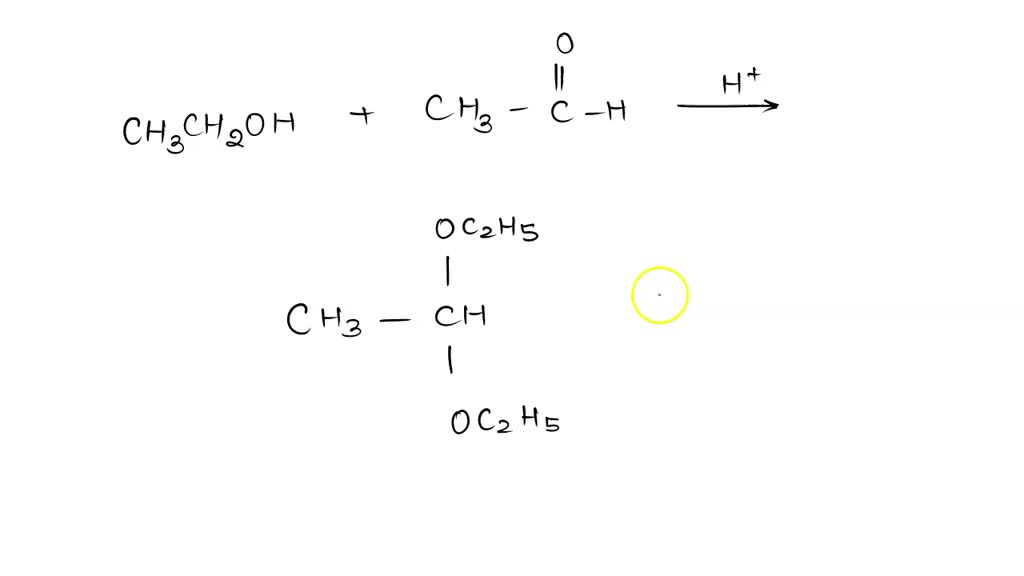
Draw The Acetal Produced When Ethanol Adds To Ethanal

Let's talk about a little chemical shindig. You know, the kind that happens when two molecules decide to get together and make something new. We're not talking about a wild party with flashing lights and questionable decisions, though sometimes chemistry can feel that way. We're talking about something a bit more… elegant. Or at least, that's what I like to tell myself. My unpopular opinion? Chemistry can be pretty darn fun, even when it sounds like it belongs in a super-serious textbook.
Today, we're going to draw something. Don't panic. This isn't about getting your artistic skills judged. It's more like sketching out a recipe. A chemical recipe, if you will. Imagine you've got two characters ready for a meet-cute. On one side, we have ethanol. Think of ethanol as that friendly, approachable alcohol. It's the stuff in your favorite drink, the one that makes karaoke nights a little more adventurous. It’s got a simple structure, kind of like a little happy family with a carbon, some hydrogens, and a hydroxyl group (that's the fancy O-H bit).
On the other side of this chemical ballroom, we have ethanal. Now, ethanal is a bit different. It's also an alcohol, but it's an aldehyde. Aldehydes are known for being a bit more reactive. They're like the slightly more energetic cousin at the family reunion. Ethanal has its own little carbon-hydrogen-oxygen setup, and the oxygen is doing a bit of a double-bond jig with the carbon. It's a bit like a single person at a dance, eager to pair up.
So, what happens when ethanol decides to tango with ethanal? They get together, they do a little molecular shuffle, and they produce something called an acetal. Yep, that's the magic word. An acetal. It sounds like something you'd find in a fancy perfume or maybe a really potent cleaning product. But in this case, it's just the result of our friendly ethanol and the slightly more zesty ethanal getting acquainted.
Now, when we talk about "drawing" this acetal, we're not reaching for our crayons. We're using our imagination and a bit of chemical shorthand. Imagine the ethanal molecule, with its eager double-bonded oxygen. Then, imagine the ethanol molecule, with its hydroxyl group, ready to offer up its hydrogen and bond with that oxygen. It's like a handshake, but with atoms. And a little bit of water is usually involved in the process, kind of like the chaperones at the dance, making sure everything is proper.
