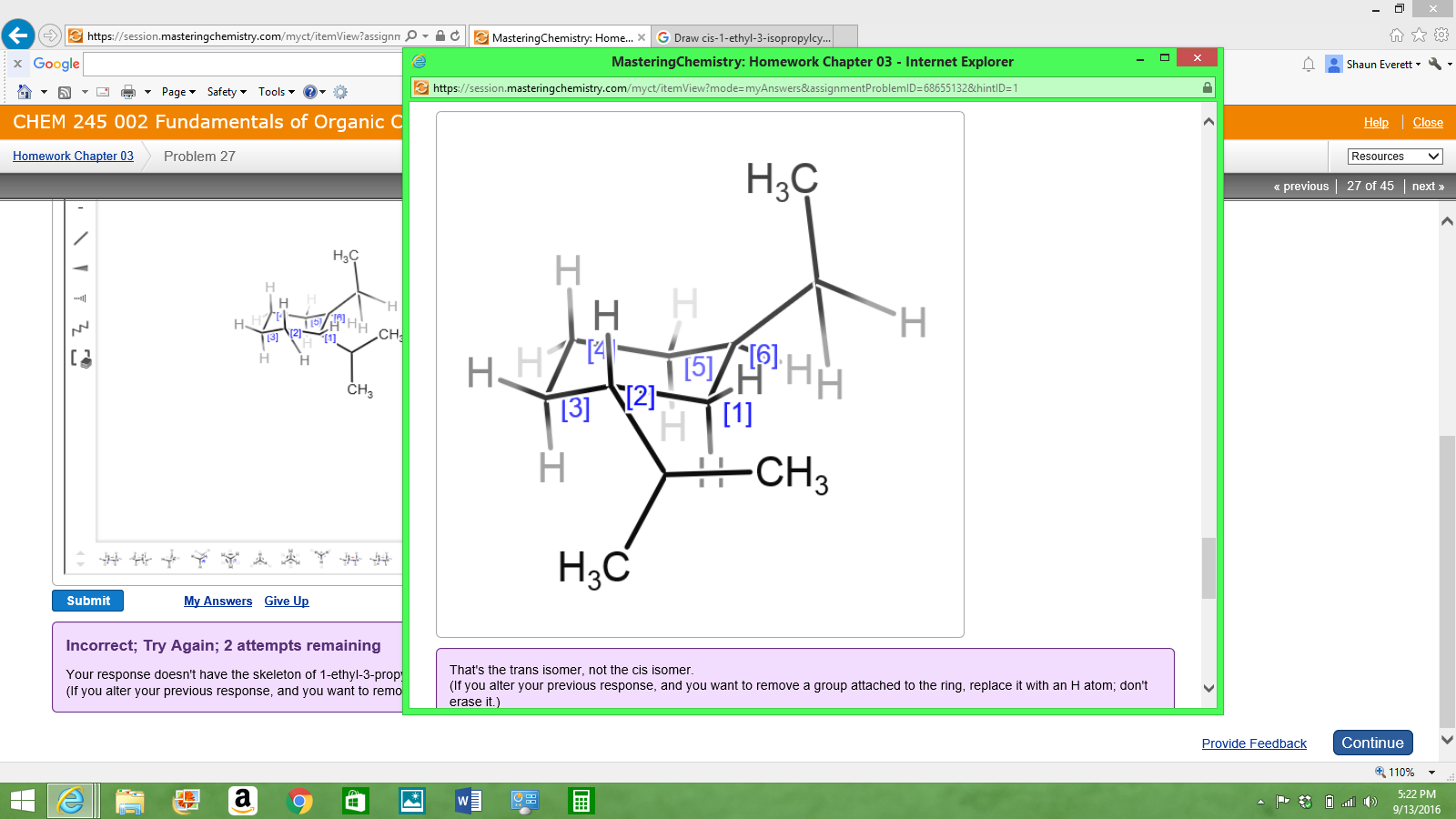
Draw Cis-1-ethyl-3-isopropylcyclohexane In Its Lowest Energy Conformation

Hey there, fellow molecule wranglers! Today, we’re diving into a bit of organic chemistry fun that’s surprisingly chill once you get the hang of it. We’re going to tackle drawing cis-1-ethyl-3-isopropylcyclohexane in its lowest energy conformation. Sounds like a mouthful, right? Don’t worry, we’ll break it down like a delicious cookie. Think of it as building with molecular LEGOs, but way cooler and with a lot less choking hazards (hopefully).
