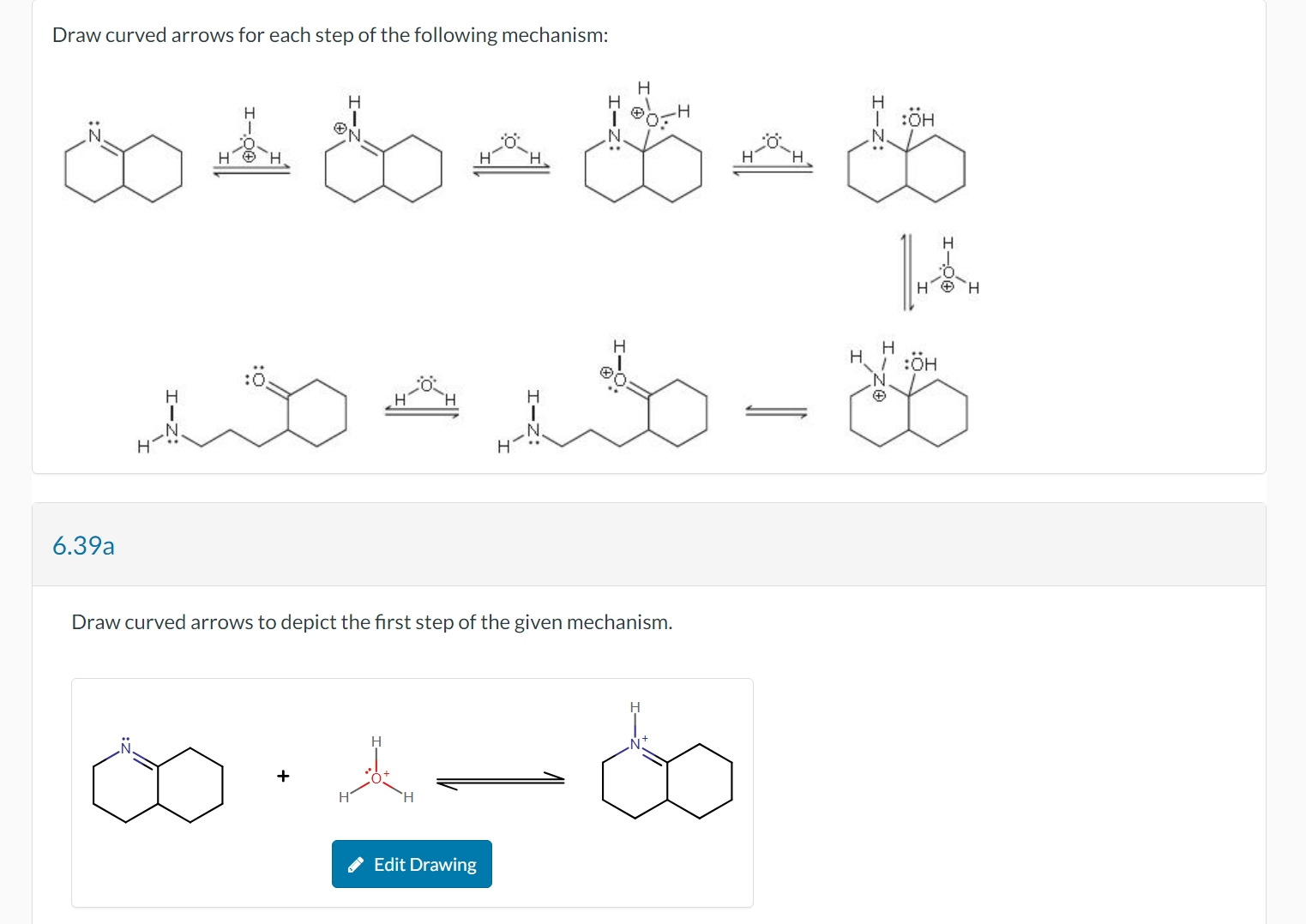
Draw Curved Arrows For Each Step Of The Following Mechanism
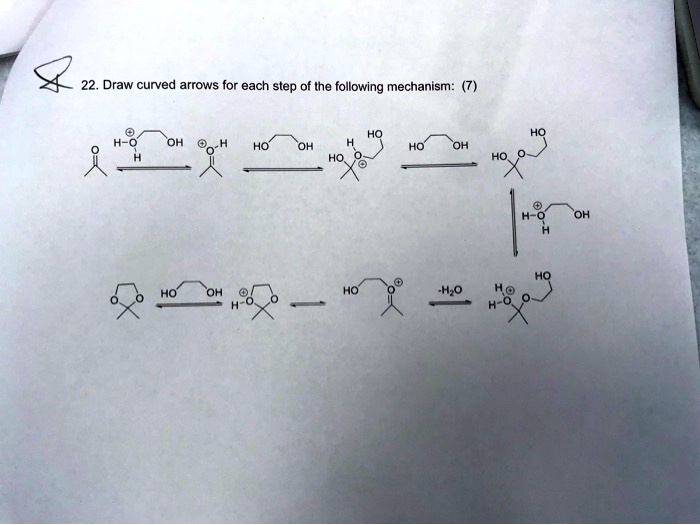
Hey there, coffee buddy! Grab your mug, settle in. We’re about to dive into something that sounds a tiny bit intimidating, but trust me, it’s actually pretty cool. We’re talking about drawing curved arrows for reaction mechanisms. Yeah, I know, “mechanism” sounds like something your car needs, but in chemistry, it’s just the step-by-step story of how molecules actually do their thing. It’s like the behind-the-scenes gossip of the chemical world.
And these curved arrows? They’re our little comic strip speech bubbles, showing where the electrons are going. Think of them as tiny, enthusiastic dancers, twirling and leaping from one atom to another. Pretty neat, right? So, imagine we’ve got this particular chemical dance happening, and our job is to show the moves. We’re going to break down each single step, and for every single step, we’ll be drawing a curved arrow. It’s all about clarity, so no one misses a beat in this molecular ballet.
So, picture this: you’ve got some reactants chilling out, maybe feeling a bit bored. Then, BAM! Something happens, and suddenly, new bonds are forming, old ones are breaking, and we’ve got products. But how? That’s where our arrow-drawing skills come in. It's like we're the directors of a tiny chemical movie, and these arrows are our storyboard. They tell us exactly what’s going on with the electrons, the real movers and shakers in any reaction. Without them, it’s just a bunch of atoms looking lost.
Let’s get our hands dirty, shall we? Metaphorically, of course. We’re not actually going to touch any chemicals unless you’ve got a fume hood handy. But we are going to look at a specific reaction and map out its electron journey. This is where the fun really begins, because you get to see the logic behind it all. It's not just random chaos; there's a beautiful, elegant flow to how these things work. You just need to know how to follow the breadcrumbs, and our arrows are those delicious breadcrumbs.
