Draw All Stereoisomers Of 1 2 Dichlorocyclohexane

Alright folks, gather ‘round! Ever had one of those days where you feel like you’re seeing double… or maybe even triple? That’s kind of what we’re diving into today, but with molecules. Yep, those tiny building blocks of everything around us. We’re talking about 1,2-dichlorocyclohexane, which sounds super fancy, like something you’d order at a molecular mixology bar. But really, it’s just a six-carbon ring with two chlorine atoms hanging out on adjacent carbons. Easy peasy, right?
Now, the fun part. Imagine you’ve got two friends, let’s call them Chris and Carol, who are identical twins. They look exactly alike, but sometimes, Carol likes to wear her favorite hat slightly tilted to the left, while Chris prefers it perfectly straight. Or maybe one of them always walks with a slight swagger. Even though they’re twins, there are these little differences, right? That’s the essence of stereoisomers. They’re like molecular twins – same atoms, same connections, but they can be arranged in space in slightly different ways. And these differences, however small, can sometimes lead to big changes in how they behave, like how one twin might be allergic to peanuts and the other isn't.
So, 1,2-dichlorocyclohexane. We’ve got our cyclohexane ring, which is like a little hexagonal bicycle frame. And then we’ve got two chlorine atoms, our “dichloros,” attached to two neighboring carbons. That “1,2” is just telling us they’re buddies, right next door to each other on the ring. Now, here’s where it gets interesting. These chlorine atoms can do a little dance. They can either be on the same side of the ring, like two besties giving each other a high-five, or they can be on opposite sides, like they’re politely nodding from across a crowded room.
Let’s break down the possibilities. Think about that cyclohexane ring. It’s not flat like a pancake, oh no. It’s got a bit of a bend to it, kind of like a deck chair. This shape is super important. When we talk about the chlorine atoms, we’re talking about their positions relative to this bending ring. Are they sticking up like little antennas, or are they sticking down like little dangling earrings?
First up, let’s consider when our two chlorine atoms are on the same side of the ring. We call this the cis isomer. Imagine you’re looking down at that bicycle frame, and both chlorine atoms are poking out on the same side. It’s like having two identical garden gnomes placed on the same side of your porch. They’re both there, doing their gnome thing, in the same general direction. Now, this cis arrangement can exist in two forms, and this is where it gets a little trippy. Because the cyclohexane ring can flip and flop, these two cis forms are actually like mirror images of each other. They’re enantiomers! Think of your hands. Your left hand and your right hand are mirror images. They’re the same, but you can’t perfectly superimpose them, right? You can’t put your left glove on your right hand and have it fit perfectly. That’s exactly what’s happening with these cis 1,2-dichlorocyclohexane molecules. They are non-superimposable mirror images of each other. We call these (1R,2R)-1,2-dichlorocyclohexane and (1S,2S)-1,2-dichlorocyclohexane. It’s like having identical twins who are left-handed and right-handed versions of themselves. Pretty wild, huh?
These two cis forms are special. They’re like a pair of magic socks – they’re identical in every way except for their "handedness." If you held one up to a mirror, you’d see the other. And you can’t stack them perfectly on top of each other. It's this subtle spatial difference that can make them interact differently with other molecules, especially other chiral molecules (molecules with that same "handedness" property). It’s like how a left-handed screwdriver won’t work perfectly in a right-handed screw. Or how your left shoe fits your left foot, not your right.
Now, let’s switch gears. What happens when our two chlorine atoms decide to be on opposite sides of the ring? We call this the trans isomer. So, one chlorine is sticking up, and the other is sticking down. They’re like two balloons tied to opposite corners of a kite. They’re definitely separated. Think of our identical twins again. This time, one twin might decide to stand on their tiptoes, while the other is perfectly grounded. Or one might be wearing a tie and the other a bowtie – same general idea of neckwear, but different presentation. Again, the cyclohexane ring has its own chair-like conformation, meaning the chlorines can be in an "axial" (up or down along the ring's axis) or "equatorial" (sticking out from the sides) position. For the trans isomer, you could have one chlorine axial and the other equatorial, or both axial but in opposite directions, or both equatorial but again, in opposite directions. It gets a bit more involved when you consider the ring flipping, but the key is that the two chlorines are on opposite sides.
