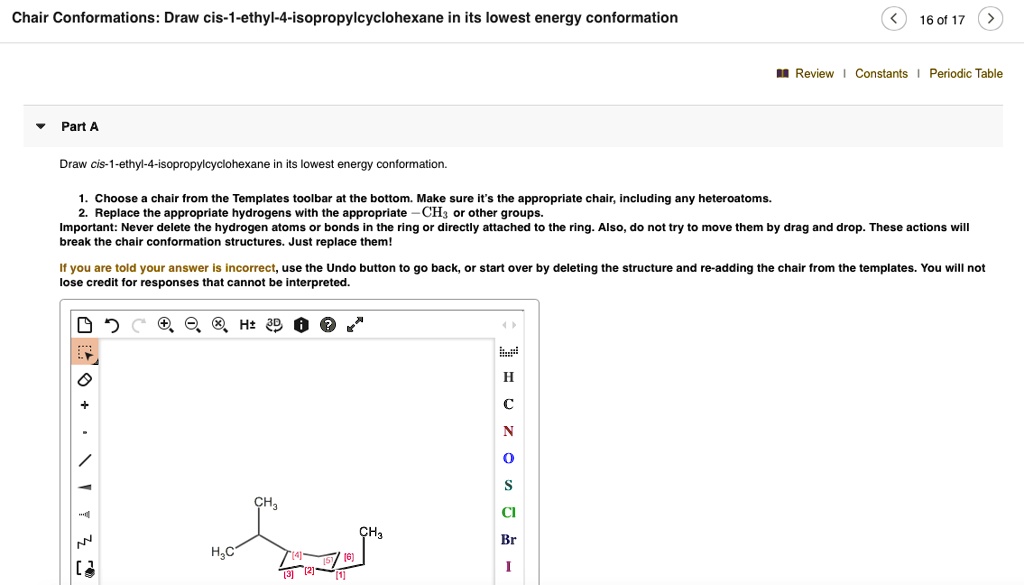
Draw Cis-1-ethyl-4-isopropylcyclohexane In Its Lowest Energy Conformation
You know, I was recently wrestling with a particularly stubborn piece of IKEA furniture. The instructions, bless their minimalist hearts, were more of a suggestion than a guide. I’m pretty sure the diagram for step three was drawn by a squirrel who’d just discovered caffeine. Anyway, after much head-scratching and at least one moment where I considered just supergluing it all together, I finally got it. And then, as I admired my slightly wobbly, yet functional, masterpiece, I realized something. It's all about finding the right way for things to fit together, isn't it? The way that’s most stable, most comfortable, least likely to spontaneously collapse.
And that, my friends, is surprisingly similar to how molecules arrange themselves. Specifically, our humble friend, cis-1-ethyl-4-isopropylcyclohexane. Yeah, I know, mouthful. But stick with me, because understanding how this dude hangs out is actually a pretty cool peek into the secret lives of organic molecules.
The Quest for the Comfiest Seat: Why Molecules Don't Just Flop Around
So, why does a molecule even care about its "conformation"? Isn't it just a bunch of atoms stuck together? Well, not exactly. Think of it like this: you wouldn’t try to read a book while standing on your head, right? It’s possible, but it’s not exactly ideal. You’d prefer a comfy chair, maybe a good lamp. Molecules are a bit like that. They have a bunch of atoms connected by bonds, and those bonds can twist and bend. This twisting and bending creates different shapes, or conformations.
Now, here's the kicker: not all conformations are created equal. Some are like that wobbly IKEA shelf – a bit… tense. They’re holding on by a thread, so to speak, and are just waiting for a slight nudge to go back to a more relaxed state. Others are like a perfectly assembled bookshelf – sturdy, stable, and happy. These are the lowest energy conformations. The molecule’s happy place, if you will. It’s where it’ll spend most of its time because it’s the most energetically favorable.
