Draw All Stereoisomers Of 1 Bromo 2 Chlorocyclohexane

Ever found yourself staring at a chemical structure and wondering, "Is that all there is to it?" Well, get ready for a little chemical adventure! Today, we're diving into the fascinating world of stereoisomers, specifically for a molecule called 1-bromo-2-chlorocyclohexane. Now, you might be thinking, "Chemistry? Fun?" And we get it! But trust us, understanding stereoisomers is like unlocking a secret level in a video game. It's not just about memorizing structures; it's about understanding how molecules can be the same in terms of their atoms but totally different in how they are arranged in 3D space. This seemingly small difference can have huge implications, from how medicines work to how flavors are perceived. So, buckle up, grab your imaginary molecular building blocks, and let's explore the wonderful world of 1-bromo-2-chlorocyclohexane!
The Magic of Molecular Arrangement
So, what's the big deal with stereoisomers? Imagine you have a pair of gloves. They're made of the same material, have the same number of stitches, and are designed for the same purpose. But you can't wear the left glove on your right hand, can you? That's because they are stereoisomers – they are mirror images of each other and cannot be perfectly superimposed. In chemistry, molecules with the same molecular formula can exist as different stereoisomers. This happens when atoms are connected in the same order but are arranged differently in three-dimensional space. For 1-bromo-2-chlorocyclohexane, we're dealing with a cyclohexane ring, which is a six-membered ring of carbon atoms. On this ring, we have a bromine atom attached to one carbon and a chlorine atom attached to an adjacent carbon.
The "fun" part comes in when we realize that these seemingly identical molecules can behave very differently. Think about it: a drug molecule might bind perfectly to a receptor in your body as one stereoisomer, but its mirror image might be completely inactive or, worse, cause harmful side effects. It’s like trying to fit a left-handed screw into a right-handed hole – it just won't work. This is why being able to draw and understand all the possible stereoisomers is a crucial skill in chemistry, especially in fields like pharmacology, biochemistry, and materials science.
Understanding stereoisomers is like learning to read the secret language of molecules. It's the difference between just seeing a shape and truly understanding its function.
Unlocking the Possibilities: Drawing 1-Bromo-2-chlorocyclohexane
Let's get down to the business of drawing. For 1-bromo-2-chlorocyclohexane, we need to consider the chirality of the molecule. Chirality essentially means "handedness," and in chemistry, it refers to molecules that are non-superimposable mirror images of each other (just like our hands!). In 1-bromo-2-chlorocyclohexane, the carbons where the bromine and chlorine are attached are usually chiral centers, meaning they have four different groups attached to them. This is where the possibilities start to branch out.
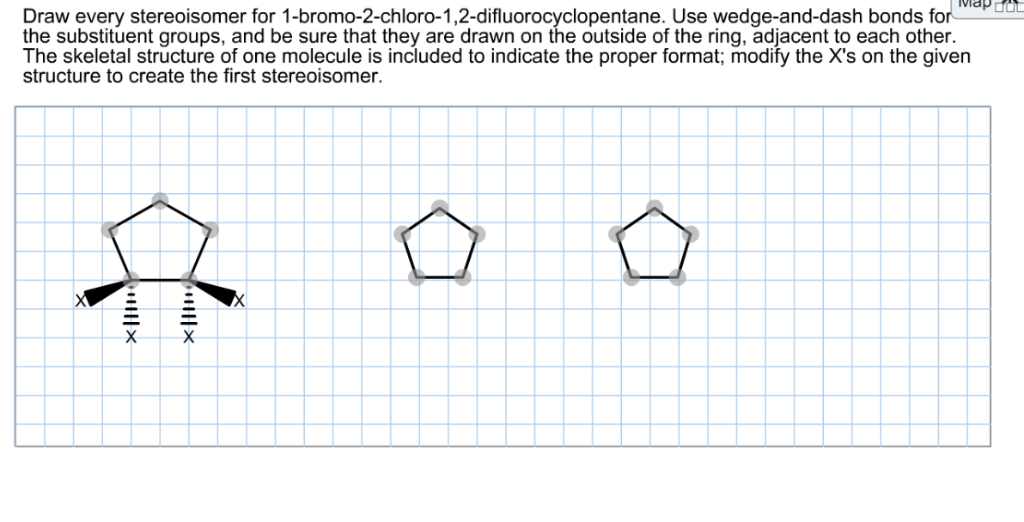
First, we need to think about the cyclohexane ring itself. Cyclohexane rings aren't flat; they exist in a 3D shape called a "chair conformation." In this chair conformation, the substituents (like bromine and chlorine) can be either pointing "up" (axial) or "outwards" (equatorial) relative to the ring. This adds another layer to the puzzle!
When we place the bromine and chlorine atoms on adjacent carbons, we have to consider their relative positions: are they on the same side of the ring (cis) or on opposite sides (trans)?
- Cis Isomers: For the cis isomers, both the bromine and chlorine atoms can be pointing "up" relative to the ring plane, or both can be pointing "down." These are two distinct molecules. Imagine looking at the ring from the side; both substituents are on the same side of the imaginary plane of the ring.
- Trans Isomers: For the trans isomers, one substituent will be pointing "up" and the other will be pointing "down." Again, there are two ways this can happen: bromine up and chlorine down, or bromine down and chlorine up. These are also distinct molecules.
But wait, there's more! Each of these cis and trans arrangements can also exist as a pair of mirror images. This is where the concept of enantiomers comes in. Enantiomers are stereoisomers that are non-superimposable mirror images of each other. So, for our cis isomers, we'll have one pair of enantiomers. Similarly, for our trans isomers, we'll have another pair of enantiomers.
Let's break it down into what we're drawing:
- Cis Isomer 1: Both Br and Cl pointing up (or both down). This molecule has a mirror image that is not superimposable. So, we have two enantiomers here.
- Trans Isomer 1: Br pointing up, Cl pointing down (or vice-versa). Again, this molecule has a mirror image that is not superimposable, leading to two more enantiomers.
So, in total, we can draw four distinct stereoisomers for 1-bromo-2-chlorocyclohexane. These are often depicted using wedge-and-dash notation or by drawing them in their chair conformations, clearly showing the spatial arrangement of the bromine and chlorine atoms. Each drawing represents a unique molecule with potentially unique chemical and physical properties. It's a beautiful demonstration of how subtle differences in molecular architecture can lead to vastly different realities!
