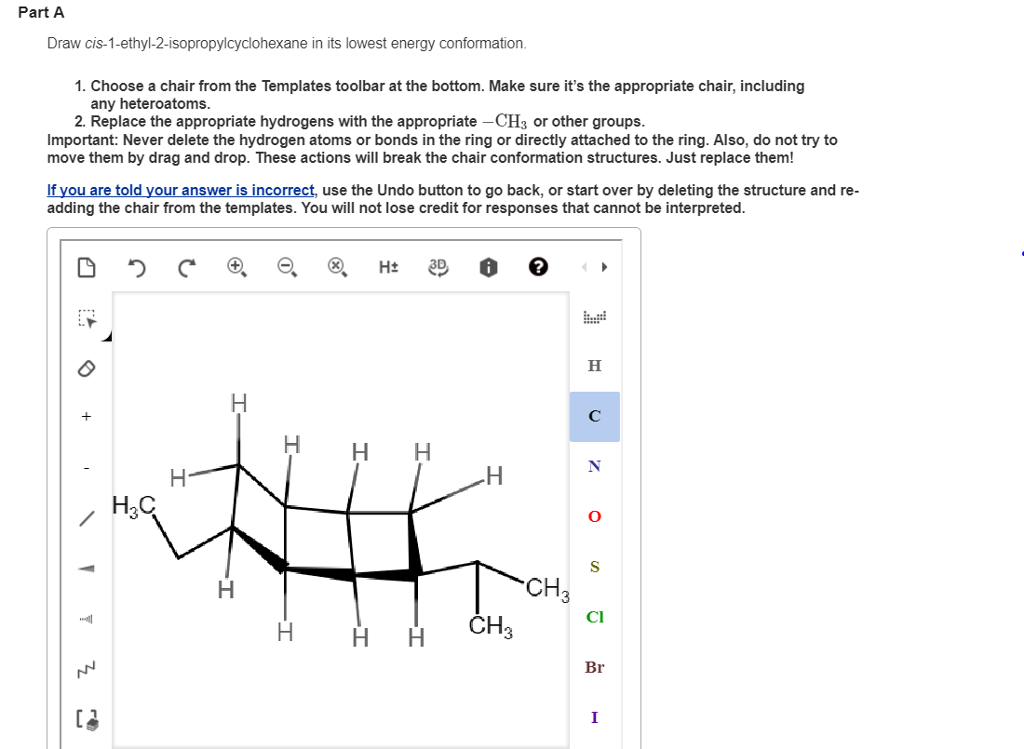
Draw Cis-1-ethyl-2-isopropylcyclohexane In Its Lowest Energy Conformation
Ever wondered what makes some molecules “happier” or more stable than others? It’s like finding the comfiest spot on the couch – there’s usually one position that just feels right. Today, we’re diving into the wonderfully wiggly world of cyclohexane conformations and exploring the lowest energy conformation for a specific molecule: cis-1-ethyl-2-isopropylcyclohexane. It might sound a bit science-y, but think of it as solving a fun, 3D puzzle! Understanding these shapes helps us unlock the secrets of how molecules behave, which is pretty cool whether you’re a budding chemist, a curious student, or just someone who enjoys a good brain teaser.
For absolute beginners, this is a fantastic way to start visualizing molecules beyond flat drawings. It’s like learning to see in 3D! You’ll begin to appreciate that molecules aren’t static drawings but dynamic structures that twist and turn. Families can turn this into a playful activity, perhaps using pipe cleaners or LEGOs to build different cyclohexane shapes and discover which arrangement is the most stable. It’s a hands-on approach to learning that can make abstract concepts relatable and engaging for everyone. For hobbyists, particularly those interested in organic chemistry, molecular modeling, or even understanding how things like medicines work at a fundamental level, this knowledge is incredibly useful. It’s the foundation for understanding reactivity and properties of countless organic compounds.
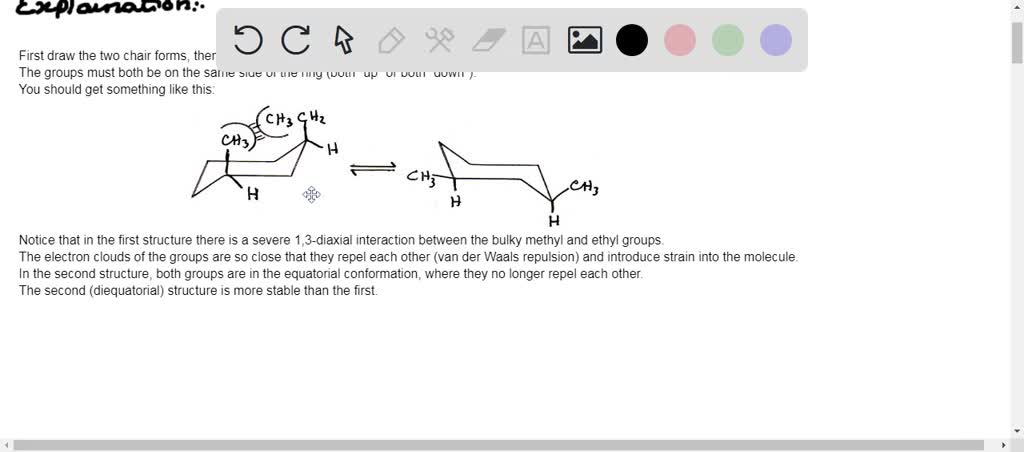
So, what exactly is the lowest energy conformation for cis-1-ethyl-2-isopropylcyclohexane? Cyclohexane itself loves to be in a chair conformation, which is the most stable shape, minimizing strain. Now, we’re adding two bulky groups: an ethyl (-CH2CH3) and an isopropyl (-CH(CH3)2). The "cis" part tells us these groups are on the same side of the cyclohexane ring. When placing these groups, we have to consider where they’ll be: either pointing “up” (axial) or “out” (equatorial) relative to the ring. Bulky groups much prefer to be in the equatorial position because it’s less crowded. Imagine trying to fit two big cushions into a small chair – you want them to lie flat rather than stick straight up!
To find the lowest energy conformation, we essentially try placing our ethyl and isopropyl groups in different combinations of axial and equatorial positions. Since they are cis, one will be up and one will be down on the ring. However, because they are on adjacent carbons, the most stable arrangement will have both groups in equatorial positions. This might seem counterintuitive with the "cis" label, but the cyclohexane ring can flip between its two chair forms. In one of these flipped forms, both bulky groups can indeed end up in the more stable equatorial positions, minimizing steric hindrance (that’s just a fancy word for crowding!).
Getting started is simpler than you think! Grab some modeling clay or even different colored play-doh. First, make a hexagonal ring for your cyclohexane. Then, attach your ethyl and isopropyl groups. Try putting them in different spots. You’ll quickly see how some arrangements make the molecule look squished and uncomfortable, while others allow it to stretch out more naturally. There are also plenty of online molecular modeling tools and diagrams that can help you visualize this. Just search for “cyclohexane chair conformation” and then look for examples with substituents!
Exploring molecular shapes is a journey that can lead to a deeper appreciation for the intricate beauty of chemistry. Discovering the most comfortable shape for a molecule like cis-1-ethyl-2-isopropylcyclohexane is not just an academic exercise; it’s a satisfying way to connect with the invisible world around us. It’s a little bit of art, a little bit of science, and a whole lot of fun!