Enter the Cyclohexane Ring: The Six-Membered Superstar
Our main character, cis-1-ethyl-4-isopropylcyclohexane, has a cyclohexane ring at its core. If you’ve ever encountered cyclohexane before, you might know it’s a bit of a shape-shifter. It’s not flat, oh no. If it were flat, it would have a lot of strain – like trying to bend a piece of metal into a perfect hexagon. It would be fighting itself.
Instead, cyclohexane prefers to adopt a glorious chair conformation. Imagine a comfy armchair. It’s got these nice, relaxed angles. In this chair, the carbon atoms are pretty much arranged in a tetrahedral geometry, which is the sweet spot for carbon bonding. This chair conformation is the key to its stability.
Visualizing the Chair: It's Not as Scary as it Sounds!
Okay, let’s get our hands dirty. To draw this, we need to get familiar with the chair. Picture two parallel lines, and then connect their ends with two more parallel lines, but slightly offset. You’ll end up with something that looks like a cube that’s been squished. Or, as I like to think of it, a somewhat grumpy looking armchair.
Now, in this chair, each carbon atom has two types of positions for its substituents (those are the bits attached to the main ring). We have axial positions and equatorial positions. Axial bonds stick straight up or straight down, parallel to the "axis" of the chair. Equatorial bonds stick out from the "sides" of the ring, roughly in the plane of the ring itself.
Think of the axial positions as standing on the very top or bottom of the chair. The equatorial positions are like sitting on the seat or resting your arm on the armrest – they’re more out to the sides, more spread out.
Our Molecule: Cis-1-ethyl-4-isopropylcyclohexane
Alright, time to introduce our players. We have an ethyl group (that’s a -CH2CH3) and an isopropyl group (-CH(CH3)2). And they’re attached to a cyclohexane ring. The "1" and "4" tell us where they are on the ring. If we number one carbon as "1", then the ethyl group is on that carbon, and the isopropyl group is on the fourth carbon away.
The "cis" part is super important. It means that the ethyl group and the isopropyl group are on the same side of the cyclohexane ring. Imagine the ring is lying flat for a moment (even though it hates doing that!). If the ethyl group is pointing up, the isopropyl group is also pointing up. If the ethyl group is pointing down, the isopropyl group is also pointing down.
Putting it All Together: The Lowest Energy Conformation
Now, we want the lowest energy conformation. Remember what we said about stability? Groups prefer to be in the equatorial position whenever possible. Why? Because axial positions are closer to other axial positions on the same side of the ring. This leads to something called axial-axial repulsion, which is a fancy way of saying the groups bump into each other and get grumpy. Equatorial positions are more spread out, less crowded, and therefore happier.
So, our goal is to place the ethyl and isopropyl groups in equatorial positions if we can. Let's imagine our chair. We have positions 1, 2, 3, 4, 5, and 6. Let’s put the ethyl group on carbon 1.
The Dance of the Substituents: Axial vs. Equatorial
We can draw the ethyl group in either an axial or equatorial position on carbon 1. Likewise for the isopropyl group on carbon 4. So, we have a few options:

Option 1: Both Axial. Ethyl axial, isopropyl axial. This is probably going to be pretty crowded and high in energy. Remember that axial-axial repulsion we talked about? Yeah, this is where it really shines. Or, more accurately, it doesn't shine. This is the grumpy teen of conformations.
Option 2: One Axial, One Equatorial. Ethyl axial, isopropyl equatorial. Or ethyl equatorial, isopropyl axial. Better, but still not ideal. One of our bulky groups is stuck in the less-than-ideal axial spot.
Option 3: Both Equatorial. Ethyl equatorial, isopropyl equatorial. This sounds like our winner, right? Everything is spread out and happy. But there's a catch. We have the "cis" configuration to consider.
The Cis-Trans Conundrum: Same Side, Different Story
Remember "cis"? It means both groups are on the same side of the ring. Let's think about our chair again. Imagine carbon 1. If the ethyl group is equatorial and pointing "up" (relative to the general plane of the ring), then on carbon 4, the isopropyl group must also be on the same side. If the isopropyl group is also equatorial, it will be pointing "down" (relative to the general plane of the ring) if the ethyl is pointing up, or "up" if the ethyl is pointing down. This is because the relative orientation of equatorial bonds at the 1 and 4 positions are opposite.
