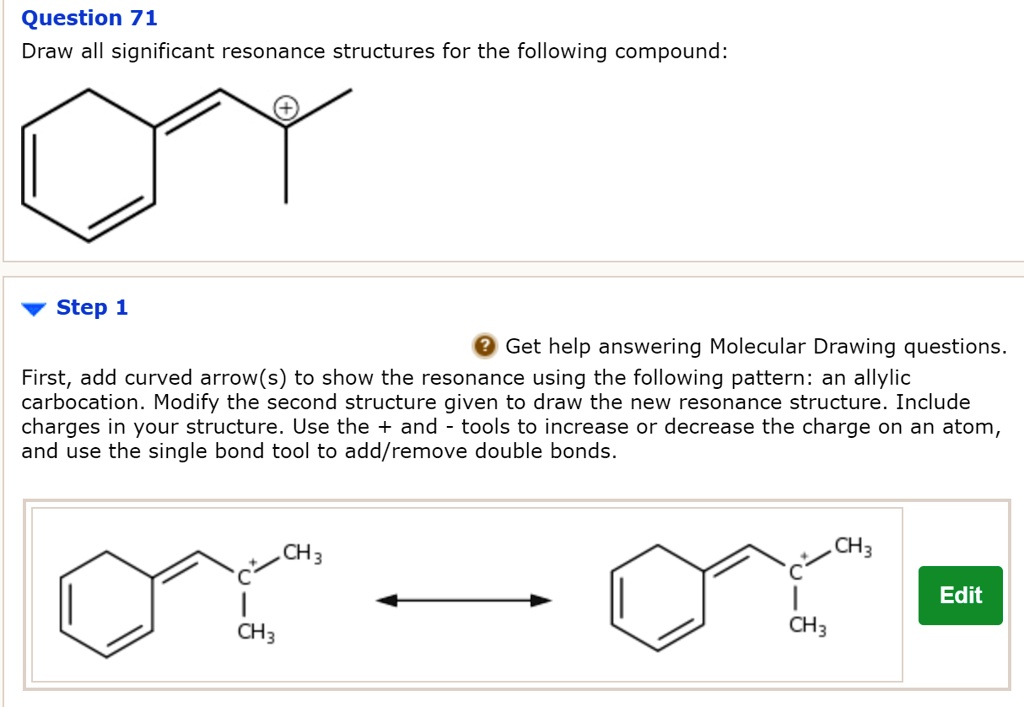
Draw Significant Resonance Structures For The Following Compound:
Ever found yourself doodling absentmindedly during a long phone call, or maybe meticulously arranging your desk for optimal feng shui? There's a delightful human impulse at play here: the desire to find patterns, to explore possibilities, and to understand the underlying order of things. And when it comes to understanding how molecules behave, a particularly fascinating and, dare I say, addictive activity emerges: drawing resonance structures! It might sound like something reserved for hardcore chemists, but the beauty of resonance structures is that they unlock a deeper appreciation for the subtle, dynamic nature of chemical bonds, a concept with surprising relevance to our everyday lives.
So, what exactly are resonance structures, and why would anyone spend time drawing them? In essence, they're a way to represent molecules where a single Lewis structure just doesn't quite cut it. Imagine trying to describe a chameleon – is it green? brown? a mix? It's constantly shifting! Molecules with resonance are a bit like that. Resonance structures are different plausible arrangements of electrons within a molecule that, when averaged together, give a more accurate picture of the molecule's true structure and stability. This concept is crucial for predicting chemical reactivity, understanding reaction mechanisms, and even designing new materials with specific properties. For instance, the stability imparted by resonance plays a role in everything from the longevity of medications to the vibrant colors of dyes.
You might not realize it, but resonance is at play in many familiar compounds. Think about the stability of the benzene ring, a fundamental building block in organic chemistry and a component of many pharmaceuticals and plastics. Or consider the ozone layer – its stability and ability to absorb UV radiation are explained by resonance. Even something as simple as the carbonate ion (CO3^2-), found in antacids and seashells, exhibits resonance. Understanding these structures helps scientists design better batteries, more efficient catalysts, and even understand how pollution disperses in the atmosphere. It’s a tiny glimpse into the invisible ballet of electrons that governs so much of the physical world.
Now, how can you get more enjoyment out of this intricate dance of electrons? Firstly, start simple. Don't jump into overly complex molecules right away. Begin with common examples like the acetate ion or nitrate ion. Secondly, visualize the movement of electrons. Think of it like a game of "musical chairs" for electrons. Curved arrows are your best friends here, showing where electrons move from one atom to another. Thirdly, embrace the imperfection. Remember, no single resonance structure is the "correct" one. They are all contributors to the overall picture, and the true structure is a hybrid. Finally, don't be afraid to make mistakes! Every wrong turn is an opportunity to learn and refine your understanding. The satisfaction of finally nailing a complex resonance contributor is incredibly rewarding. So, next time you encounter a molecule that seems a bit… elusive, try drawing its resonance structures. You might just discover a newfound appreciation for the dynamic beauty of chemistry!
