Draw All Resonance Structures For The Acetate Ion Ch3coo

Hey there, coffee buddy! Grab another refill, because we're about to dive into something super cool, but, like, in a totally chill way. We're talking about the acetate ion. Ever heard of it? No worries if not, it's not exactly front-page news, but it's kinda a big deal in the chemistry world. Think of it as the quiet achiever, you know? It's that little guy that makes stuff work, and today, we're gonna uncover its little secrets by drawing out all its resonance structures. Sounds fancy, right? But honestly, it's more like a fun puzzle. Like LEGOs for chemists, but with electrons. So, let's get our thinking caps on, but keep 'em loose, okay? We’re not cramming for an exam here. This is just us, exploring some chemistry together. Ready to peek behind the curtain?
So, what even is the acetate ion? Imagine a molecule that's lost one of its tiny little electron friends. Yep, it's got a negative charge hangin' out. And it comes from, you guessed it, acetic acid. That's the stuff in vinegar, by the way! So, every time you put vinegar on your salad – and you should, it's healthy! – you're dealing with the acetate ion. Wild, huh? It’s like a celebrity ingredient in your pantry. But the coolest part about this ion? It’s not just one thing. It’s like a shape-shifter, a molecular chameleon. And that’s where our resonance structures come in. They're like different snapshots of the same molecule, trying to capture its true essence. It's a bit like trying to describe a person – you can talk about their laugh, their kindness, their quirky habits, but no single description is the whole person, right? Same with our acetate friend. It's got a few ways it likes to hang out, and we gotta show 'em all.
Let's get down to business, shall we? The formula for the acetate ion is CH₃COO⁻. See that little minus sign? That’s our clue that it’s an ion. And the CH₃ bit? That’s just a carbon with three hydrogens chilling around it. Pretty standard stuff. The real action, the drama, happens in the COO⁻ part. That’s the carboxylate group. It's where the magic, and the electron shuffling, goes down. So, we’ve got this central carbon atom bonded to two oxygen atoms. And one of those oxygens is carrying that negative charge. Or is it? That’s the million-dollar question, my friend!
First things first, let’s draw the basic skeleton. We have a carbon atom (let's call him 'Carbon Central') in the middle, and it's attached to the CH₃ group. Then, Carbon Central is also attached to two oxygen atoms. Let's label them Oxygen 1 and Oxygen 2. Now, a carbon atom usually likes to have four bonds. And those hydrogens on the CH₃ group? They're happily making their single bonds. So, Carbon Central is also making a bond to the CH₃ group. That leaves us with Carbon Central needing to bond to our two oxygens. How do we do that? Well, one of the oxygens is definitely single-bonded to Carbon Central. And this oxygen, the one that's only making one bond to Carbon Central, is the one that’s carrying the negative charge. So, it needs to have six valence electrons around it (because oxygen usually has six, and when it’s single-bonded and has a negative charge, it has an extra electron). That makes sense, right? It's feeling a bit heavy with that extra electron.
Now, the other oxygen. This oxygen is also bonded to Carbon Central. And for Carbon Central to be happy with its four bonds, this second oxygen must be involved in a double bond with Carbon Central. When you have a double bond, you’re sharing two pairs of electrons. So, this oxygen, the one in the double bond, will have four valence electrons around it (two lone pairs). It’s looking pretty content, not carrying any extra baggage. So, in this first picture, we have a single bond to Oxygen 1 (with the negative charge) and a double bond to Oxygen 2 (no charge). This is our starting point, our first guess at what the acetate ion looks like. It's a perfectly valid way for it to exist, and chemists call this a Lewis structure. It's like the blueprint, showing us where all the atoms are and how many electrons are hanging out. Pretty neat, huh?