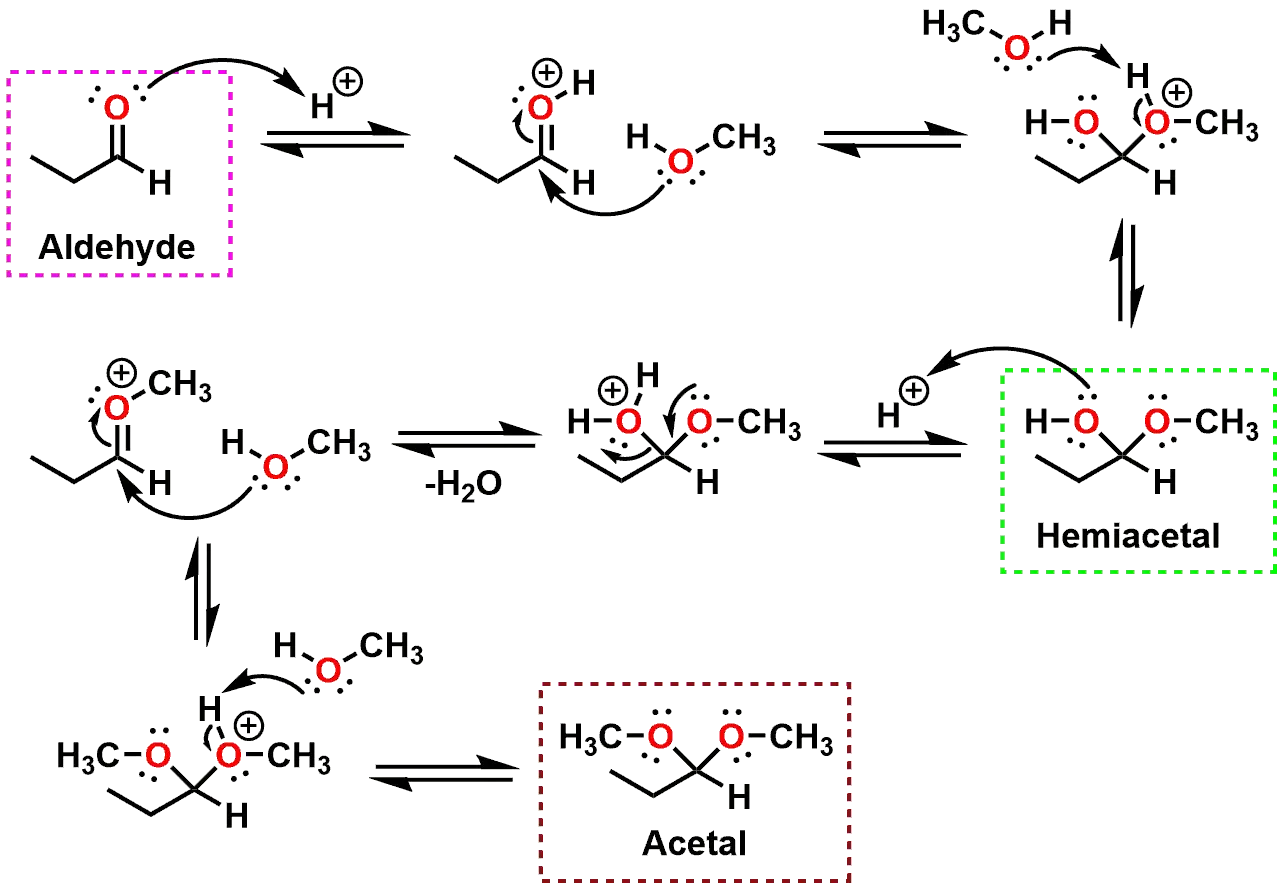
So, you've got the ethanal. It's got its carbon atom with a double bond to an oxygen, and then it's attached to a hydrogen and a methyl group (that's a carbon with three hydrogens). Think of it like this: a central carbon, a hanging hydrogen, a little CH3 group, and then that oxygen doing a lively dance with the carbon. Now, the ethanol comes along. It's a CH3-CH2-OH. That OH part is where the action is.
What happens is that the oxygen from the ethanol (the one with the H still attached) comes and snuggles up to the carbon of the ethanal. And the hydrogen from that ethanol's OH group? Well, it decides to take a little vacation and pair up with the oxygen that was already double-bonded to the ethanal's carbon. And poof! You get water. It's like a little chemical divorce and remarriage all at once. And the carbon that used to be doing a double-time dance with oxygen is now doing a single, more committed dance with two different oxygen atoms. One of those oxygen atoms is the one that used to be part of the ethanal, and the other one is the one that came all the way from the ethanol.
The result is our star of the show: the acetal. It's a carbon atom that is now bonded to two oxygen atoms. One of those oxygen atoms is also bonded to a hydrogen atom (that's the leftover from the ethanol's OH). The other oxygen atom is bonded to a carbon chain (that's the ethyl part from the ethanol). And then, of course, you still have the original hydrogen and the methyl group attached to that central carbon. It's like the carbon went from being a social butterfly to a rather committed partner, holding hands with two different oxygen atoms. And one of those oxygen atoms is holding hands with a hydrogen, and the other oxygen atom is holding hands with another carbon chain.
When you draw it out, it looks something like this: a central carbon atom. To this carbon, you attach a hydrogen. You also attach a methyl group (CH3). Then, you attach two oxygen atoms. One of these oxygen atoms is attached to a hydrogen atom. The other oxygen atom is attached to an ethyl group (CH2CH3). And there you have it. The grand and glorious acetal! It's got a bit of the ethanal's character and a whole lot of the ethanol's influence. It’s a true hybrid, a chemical love child, if you will. And to think, it all started with a simple meet-cute between two relatively straightforward molecules. It's not exactly a Rembrandt, but it's got its own quiet beauty, don't you think? A little bit of molecular art for your mind.