First off, let’s unpack that name, shall we? Cyclohexane is our basic building block. Imagine a six-membered ring of carbon atoms, kind of like a perfectly formed hexagon, but with those carbons all connected in a circle. It’s pretty stable, but it loves to twist and turn to find its happy place, which we call its lowest energy conformation. Usually, this happy place is a chair conformation. Think of a comfy armchair – that’s the vibe.
Now, let’s talk about our fancy little decorations on this ring: ethyl and isopropyl groups. An ethyl group is just two carbons hooked together (CH₂CH₃). Think of it as a tiny little arm waving from our ring. The isopropyl group is a bit bulkier – it’s three carbons arranged in a Y-shape (CH(CH₃)₂). This one’s like a slightly more ambitious, three-pronged arm.
The “1-ethyl-3-isopropyl” part tells us where these arms are attached to our cyclohexane ring. We number the carbons in the ring, starting from one of the attachment points. So, we’ll have an ethyl group on carbon number 1, and an isopropyl group on carbon number 3. Easy peasy, right? You can pick any carbon to start your numbering, but stick with it!
The real kicker is the “cis” part. This is where things get a little more spatial. In a cyclohexane ring, carbons have two types of positions: axial and equatorial. Axial bonds point straight up or straight down, like the legs of a chair. Equatorial bonds point out to the sides, more or less horizontally, like the arms of a chair. When we say “cis,” it means that our ethyl and isopropyl groups are on the same side of the ring. Imagine the ring is lying flat on a table for a sec (even though it’s never really flat, but humor me). If the ethyl group is pointing up from the table, then the isopropyl group must also be pointing up from the table. Or, if ethyl is down, isopropyl must also be down.
Let's Get Our Chair On!
Okay, so the chair conformation is where our molecule wants to chill. Why? Because it minimizes steric strain. This is fancy talk for “things bumping into each other.” When our groups are too close for comfort, the molecule gets a bit grumpy and uses up extra energy. The chair conformation spreads things out nicely, like people sitting comfortably in a circle instead of crammed into a tiny box.

To draw a chair, start with a slightly squashed hexagon. Imagine one side of the hexagon is higher than the other, and then you have a valley. Or, think of drawing a capital ‘M’ and then connecting the ends. It’s a bit of an art form, honestly. Once you have your basic chair shape, you need to add those axial and equatorial bonds. For each carbon in the ring, there’s one axial bond pointing up and one equatorial bond pointing sideways, and then the other set points down and sideways respectively. It’s like each carbon has two potential parking spots for its substituents (our ethyl and isopropyl friends).
Now for the crucial part: the cis relationship. Since we have substituents on carbons 1 and 3, let’s number them. Pick a carbon for C1. Let’s imagine it’s one of the carbons forming the “seat” of our chair. Now, move along the ring to C3. You can go either clockwise or counterclockwise, just be consistent!
Let’s say we choose C1 and move clockwise to C3. On C1, we need to attach our ethyl group. We have an axial and an equatorial option. On C3, we’ll attach our isopropyl group, also with axial and equatorial options. The “cis” rule means both groups must be on the same side of the ring. If we define “up” on C1 as axial, then “up” on C3 is also axial. If we define “up” on C1 as equatorial, then “up” on C3 is also equatorial.
So, we have two main possibilities for placing our groups in a cis arrangement: both axial or both equatorial. Here’s where the “lowest energy” part comes in. Remember that bulky isopropyl group? It’s like the big uncle at Thanksgiving dinner – it takes up a lot of space! When groups are in axial positions, they can get a bit too cozy with other axial bonds on the same side of the ring, leading to 1,3-diaxial interactions. These are like awkward social encounters where people are standing too close and bumping elbows. It’s not ideal for energy.
The Energy Showdown!
Generally, equatorial positions are preferred for bulky substituents because they stick out further from the ring and avoid those pesky 1,3-diaxial interactions. It’s like giving your guests more elbow room!
So, let’s draw our chair. Let’s start by drawing C1 pointing up. If C1 is pointing up, its axial bond points up, and its equatorial bond points down and out. Now, let’s go to C3. If we number clockwise, C3 will also be pointing up. Its axial bond will point up, and its equatorial bond will point down and out.
Now, for the cis relationship. If we put the ethyl group on C1, and we want it to be cis to the isopropyl group on C3, they need to be on the same side. Let’s try putting the ethyl group in the equatorial position on C1. This is the preferred spot because it’s less crowded.
