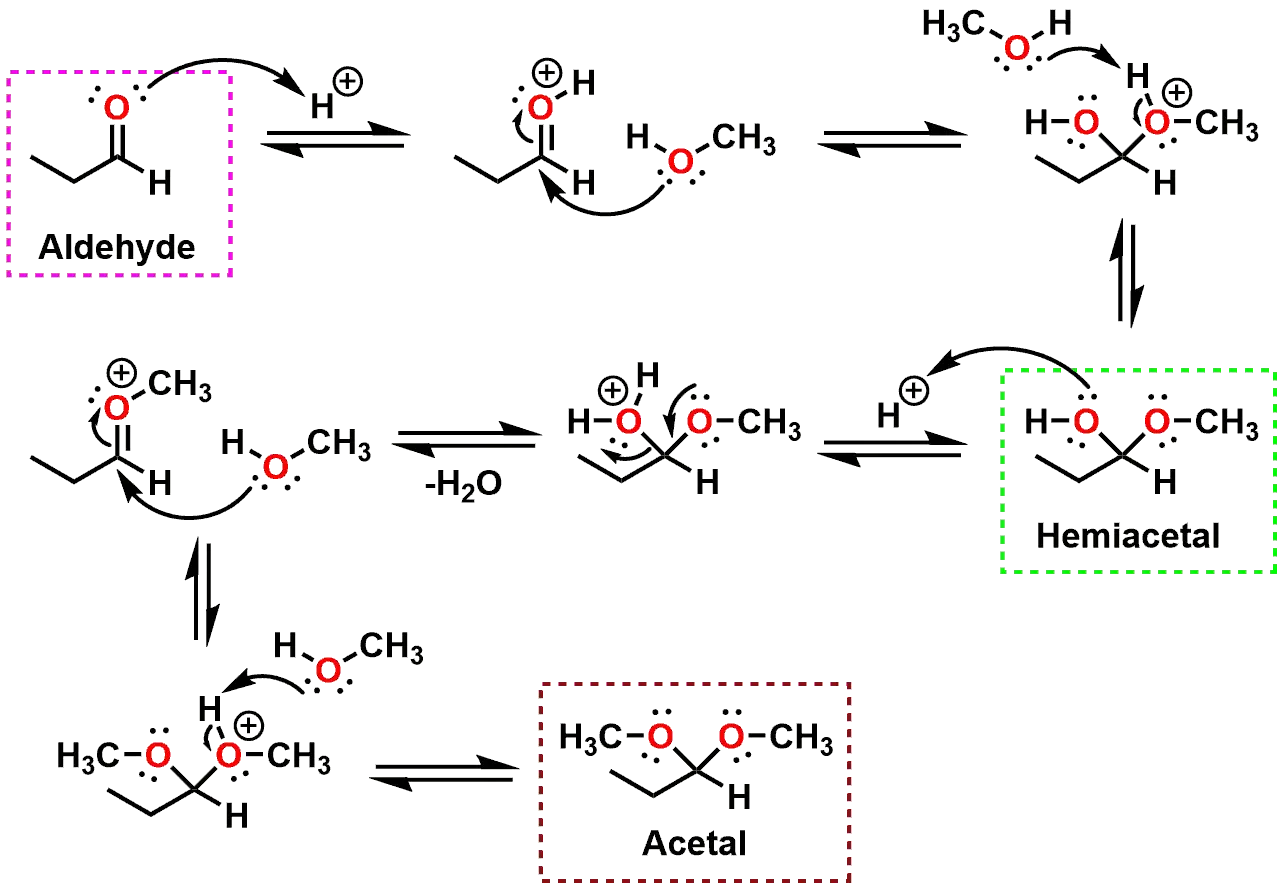
Ever wondered how chemists whip up all sorts of interesting molecules? Sometimes, it's a bit like following a recipe, but instead of flour and sugar, we're using smaller building blocks called molecules. Today, let's peek behind the curtain and explore a neat chemical reaction: the formation of an acetal from ethanol and propanone. It might sound a bit technical, but understanding this process is like unlocking a tiny piece of how the world around us works, and honestly, it's pretty cool!
So, what exactly is an acetal, and why should we care about it? Think of acetals as versatile chemical intermediaries. They're formed when an alcohol (like our friend ethanol) reacts with a carbonyl compound (in this case, propanone, also known as acetone). The main purpose of creating acetals is often for protection. In complex chemical syntheses, certain parts of a molecule might be reactive and get in the way of the desired reaction. By converting them into acetals, chemists can temporarily 'mask' these reactive groups, allowing other transformations to happen smoothly. Once the main reaction is done, the acetal can be easily removed, revealing the original functional group. This is a fundamental technique in organic chemistry, making it a key concept for anyone studying the subject.
The benefits are quite significant. Beyond just protecting functional groups, acetals are also used as solvents and can be found as components in certain fragrances and even in some pharmaceuticals. In educational settings, drawing the formation of an acetal is a classic exercise that helps students understand reaction mechanisms, stereochemistry, and the principles of functional group manipulation. It’s a tangible way to visualize how atoms rearrange and bonds form and break. In daily life, while you might not directly encounter an acetal being formed, the products of reactions that utilize acetal protection could be part of the materials, medicines, or even food additives we interact with every day.
Curious to explore this further? You don't need a fancy lab coat! The most straightforward way is to draw the reaction. Grab a piece of paper and a pencil. First, draw the structure of ethanol – that's two carbons with an -OH group attached. Then, draw propanone, which is a three-carbon chain with a double-bonded oxygen in the middle. The reaction typically requires an acid catalyst. You'll see the oxygen from the alcohol's -OH group attack the carbon of the double-bonded oxygen in propanone, and then another ethanol molecule gets involved. The result is a cyclic or acyclic structure with two ether linkages originating from the same carbon. Looking up "acetal formation mechanism" online will give you plenty of visual aids to follow along. You can even find interactive simulations that let you build molecules and watch reactions unfold!
It’s a fascinating journey from simple starting materials to a more complex, yet incredibly useful, molecule. So, next time you hear about a chemical reaction, remember there's often a clever reason and a useful application behind it, just like the humble acetal.