This is where the magic happens. When we have cis substituents at the 1 and 4 positions of a cyclohexane ring, one of them must be axial if the other is equatorial, and vice versa. It’s a mathematical necessity of the chair conformation and the cis relationship.
The Crucial Flip: Ring Flip and Energy Minima
So, if we have cis-1,4-disubstituted cyclohexane, and we try to put both groups equatorial, we'll find it's impossible while maintaining the cis relationship and the chair conformation. This is where the molecule has to make a choice.
Let's draw our chair. Let's say we place the ethyl group in an equatorial position on carbon 1, pointing "up". Because it’s cis, the isopropyl group on carbon 4 must also be on the "up" side. Now, the equatorial position on carbon 4 points "down" relative to the ring plane. So, if we want the isopropyl to be equatorial, it would be pointing "down". This contradicts the "cis" requirement if our ethyl is pointing "up".
Therefore, in the cis isomer, if one substituent is equatorial, the other must be axial to maintain the cis relationship. And the opposite is also true: if one is axial, the other is equatorial. They can't both be axial (too crowded) and they can't both be equatorial (doesn't fit the cis rule in the chair conformation).
This means for cis-1-ethyl-4-isopropylcyclohexane, we have two possible low-energy conformations (because the other options, like both axial, are much higher energy):
- Ethyl is equatorial, isopropyl is axial.
- Ethyl is axial, isopropyl is equatorial.
Now, which one is the lowest energy? We need to consider the size of our groups. The isopropyl group is bigger and bulkier than the ethyl group. Bigger groups create more of that dreaded axial-axial repulsion when they’re in the axial position. So, the conformation where the larger group (isopropyl) is equatorial and the smaller group (ethyl) is axial will be the most stable. It minimizes the steric hindrance.
Drawing it Out: The Moment of Truth
Okay, deep breaths. Let’s draw this. We’ll start with our chair conformation. I’m going to draw it so the front bottom carbon is pointing towards me, and the back top carbon is pointing away. It’s a classic way to draw it.
First, let's draw the cyclohexane ring. Two downward-pointing bonds at the bottom, two upward-pointing bonds at the top, connected by two bonds going back and up, and two going back and down. Like a squished, angular ‘M’ if you tilt your head right.

Now, let’s label our carbons 1 through 6, starting from a bottom carbon and going clockwise. So, bottom right is 1, top right is 2, top middle is 3, top left is 4, bottom left is 5, and bottom middle is 6.
We know the isopropyl group is larger and wants to be equatorial. So, on carbon 1, we’ll draw the ethyl group in the axial position, pointing straight down. On carbon 4, we’ll draw the isopropyl group in the equatorial position, sticking out to the right and slightly down (relative to the ring). Make sure they are on the same side of the ring in your mind's eye – if you imagine the ring flattened for a second, they'd both be sticking up, or both sticking down. In our chair drawing, this means if the ethyl axial is pointing down, the isopropyl equatorial will also be on that same general "down" side of the ring.
The other option, where the ethyl is equatorial and the isopropyl is axial, would have higher energy because the bulkier isopropyl group is experiencing more axial-axial repulsion.
So, the lowest energy conformation for cis-1-ethyl-4-isopropylcyclohexane has the ethyl group in an axial position and the isopropyl group in an equatorial position. This is because the bulkier isopropyl group is better off in the less hindered equatorial position, even if it means the smaller ethyl group has to take the axial spot.
It’s like having to put the awkwardly shaped luggage in the trunk and the precious, but smaller, vase on the passenger seat. You optimize for the most problematic item. Molecules do the same thing, just with invisible bumps and electron clouds!
So there you have it. The seemingly complex cis-1-ethyl-4-isopropylcyclohexane, in its most relaxed, happiest, lowest-energy conformation. It’s all about finding that comfy chair and making sure the biggest guests get the most space. A valuable lesson, both in chemistry and in life, don't you think? Now, if you'll excuse me, I have a slightly wobbly bookshelf to admire.