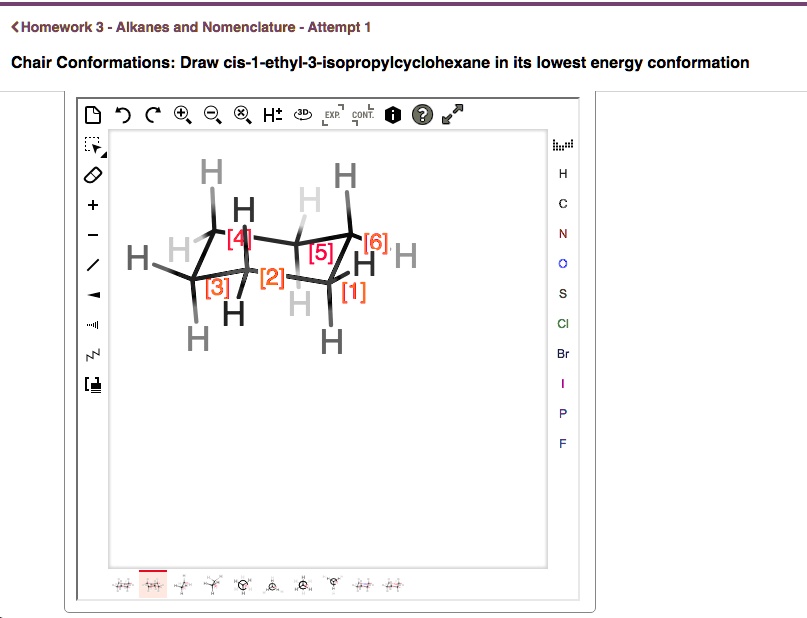
Now, for C3, we need to attach the isopropyl group. Since it’s cis to the ethyl group, it also needs to be on the same side of the ring. If our ethyl is equatorial on C1, and we want them both on the same side, we’d also want the isopropyl group to be in its equatorial position on C3. This means both groups will be pointing generally “outward” from the ring in a similar direction.
Let’s visualize this. Imagine our chair is sitting on a table. If the ethyl group is sticking out and slightly up (equatorial), the isopropyl group will also be sticking out and slightly up (equatorial) on C3. This arrangement minimizes steric strain because these bulky groups are as far away from each other and from the ring’s axial bonds as possible. This is our lowest energy conformation!
What if we tried the other cis arrangement: both axial? If we put the ethyl group axial-up on C1, and the isopropyl group axial-up on C3, they’d be pretty close to each other, and also close to the axial hydrogens on other carbons. This would create more steric strain. Similarly, if they were both axial-down, they’d still have issues. So, the equatorial-equatorial arrangement for cis is definitely the winner.
Drawing Steps (The Fun Part!)
Okay, let’s actually draw it! Grab a pencil and paper, or your favorite drawing app.
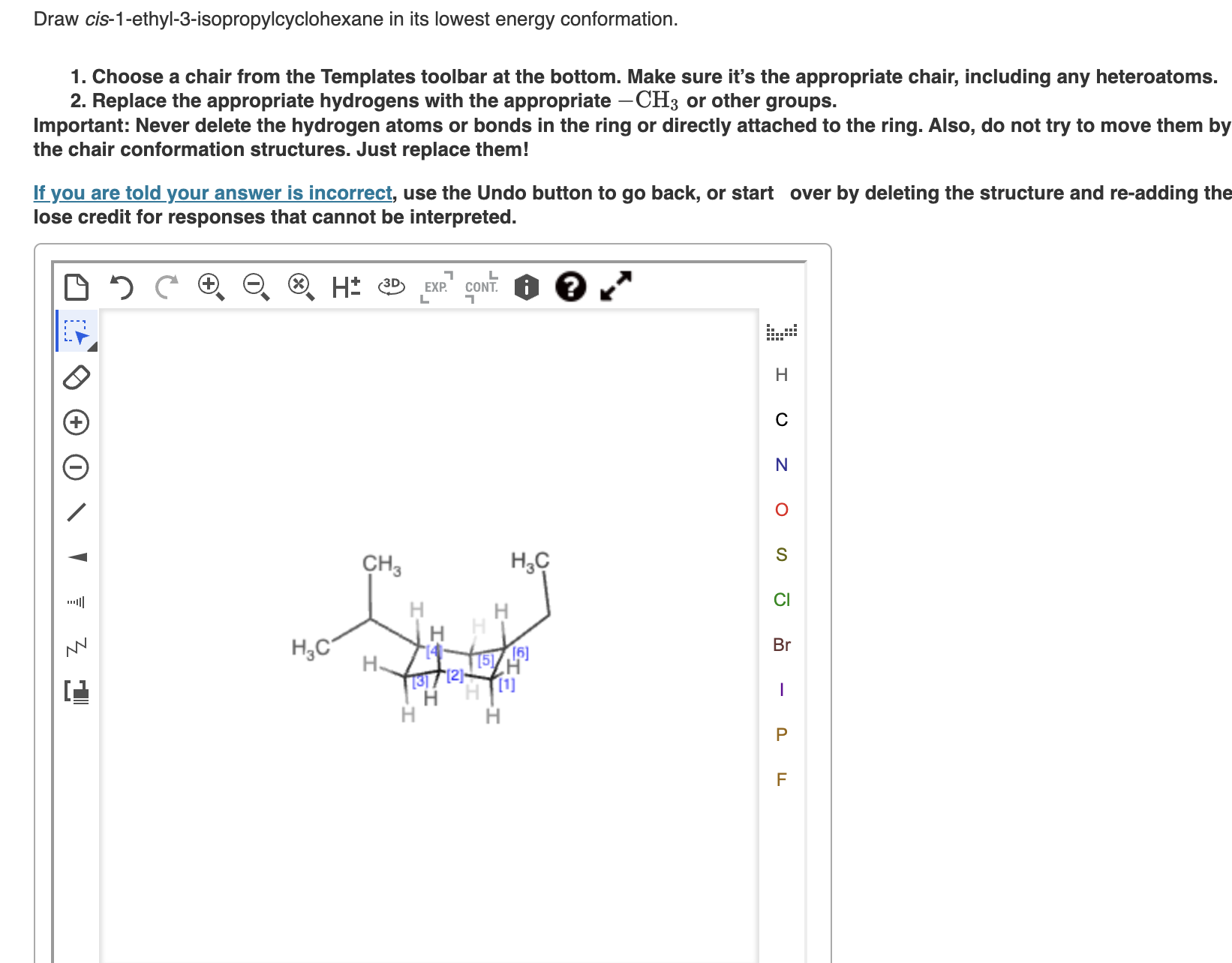
- Draw the chair conformation. Start with that slightly tilted hexagon. Imagine one carbon is a bit higher, then two carbons go down, then one carbon is lower, then two carbons go up. It looks like a comfy chair!
- Number your carbons. Pick any carbon and call it C1. Number around the ring (clockwise or counterclockwise, your choice!) until you get to C3.
- Place the ethyl group. Since we want the lowest energy conformation for a cis relationship, we’ll put the ethyl group in the equatorial position on C1. Remember, equatorial bonds are the ones that stick out to the sides. If C1 is pointing generally upwards, the equatorial bond will be pointing outwards and slightly downwards.