So, what kind of reaction are we talking about today? Let’s pick something relatively straightforward, something that doesn’t require a Ph.D. in advanced organic chemistry just to understand the starting materials. Maybe something involving a simple acid-base reaction, or a little bit of nucleophilic attack. These are the bread and butter of many chemical transformations, and they’re perfect for practicing our arrow-pushing. Don't worry if it looks like gibberish at first. We'll untangle it together, one arrow at a time.
The goal here is to be super precise. Every arrow has a meaning, a direction, and a starting point and an ending point. It’s like sending a text message with a very specific emoji sequence. You can’t just send a random smiley face; it has to convey the exact feeling. Same with our arrows. They have to show the exact electron movement. So, pay attention to the tails of the arrows and the heads. They’re not interchangeable!
Step 1: Setting the Scene
Alright, first things first. We need our reactants. Let’s imagine we have molecule A and molecule B. Maybe molecule A is feeling a bit electron-deficient, like it’s missing something. And molecule B? Well, maybe it’s got a pair of electrons just hanging around, looking for a purpose, like a pair of socks without a match. These electrons are often called a lone pair, and they're super important players in our story.
So, in our very first step, we need to identify where the action is going to start. Where are these free-roaming electrons? And where are they going to be welcomed? This is like scouting out the dance floor. You find the people who look ready to groove and the space where the music is hitting just right. We're looking for a nucleophile (the electron-rich one, the giver) and an electrophile (the electron-deficient one, the receiver).
