Draw O-nitroanisole. Draw Out The Nitro Group Including Formal Charges.
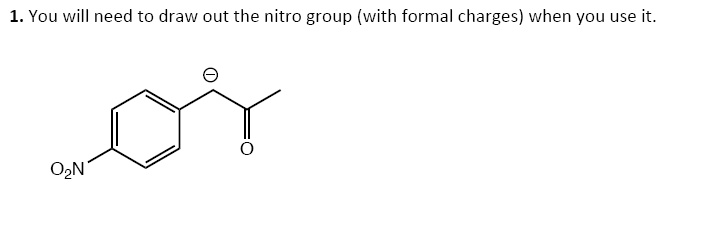
Hey there, coffee buddy! So, guess what we're diving into today? No, it's not the latest gossip or figuring out what to have for lunch (though those are important, obviously!). We're gonna doodle a little. Yep, you heard me. We're talking about drawing O-nitroanisole. Sounds fancy, right? But honestly, it's just a molecule. A bunch of atoms hanging out, being all… molecular. So grab your imaginary pencil, or a real one if you're feeling ambitious, and let's get this drawing party started!
First things first, what is O-nitroanisole? Think of it as a cozy little organic compound. It's got this benzene ring, which is like, the ultimate party animal of the molecule world. Always showing up, looking all hexagonal and stable. And then, attached to this party ring, we've got a couple of other interesting guests. One is an —anisole part. That's basically an oxygen atom chilling with a methyl group (that's a carbon with three hydrogens, super simple). It’s like the friendly neighbor who always brings cookies.
And then, the main event, the star of our drawing show: the nitro group. This guy is a bit more dramatic. It's not just a simple atom; it's a whole little crew. And drawing it right, with all its bits and bobs, is key. We have to get the formal charges in there, no skipping this step! It’s like sending a postcard with the wrong address; it just won't get to where it needs to be, you know?
So, let's break down the O-nitroanisole drawing. We’ll start with our trusty benzene ring. Remember those? Six carbons, all bonded to each other in a hexagon. And each carbon, usually, has a hydrogen attached to it. But in our case, some of these hydrogens are gonna get evicted to make room for our other guests. It’s like a tiny apartment building, and sometimes you need to make space for new tenants.
Okay, so the ring is drawn. Looking good! Now, for the anisole part. We're attaching this to one of the carbons on the ring. Remember, it’s an oxygen atom. Just a big ol' 'O'. And this oxygen is also connected to a methyl group. So, that's a carbon atom (let's call it C) bonded to three hydrogen atoms (H). So, you'll have a C-H bond, and then another C-H bond, and a third C-H bond, all radiating from that single carbon. Think of it as a little tripod of hydrogens. Cute, right?
This oxygen atom, the one connecting the anisole part to the benzene ring, is super important. It’s kind of the bridge. It’s also where the 'O' in O-nitroanisole comes from. Like, 'O, look, it's oxygen!' Get it? Okay, maybe that joke was a bit much. My bad. Coffee hasn't kicked in fully yet.
Now, the real fun begins: the nitro group. This is where things get a little more… electrified. The nitro group is written as -NO₂. But that's just the shorthand, the quick and dirty way. When we draw it out, we're gonna see a nitrogen atom and two oxygen atoms. But they aren't just chilling side-by-side. Oh no. They've got bonds, and those bonds have charges. This is the juicy part!