Here’s the kicker about the trans isomer: it’s different from the cis isomer in a very fundamental way. Unlike the cis form, the trans 1,2-dichlorocyclohexane is achiral. What does achiral mean? It means it’s not like your hands. It’s like a simple, symmetrical ball. If you put it in a mirror, the reflection is exactly the same as the original. You can perfectly superimpose the mirror image onto the original. It’s like looking at a perfectly round dinner plate in a mirror – it’s still a dinner plate, and you can’t tell the difference. So, there’s only one trans isomer of 1,2-dichlorocyclohexane. No handedness, no mirror-image twins to worry about here. It’s just… trans.
Why is this whole stereoisomer thing even a thing? Well, imagine you’re baking cookies. You’ve got flour, sugar, eggs, and chocolate chips. You can mix them all up in a bowl and bake them, and you get cookies. But what if you arranged those ingredients in a very specific way before baking? That’s kind of what stereochemistry is about. The same ingredients (atoms) are there, but their arrangement in three-dimensional space can totally change the outcome. In the case of 1,2-dichlorocyclohexane, the cis isomers, with their "handedness," might react differently with other chiral molecules, like enzymes in your body. One might fit perfectly into a receptor site like a key in a lock, while its mirror image might be a terrible fit, or even bind to a different receptor altogether.
Think about how some medicines work. A drug molecule has a specific 3D shape. If a drug has stereoisomers, one isomer might be super effective at treating an illness, while the other isomer might be inactive, or worse, cause unwanted side effects. It’s like having two keys that look almost identical. One key opens your front door, but the other, with a slight difference in its bitting, might open your neighbor’s door (and that could be problematic!). So, understanding stereoisomers is super important in fields like pharmaceuticals and biochemistry. We need to know if we’re making the right "key" for the job.
Let’s recap the players in our little stereoisomer party for 1,2-dichlorocyclohexane:
The Cis Crew: Mirror Images Galore!
We’ve got two members in this crew, and they’re inseparable, like a matched pair of socks that somehow always go missing together. They are enantiomers, meaning they are non-superimposable mirror images of each other.
- (1R,2R)-1,2-dichlorocyclohexane: This is one of our “handed” twins. Imagine it with the two chlorines pointing up (or down, depending on how you look at the ring), but with a specific arrangement that gives it a particular "chirality."
- (1S,2S)-1,2-dichlorocyclohexane: This is the mirror image of our first friend. It’s got the same structure, same atoms, same connections, but its 3D orientation is flipped, like looking at it in a mirror. They are absolutely distinct molecules, even though they look so alike!
These two are like identical twins who have mastered the art of looking different just enough to confuse you. They have the same properties in most situations, except when they encounter other chiral things. It’s like they have a secret handshake that only certain molecules understand.
The Trans Titan: The Lone Wolf
This one is a bit of a different character. It’s not playing the mirror-image game with anyone.
- (1R,2S)-1,2-dichlorocyclohexane (or (1S,2R)-1,2-dichlorocyclohexane): Now, this is where it gets a little neat. Because of the symmetry within the trans arrangement and how the cyclohexane ring can flip, the trans isomer is actually what we call meso. Wait, what’s meso? It's like a molecule that looks like it should have mirror images, but because of an internal symmetry, its mirror image is identical to itself. It's like a palindrome – "madam" reads the same forwards and backward. The trans isomer, despite having two chiral centers (the carbons with the chlorine atoms), possesses an internal plane of symmetry that makes it achiral. It’s like a perfectly symmetrical snowflake; even though it has intricate details, it can be cut in half perfectly symmetrically. So, there’s only one trans form. It’s not a pair of twins; it’s a singular entity.
So, when we talk about drawing all the stereoisomers of 1,2-dichlorocyclohexane, we’re really talking about identifying these distinct spatial arrangements. We’ve got our cis pair (the enantiomers) and our trans single (the achiral, or meso, compound). That makes a total of three stereoisomers!