Think about it: electrons always move from a region of high electron density to a region of low electron density. It's like water flowing downhill, or a celebrity seeking out a paparazzi scrum. It’s just natural! So, our first task is to spot that electron surplus and that electron deficit.
Let’s say molecule B has a negatively charged atom, or that aforementioned lone pair. That’s our electron-rich superstar, ready for its close-up. And molecule A has a positively charged atom, or an atom that’s bonded to a very electronegative atom, making it partially positive. That’s our electron-poor stage, waiting for a performance. You’ve found your dancers!
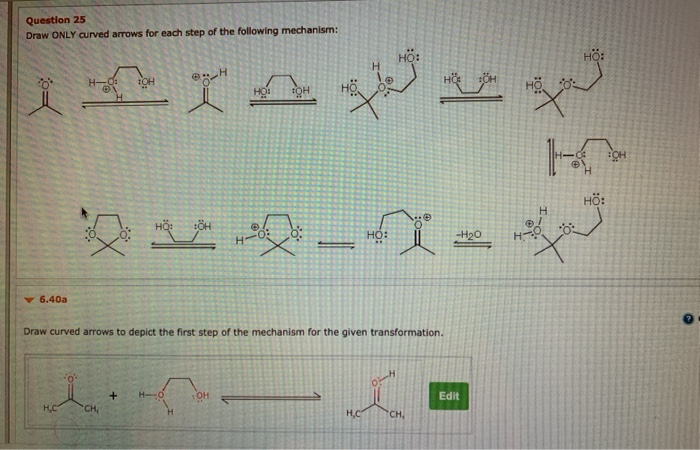
Now, for our very first curved arrow in this step, we’re going to draw it originating from the source of the electrons. This is the tail of our arrow. If it's a lone pair, the tail starts right on that pair of dots. If it's a negative charge, the tail comes from the symbol for the negative charge. It’s like pointing a spotlight directly at the electron party.
And where does it go? It points towards the electron-deficient atom. This is the head of our arrow. This shows where those eager electrons are going to land, where they’re going to make their new home, or where they’re going to form a new bond. It’s a direct route from abundance to need. Easy peasy, right? One arrow down, many to go!
The First Dive: From Lone Pair to Positive Charge
Okay, let's get really specific. Imagine we have an alcohol (like ethanol, CH3CH2OH) and a strong acid (like HBr). The oxygen in the alcohol has those lovely lone pairs, right? And the hydrogen in HBr is a bit exposed, ready to accept some electrons. So, the oxygen, with its electron-rich abundance, sees the slightly positive hydrogen. It's like a siren song for those electrons!
So, for our first step, we draw a curved arrow starting from one of the lone pairs on the oxygen atom of the alcohol. You can pick either lone pair, they’re equally eager! The tail of this arrow sits right on those little dots representing the electrons. And where does it go? It points directly to the hydrogen atom in HBr. This signifies that the oxygen is donating those electrons to form a new bond with the hydrogen.
When this happens, this hydrogen is going to leave its bond with the bromine. Bromine is a bit of a drama queen, always wanting to hog electrons. So, as the oxygen-hydrogen bond starts to form, the bond between hydrogen and bromine has to break. It’s like a handshake between oxygen and hydrogen, and as they clasp hands, hydrogen has to let go of bromine. This is crucial!
And that’s it for our first arrow in this step! We’ve shown how the electrons from the alcohol are attacking the hydrogen of the acid. It’s the initial flirtation, the spark that ignites the whole reaction. Don't forget, every arrow shows the movement of two electrons. It's a package deal. Always remember that.
Step 2: The Aftermath and Another Move
So, what happens after our first arrow has done its magic? Well, we’ve formed a new bond: O-H. The oxygen now has three bonds and a positive charge. It’s a bit overloaded, like someone who just ate way too much Thanksgiving dinner. It’s now a positively charged oxygen species, also known as an oxonium ion. Fancy, huh?