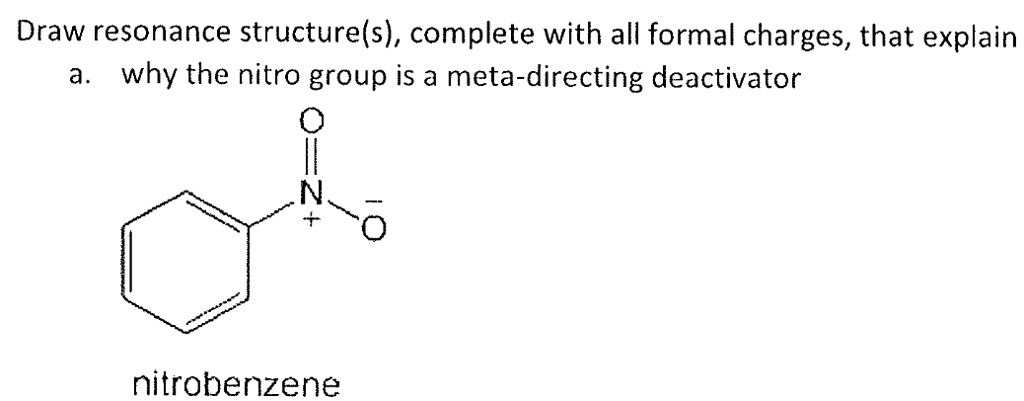
So, we have a nitrogen atom. Let's call him N. He’s gonna be bonded to one of the carbons on our benzene ring. And then, attached to this nitrogen, we have our two oxygen atoms. Let's call them O1 and O2. Now, here's where the magic happens. The nitrogen atom in a nitro group doesn't just have single bonds with both oxygens. That would be too simple, wouldn't it? Life's never that easy, especially in chemistry.
Typically, in the nitro group, you'll see one double bond and one single bond. So, the nitrogen will be double-bonded to one oxygen, and single-bonded to the other. Think of it as one oxygen getting all the attention with a strong, shared-electron relationship, and the other oxygen getting a more… exclusive, one-on-one connection. Chemistry is all about relationships, you know?
But here's the crucial bit. We need to show the formal charges. This is what makes the drawing complete and tells the story of how these electrons are distributed. For the nitrogen atom in the nitro group, it usually carries a positive formal charge. Yep, positive! So, you’ll draw a little '+' sign next to the nitrogen. It’s like the nitrogen is feeling a bit… generous with its electrons, or perhaps a bit needy, leaving it with a positive vibe. Who knows what goes on in the minds of atoms!
And then, one of the oxygen atoms, the one that's single-bonded to the nitrogen, will carry a negative formal charge. So, you'll put a little '-' sign next to that oxygen. This is the balancing act. The positive nitrogen and the negative oxygen are like two halves of a magnet, holding each other together. It's a beautiful, if slightly charged, partnership.
What about the oxygen atom that's double-bonded to the nitrogen? Well, this one is usually neutral. No charge to see here! It's just happily sharing its electrons in that double bond, playing its part without any extra drama. It’s the chill one in the group.
So, to recap the nitro group drawing: you've got your nitrogen, often with a '+' charge. Then, you've got your two oxygens. One is double-bonded to the nitrogen and is neutral. The other is single-bonded to the nitrogen and has a '-' charge. Make sure you draw those bonds clearly! The double bond is two lines, the single bond is one. And don't forget those little charge symbols! They are the punctuation marks of our molecular sentences.
Now, where does this nitro group attach to our O-nitroanisole? The 'O' in O-nitroanisole tells us that the nitro group is attached to an oxygen atom which is then attached to the benzene ring. This is a common way to name things in organic chemistry, by the way. The 'O-' prefix usually means the substituent is attached via an oxygen atom. So, it's not directly attached to a carbon on the ring. It's a bit more indirect, a bit more sophisticated. Like, instead of knocking on the front door, it's calling the concierge.
So, you’ll have your benzene ring. Then, attached to one of the carbons of the benzene ring, you’ll have an oxygen atom. This oxygen atom is where our nitro group will be hanging out. So, the sequence goes: Benzene Ring — Oxygen — Nitrogen (with its double-bonded, neutral oxygen and single-bonded, negatively charged oxygen). And don’t forget the methyl group is also attached to that same oxygen atom. So, it’s actually: Benzene Ring — Oxygen — (Methyl Group) AND Oxygen — Nitrogen (with its charged oxygens).

