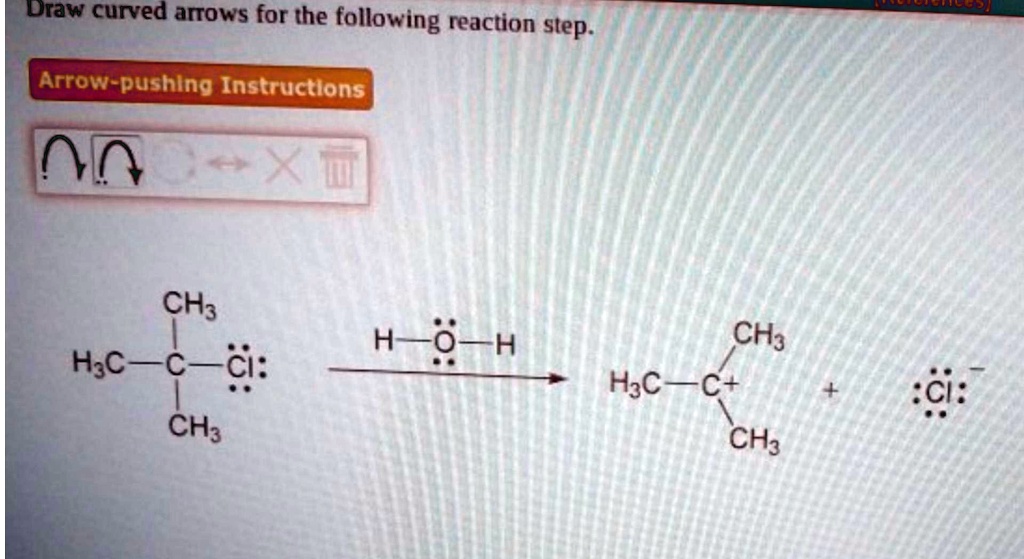
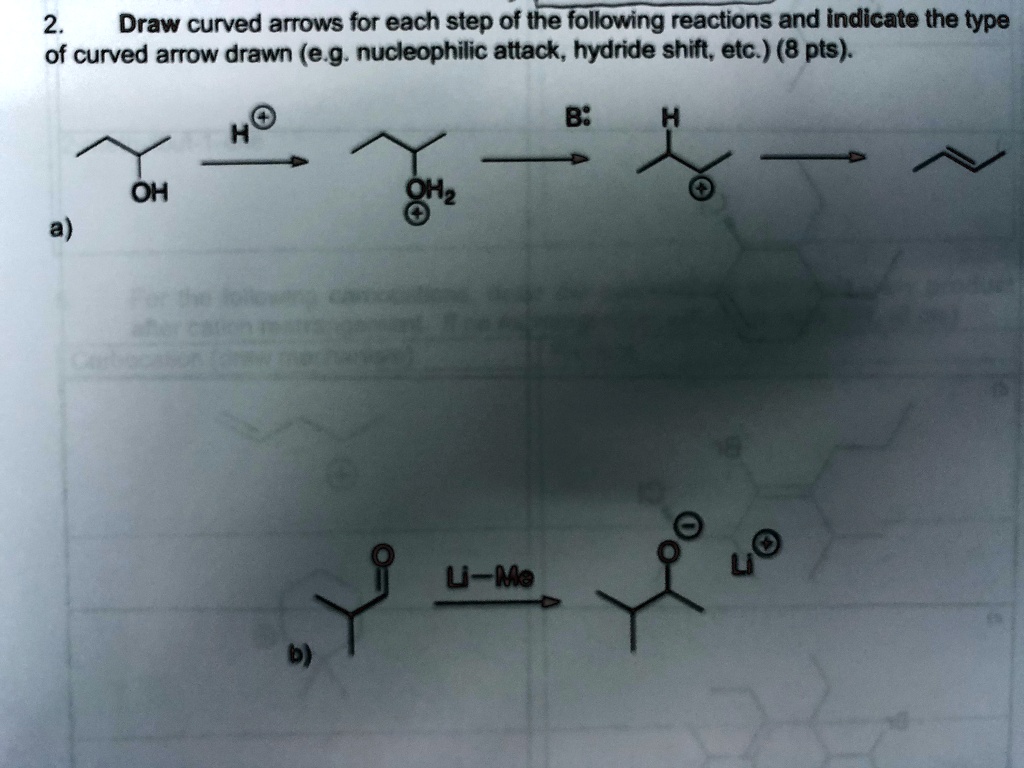
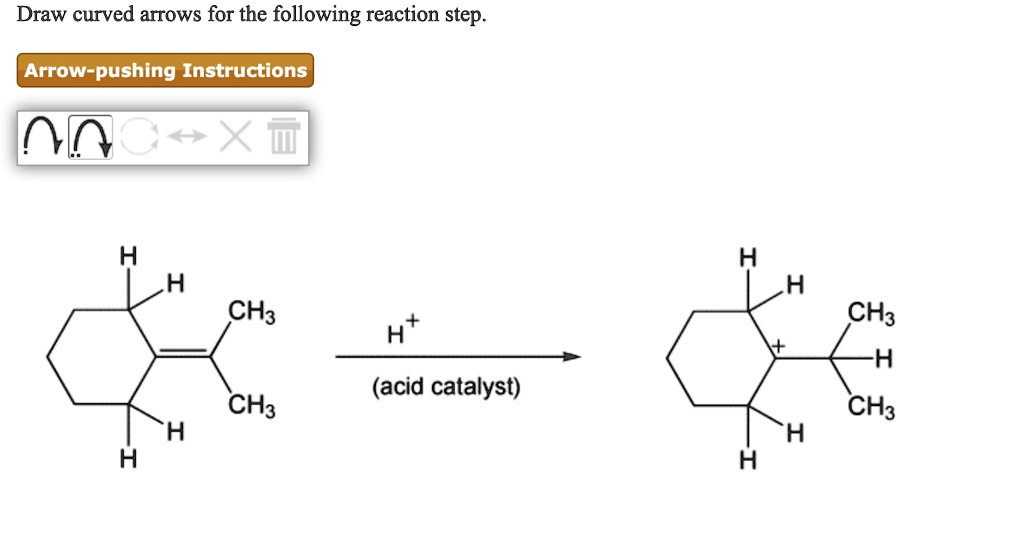
Draw Curved Arrows For The Following Reaction Step.