But remember what we said about the H-Br bond? It had to break. Where do those electrons from the broken H-Br bond go? They don’t just disappear into thin air! That would be chaotic. Nope, those electrons, now free from their hydrogen pal, are going to find a new home. And in this case, they’re going to land on the bromine atom.
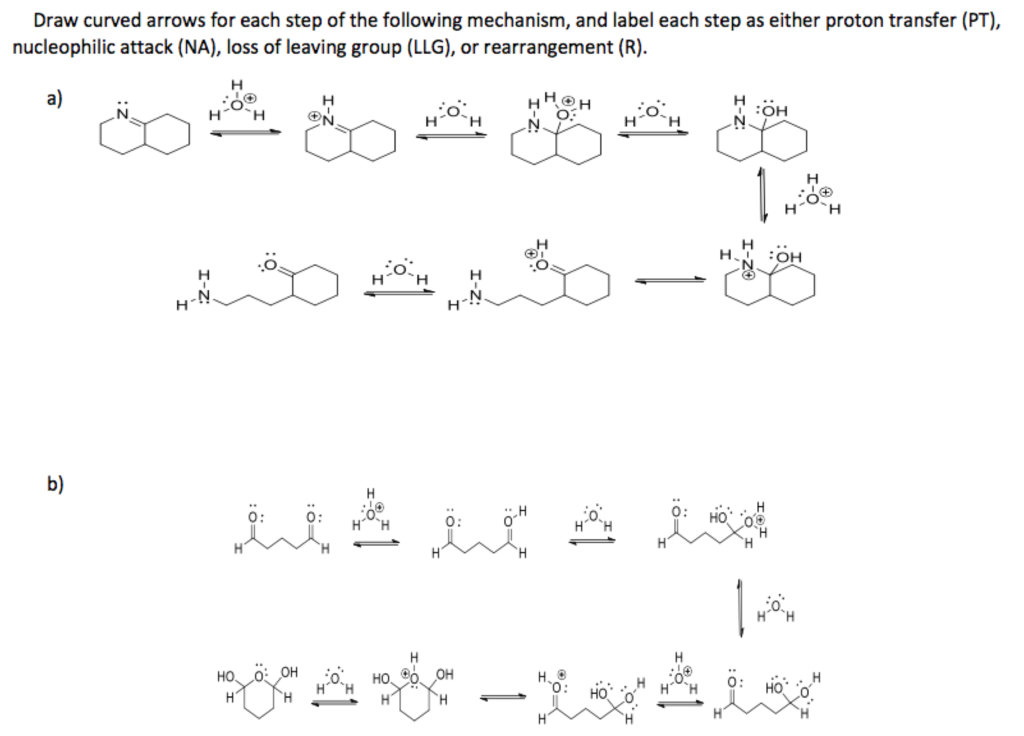
So, for our second curved arrow, we need to show this bond breaking. How do we draw an arrow for a bond breaking? It’s a little different. Instead of starting from a lone pair or a negative charge, this arrow’s tail will be placed in the middle of the bond that is breaking. You know, right where the shared electrons are. It’s like drawing a line through the middle of the H-Br connection.
And where does the head of this arrow go? It points directly to the bromine atom. This signifies that both electrons from the H-Br bond are now belonging to the bromine. Bromine was already pretty electronegative, so it happily takes on these extra electrons, becoming a bromide ion (Br-). It’s now negatively charged and probably feeling a bit smug about its electron fortune.
So, in this second step, we’ve used our second arrow to show the departure of the leaving group and the formation of the bromide ion. We’ve cleaned up the mess from the first step and dealt with the consequences. It’s all about balance and where those electrons feel most comfortable.
The Second Dive: Bond Breaking and Ion Formation
Let’s stick with our alcohol and HBr example. After that first arrow, we have our protonated alcohol (the oxonium ion) and the bromide ion (Br-). The H-Br bond is no longer there. It’s history!
So, for our second step's curved arrow, we look at the bond that just broke: the H-Br bond. We place the tail of the arrow exactly in the middle of that H-Br bond. It's like putting a marker on the line where the electrons used to be shared.