- Place the isopropyl group. Now, go to C3. For the cis relationship, and to keep it low energy, we’ll also put the isopropyl group in the equatorial position on C3. Crucially, it needs to be on the same side as the ethyl group. So, if your ethyl group is pointing out and slightly down from C1, your isopropyl group on C3 should also be pointing out and slightly down.
- Add the hydrogens. Don’t forget the little hydrogen atoms! Each carbon has one hydrogen remaining. These will occupy the axial positions if your substituents are equatorial, and vice-versa. Since our ethyl and isopropyl are equatorial, the hydrogens on C1 and C3 will be axial.
It’s a bit of a puzzle, and the first few times might feel like you’re doing brain surgery with chopsticks. But once you get the hang of the chair and the axial/equatorial dance, it becomes second nature. Think of it as a secret handshake for molecules!
A quick check: Are the ethyl and isopropyl groups on the same side of the ring? Yes, because both are in equatorial positions that point roughly in the same direction relative to the plane of the ring. Is it the lowest energy conformation? Yes, because bulky groups are in equatorial positions, minimizing steric clashes. Hooray!
So, there you have it! Cis-1-ethyl-3-isopropylcyclohexane chilling in its favorite, most relaxed pose. It’s like the molecule wearing its comfiest pajamas. And honestly, that’s what organic chemistry is all about – understanding how molecules arrange themselves to be as happy and stable as possible. It’s a beautiful dance of atoms, and you just learned a few steps!
Don’t be discouraged if your drawings aren’t perfect on the first try. Every chemist has been there, squinting at their diagrams and muttering about axial hydrogens. The important thing is to understand the concepts: the chair, axial vs. equatorial, and how “cis” affects the placement. With a little practice, you’ll be drawing these conformations like a pro. And who knows, maybe you’ll start seeing chair conformations in everyday objects. That park bench? Total chair conformation. That comfy armchair? Definitely a low-energy conformation. Keep exploring, keep drawing, and keep that molecular curiosity burning bright!