Wait, that's not quite right. My brain is doing a little somersault. Let's clarify! O-nitroanisole. The 'anisole' part is the methoxy group attached to the benzene ring. So, you have a benzene ring with an -OCH₃ group attached. The 'O-nitro' means the nitro group is also attached to that same oxygen atom of the methoxy group. That’s a bit unusual, right? Usually, the nitro group would be on the benzene ring itself. Let’s re-read the name very carefully.
Ah, I see the potential for confusion! The common understanding of "O-nitroanisole" is actually p-nitroanisole or o-nitroanisole or m-nitroanisole, where the 'O' refers to the position on the benzene ring. So, the nitro group is attached to the benzene ring, and the anisole (methoxy) group is also attached to the benzene ring. The 'O-' in o-nitroanisole refers to the ortho position on the benzene ring, relative to the methoxy group. That makes way more sense! My apologies for the detour, folks. Sometimes even the simplest things can twist your brain into a pretzel.
So, let's re-do the drawing with the correct understanding. We're drawing ortho-nitroanisole. That means we have our benzene ring, looking all chic. And then we have our methoxy group (-OCH₃) attached to one of the carbons on the ring. Let’s say we pick a carbon and attach the oxygen, and then the methyl group to that oxygen. Simple enough.
Now, the 'ortho' part. On a benzene ring, carbons are numbered. If our methoxy group is on carbon 1, the 'ortho' position is carbon 2. So, we’re attaching our nitro group to the carbon at the ortho position. Remember, the nitro group is -NO₂ with its charges. So, you'll have a nitrogen atom bonded to that ortho carbon. And then, attached to that nitrogen, you'll have your double-bonded neutral oxygen and your single-bonded negative oxygen, and the nitrogen itself will have that positive charge.

So, the structure is: Benzene Ring, with a -OCH₃ group on one carbon, and a -NO₂ group on the adjacent carbon (the ortho position). And within that -NO₂ group, you draw the nitrogen with its double bond to one oxygen and single bond to another, and their respective formal charges: '+' on the nitrogen, '-' on the single-bonded oxygen, and neutral on the double-bonded oxygen. This is it! This is the money shot!
Why are these charges important, you ask? Well, they tell us a lot about how the molecule behaves! It’s like reading a person’s body language. The charges indicate electron distribution, which influences reactivity, solubility, and all sorts of other cool chemical stuff. Without them, the drawing is incomplete, like a story without an ending. And who wants a story without an ending? Not me, that’s for sure!
So, to nail this drawing, remember: 1. The Benzene Ring: Six carbons, alternating double and single bonds (or a circle in the middle to show delocalization – your call!). 2. The Methoxy Group (-OCH₃): An oxygen atom bonded to one carbon of the ring, and a methyl group (CH₃) bonded to that oxygen. 3. The Nitro Group (-NO₂): Attached to an ortho carbon of the ring. This is the crucial part for drawing ortho-nitroanisole. 4. Within the Nitro Group: A nitrogen atom bonded to the benzene ring. This nitrogen is double-bonded to one oxygen and single-bonded to another. 5. Formal Charges: The nitrogen gets a '+' charge. The single-bonded oxygen gets a '-' charge. The double-bonded oxygen is neutral.
And there you have it! You've just drawn O-nitroanisole (or rather, ortho-nitroanisole, the most common interpretation). Pretty neat, huh? It might seem like a lot of little details, but once you get the hang of it, it becomes second nature. Like riding a bike, or remembering your friend’s birthday (ahem, are you listening?).
So, next time someone asks you to draw this molecule, you can confidently whip out your imaginary pencil and show them exactly what’s what. You’ll be the molecule drawing guru, the atom artist, the chemical cartoonist! And all it took was a little bit of focus, a dash of understanding, and maybe another cup of coffee. Happy drawing!