And the head of the arrow? It points directly to the bromine atom. This clearly shows that the bromine atom is now taking both electrons that used to be in the H-Br bond. Voilà! We have our bromide ion (Br-). It’s now a stable, negatively charged species. This arrow represents the heterolytic cleavage of the H-Br bond, meaning the bond breaks unevenly, with one atom getting both electrons.
So, by the end of these two steps, we’ve used two arrows. The first arrow showed the attack of the nucleophile (oxygen) on the electrophile (hydrogen). The second arrow showed the departure of the leaving group (bromine) and the formation of the bromide ion. We've gone from reactants to a protonated intermediate and a free ion. It’s like a mini-story arc in the life of these molecules.
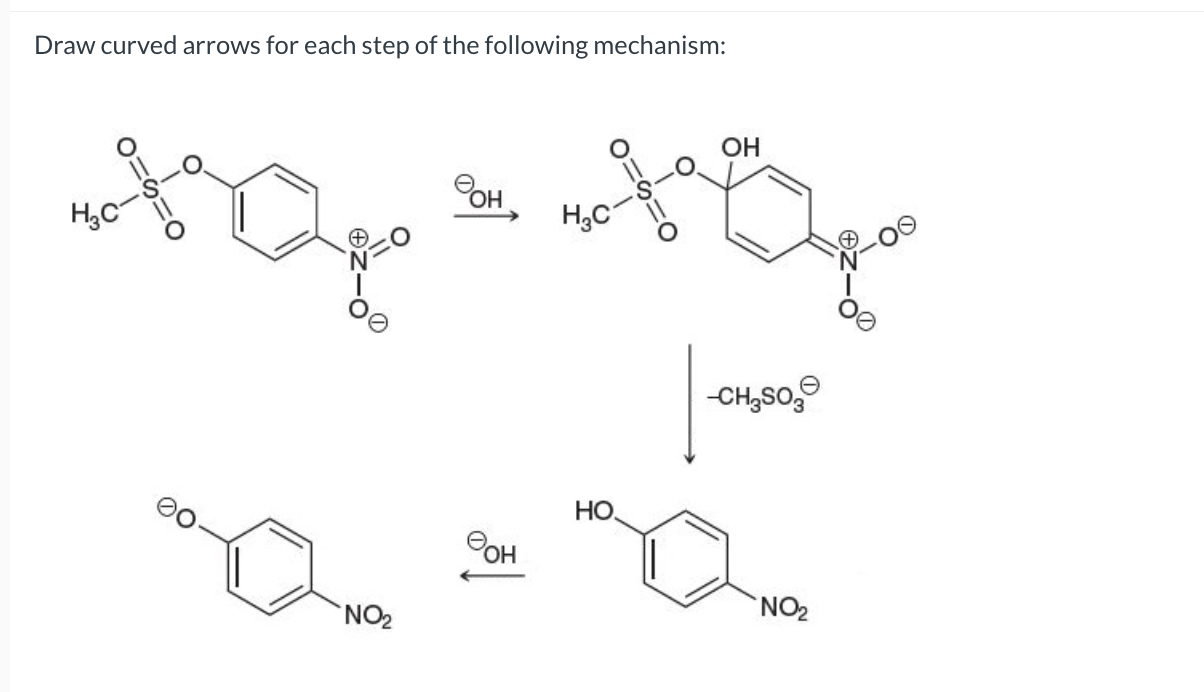
Step 3: And Then What? More Arrows!
Now, depending on what the actual overall reaction is trying to achieve, there might be more steps. This is where things can get really interesting. Let’s say, in our example, we wanted the alcohol to react further with something else. Or maybe, the bromide ion we just created is going to do something.
Let’s imagine a different scenario for a moment, to illustrate more arrow-drawing. Suppose we have a molecule with a double bond, and something with a positive charge wants to attack it. The double bond is a region of high electron density, so it’s ready to donate electrons. The positive charge is hungry!
So, for a third step, we might draw an arrow from the pi bond (that’s the second bond in a double bond, the one that’s a bit more flexible) of the double bond. The tail of the arrow goes right onto the double bond, indicating those electrons are on the move. And the head? It points to the positively charged atom of our attacking species. This forms a new bond, and the positive charge gets neutralized.