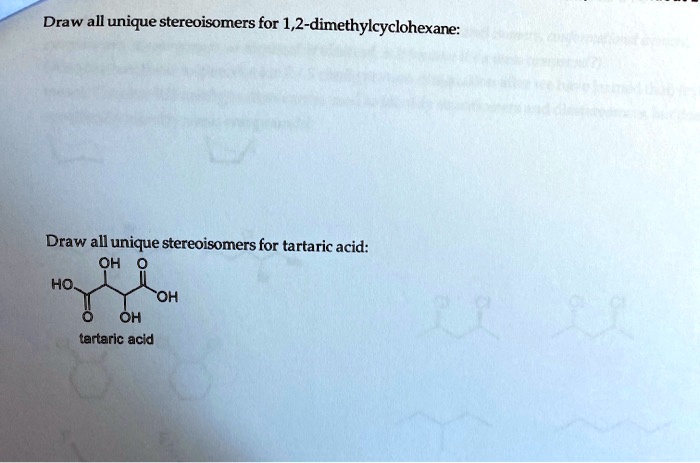
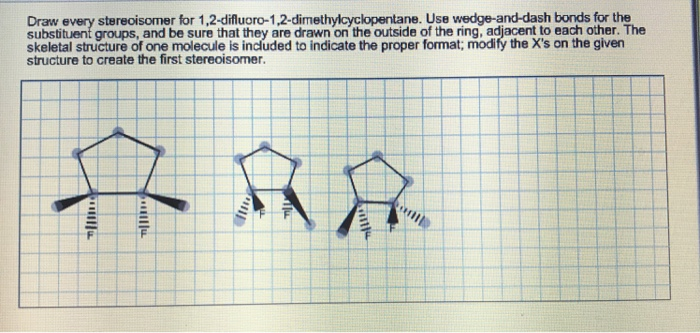
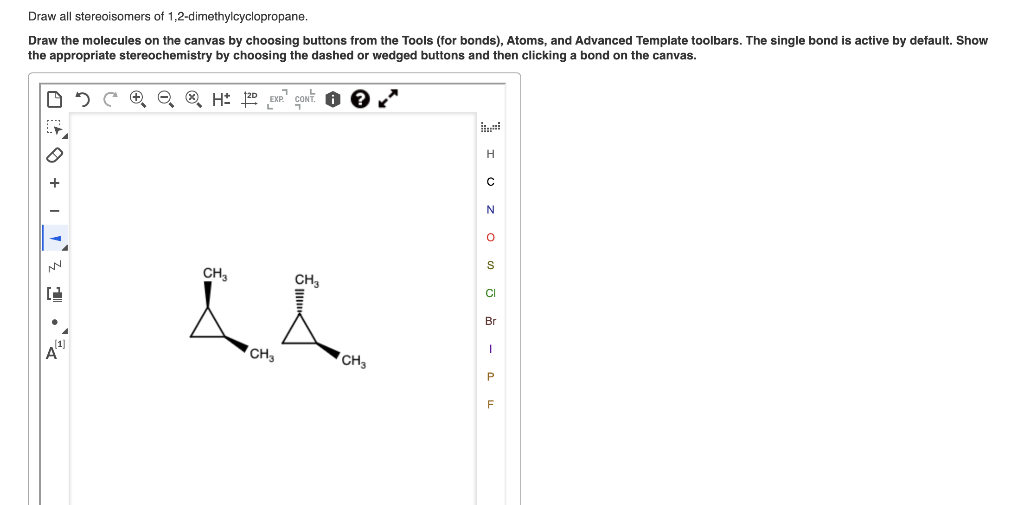
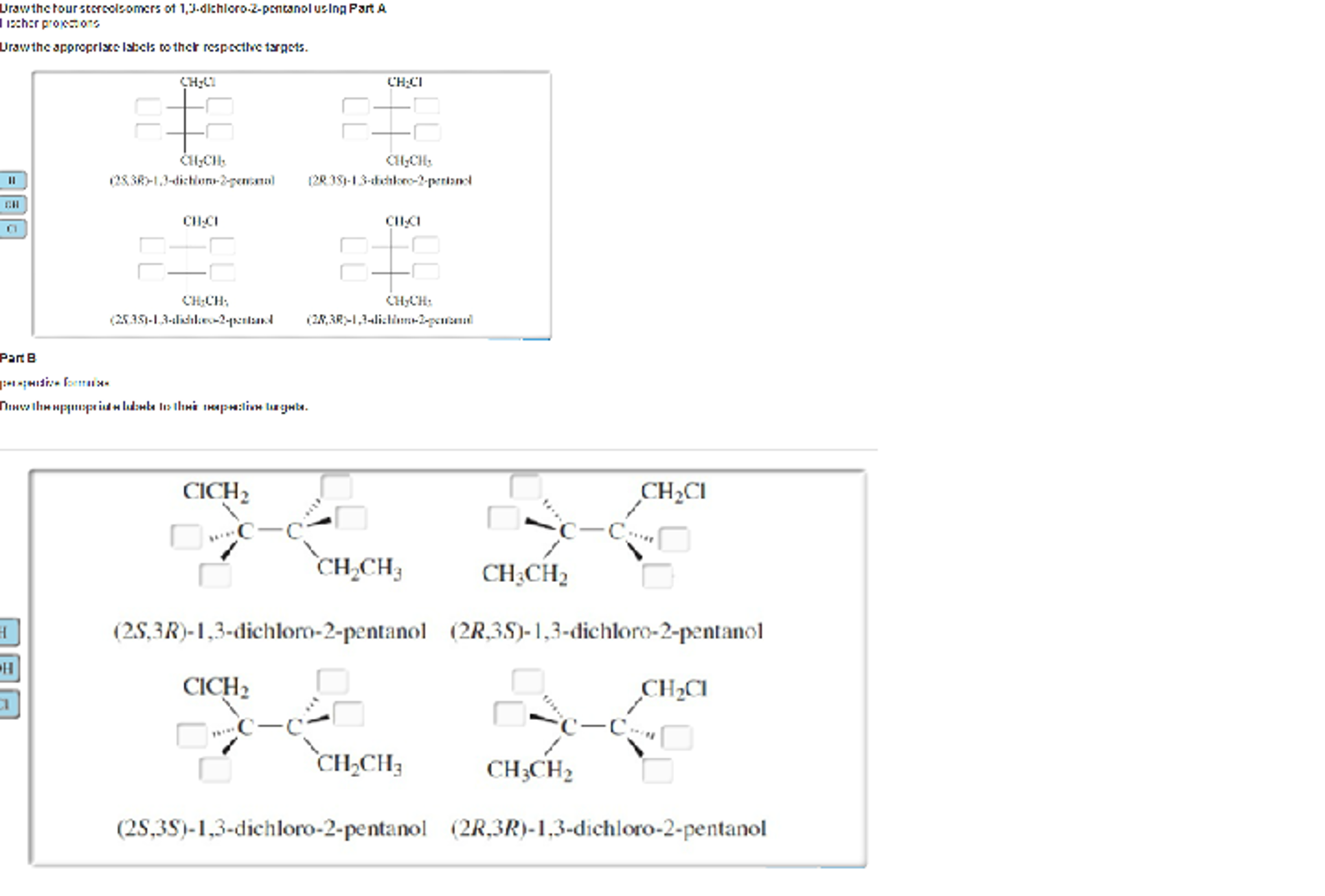
Drawing them out can be a fun little puzzle. You’d use those wedge-and-dash notations to show which atoms are coming out of the page (wedges) and which are going into the page (dashes), all within the context of that somewhat wiggly cyclohexane ring. You'd draw the cyclohexane ring, usually in its chair conformation to be more accurate, and then place your chlorine atoms. For the cis isomers, you’d have both chlorines pointing "up" relative to the ring's plane (or both "down"), and then draw their mirror images. For the trans isomer, you’d have one chlorine pointing "up" and the other "down," and realize that its mirror image is the same molecule. It’s like trying to build Lego models with the same bricks but in slightly different orientations – the final structure looks similar but isn't quite the same.
So, next time you hear "stereoisomer," don’t get all flustered. Just think of identical twins who have slightly different haircuts, or socks that are left and right. They’re related, they’re similar, but they’re not exactly the same, and sometimes, that difference is a really big deal in the grand scheme of things. Especially in the world of molecules, where even the slightest twist can change everything!
It’s all about how things are arranged in 3D space, like carefully placing your furniture in a room. You can have the same pieces, but if you arrange them differently, the whole feel of the room changes. Same with these molecules. Same atoms, same connections, just a different dance in space. And that’s the magic (and sometimes the headache!) of stereochemistry!