Alright, chem-curious pals! Ever looked at a chemical reaction and thought, "Whoa, what’s going on there? It’s like a secret dance party of atoms!" Well, sometimes, to really see that dance, we need a little help from our trusty sidekick: the curved arrow. Think of them as the little spotlights that show us exactly where the electrons – those tiny, energetic troublemakers – are deciding to go next. It's like following a breadcrumb trail, but way more exciting because instead of finding a cookie, we're uncovering the magic of how molecules change!
Now, don’t let the fancy name scare you. "Curved arrow" just means… well, an arrow that’s curved! Like a tiny, animated thought bubble for electrons. And the "following the reaction step" part? It’s simply about observing the very next move in this molecular ballet. We’re not trying to choreograph the whole grand finale, just one little, perfectly executed pirouette. Easy peasy, lemon squeezy!
Imagine you're at a super-exclusive party, and all the cool kids (those are our electrons) are hanging out. Suddenly, one group of electrons decides it’s time for a change of scenery. A curved arrow is like you pointing and saying, "Hey, look! Those electrons are shuffling over to that other molecule over there!" It’s that direct. It’s the ultimate gossip column for the microscopic world.
Let’s say we have a situation where one molecule has a whole bunch of electrons just chilling, practically begging for an adventure. We call this a nucleophile – a fancy word for "electron-lover." They're like the life of the party, always ready to share their electron friends. On the other hand, we might have another molecule that’s feeling a bit… electron-deprived. Maybe it has a spot that’s desperately looking for some electron company. That’s an electrophile, the "electron-avoider" or, more accurately, the electron-seeker!
When these two meet, it’s like a romantic comedy, but with atoms. The nucleophile, with its abundance of electron goodness, sees the electrophile’s empty spot and thinks, "Oh, you poor, lonely atom! Let me share my electron treasures with you!" And poof! That’s where our curved arrow comes in. It starts at the source of the electrons – the nucleophile – and swoops over to the spot where they’re going to be welcomed with open arms – the electrophile.
It's like a cosmic high-five, but with the ultimate reward: a brand new bond!
Sometimes, the electrons aren’t just moving to a new molecule; they’re shifting within the same molecule. Think of it like rearranging the furniture in your living room. You’re not buying new stuff; you’re just moving what you already have to make things look and feel better. Maybe a double bond decides it needs to chill out and become a single bond, or a lone pair of electrons decides to form a brand new bond. Again, the curved arrow is our trusty guide, showing us the direction of that electron migration.

So, when you see a chemical reaction and the question pops up, "Draw curved arrows for the following reaction step," don’t panic! Just remember the fundamental rule: electrons move. And our curved arrows are here to tell us where they’re moving and from where. They’re the silent storytellers of chemical transformations.
Let's get a little more specific, shall we? Imagine you've got a molecule that’s like a grumpy old man, hoarding all his spare change (those are his lone pair electrons). He’s not using them, and they’re just sitting there. Then, a cheerful young person (an electrophilic atom with an empty space) walks by, looking a bit down. Our grumpy old man, in a surprising act of generosity (or maybe just to get rid of them!), decides to offer some of his spare change. The curved arrow would start right at the lone pair of electrons on the grumpy old man and swing over to the empty spot on the cheerful young person. It's a beautiful thing, really. Electrons finding their happy place.
Or, what if a double bond, which is basically like two people holding hands really tightly (two pairs of shared electrons), decides that one of them is tired of holding on? One of those hands might let go and reach out to grab onto something else. The curved arrow would start from the middle of that double bond and point to where the electron is going. It’s all about following the flow, the electron traffic jam, or the electron getaway!
The key is to look for the electron-rich areas and the electron-poor areas. The electron-rich ones are like overflowing picnic baskets, ready to share. The electron-poor ones are like empty stomachs, eagerly awaiting a delicious meal. And the curved arrow is the waiter, showing you exactly which picnic basket is serving which hungry tummy. It’s a symphony of electron transfer, and the arrows are the conductor’s batons, guiding the entire performance.
So, next time you’re faced with drawing these little wonders, take a deep breath. Remember the electron party. Remember the electron migration. Remember that you’re not just drawing lines; you’re tracing the path of pure, unadulterated chemical change. You’re becoming a detective of the molecular realm, and the curved arrow is your magnifying glass. It’s fun, it’s visual, and it’s the absolute heart of understanding how the world around you transforms, one electron dance at a time. Go forth and draw with glee!