But! This might leave another part of the molecule with a negative charge, or a lone pair that can now act as a nucleophile. Or, it might create a new spot that’s electron-deficient, setting up the stage for another arrow. It’s a chain reaction, a domino effect of electron movement.
The Third Dive: Nucleophilic Attack on a Double Bond
Let’s imagine we have ethene (CH2=CH2) and we’re reacting it with something like H+ (a proton). We already did this in a way with the alcohol, but let’s look at the double bond itself.
The double bond in ethene is made of a sigma bond and a pi bond. The pi bond is the one that’s more exposed and reactive. So, if we have a free proton (H+), it’s an electrophile, right? It’s looking for electrons.
For our third step's curved arrow, we draw it originating from the pi bond of the ethene molecule. The tail of the arrow goes right onto the double bond, signifying the donation of those pi electrons. And the head of the arrow points directly to the proton (H+). This shows the proton attaching to one of the carbon atoms, forming a new C-H bond.
Now, what happens to the other carbon atom in the double bond? When the pi bond breaks to form a new bond with the hydrogen, the other carbon atom is left with a deficiency of electrons. It loses its share of the pi electrons. This carbon becomes positively charged! So, this arrow leads to the formation of a carbocation intermediate: CH3-CH2+.
This is a classic example of electrophilic addition to an alkene. We used one arrow to show the pi electrons attacking the electrophile, and in doing so, we created a new reactive species (the carbocation) which might then react further. Every arrow tells a part of the story.
The Art of the Arrow: It's All About Electron Flow
So, you see the pattern, right? We identify electron-rich areas (lone pairs, negative charges, pi bonds) and electron-deficient areas (positive charges, partial positive charges, atoms with incomplete octets). Then, we draw arrows to show the electrons moving from the rich to the poor.
A few more pointers to keep your arrow-drawing game strong:
- Arrow Tail: Always starts at the source of the electrons. This could be a lone pair, a bond, or a negative charge.
- Arrow Head: Always points to where the electrons are going. This is usually an atom that needs electrons, a bond that's forming, or where a negative charge will end up.
- Full Head vs. Half Head: A full arrow with a normal arrowhead shows the movement of a pair of electrons (like from a lone pair or a bond). A half-headed arrow (a fishhook) shows the movement of a single electron. We're mostly dealing with full arrows for mechanisms.
- “Curved” is Key: The arrows are curved to show the path of electron movement. They're not straight lines; they're showing a trajectory.
- No Magic: Electrons don't just teleport. Their movement is dictated by the stability of the resulting species. Nature likes things to be stable, so reactions tend to proceed in ways that lead to more stable intermediates or products.
- Multiple Arrows, Multiple Steps: Each arrow in a mechanism represents a distinct step. If you have a reaction that takes three steps, you'll likely draw three primary arrows to show the electron flow for those steps.
It's like learning a new language, the language of electrons. And once you start speaking it, you'll see reactions everywhere! You'll be able to predict what might happen, and understand why it happens. It’s empowering, really. You’re not just memorizing; you’re understanding the molecular choreography.
So, let’s recap our original idea: draw curved arrows for each step of the following mechanism. This means for every distinct electron-moving event, you get your own arrow. No cutting corners, no skipping beats. It’s about showing the full, unadulterated electron journey. Each arrow is a whisper of what the electrons are up to, and together, they tell the whole epic tale of the reaction.
Don't be discouraged if it takes a few tries. Even the best chemists had to start somewhere. Think of it as doodling, but with a purpose! You’re creating a visual narrative of chemical transformation. And who knows, maybe you’ll start seeing curved arrows in your dreams. (Just kidding… mostly.)
The beauty of drawing these arrows is that it forces you to think critically about electron distribution and stability. You're not just passively receiving information; you're actively engaging with it. You're the detective, and the arrows are your clues leading you to the solution of the reaction mystery. So, go forth and draw! May your arrows be ever so clear and your electron movements ever so logical.
And remember, if you’re ever unsure, just ask yourself: where are the electrons happiest? Where do they want to go? Usually, the answer will guide your arrow. It’s all about following that electron trail, that invisible, yet incredibly powerful, force that drives the entire chemical universe. Happy arrow-pushing!