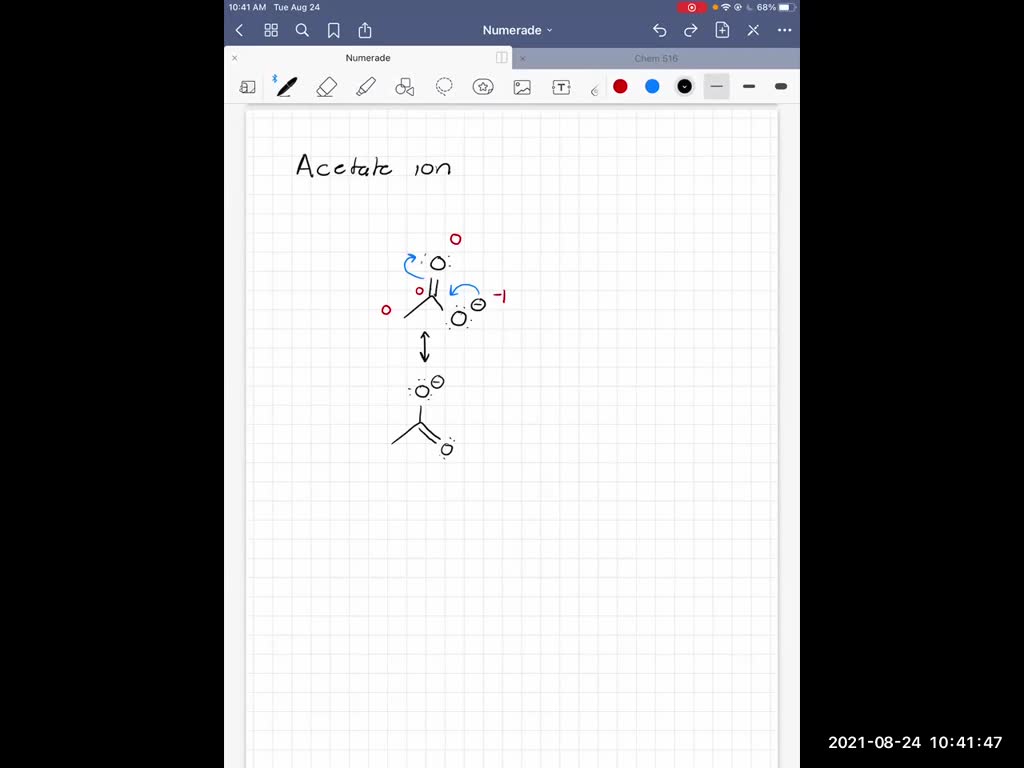
But here's where the plot thickens, and things get really interesting. Remember how I said acetate is a shape-shifter? Well, this first Lewis structure isn’t the whole story. It’s like saying a picture of a smile is the whole person. It’s part of it, but there’s more. You see, the negative charge and the double bond could be swapped around! What if the oxygen that was single-bonded and had the negative charge in our first picture actually decided to play the role of the double-bonded oxygen? And what if the oxygen that was double-bonded decided to take on the single bond and the negative charge?
So, let's draw this second possibility. We still have Carbon Central attached to the CH₃ group. And Carbon Central is still attached to two oxygens. But this time, Oxygen 1 is double-bonded to Carbon Central. This oxygen (Oxygen 1) will have its two lone pairs of electrons, totaling four valence electrons. And Oxygen 2? This oxygen is now single-bonded to Carbon Central, and this is the oxygen that’s carrying the negative charge. So, Oxygen 2 has its three lone pairs of electrons, giving it a total of six valence electrons. Tada! A completely different, yet equally valid, Lewis structure for the acetate ion.
Are you seeing it? We have two distinct drawings, two different ways to arrange the electrons, specifically the location of the double bond and the negative charge. In the first structure, Oxygen 1 is single-bonded and negative. In the second structure, Oxygen 2 is single-bonded and negative. And the double bond is with the other oxygen in each case. It's like they've swapped dance partners! And here's the kicker: the actual acetate ion isn't just the first structure, and it isn't just the second structure. It's like a blend, a hybrid of both. This is what we mean by resonance. These are our resonance structures, or sometimes called resonance contributors.

When we draw these resonance structures, we use a special arrow in between them. It’s a double-headed arrow that looks like this: ↔. This arrow doesn't mean the molecule is flipping back and forth between these two forms, like a coin being tossed. Nope. It means that the real molecule is somewhere in between these representations. It’s like saying, "These are the extreme possibilities, and the truth lies in the middle." Think of it as a fuzzy photograph versus a perfectly sharp one. The fuzzy one captures the general idea, but the sharp one shows details that might be blurred in the other. Our resonance structures are like those sharp, individual photos, and the actual acetate ion is the blurry, composite image that encompasses both.
Why is this even a thing? Why does it matter? Well, it has a huge impact on the stability of the molecule. When electrons can spread out, or be delocalized, over multiple atoms, the molecule becomes much more stable. Imagine you have a really heavy burden to carry. If you can share that burden with a friend, or even two, it's going to feel a lot lighter and easier, right? That’s what’s happening with the negative charge on the acetate ion. Instead of being stuck on one oxygen atom, that negative charge is actually shared between the two oxygen atoms. This spreading out of the negative charge makes the acetate ion a lot more stable than if the charge was localized on just one oxygen. And that’s why it loves to exist as an ion.

So, let’s recap the structure of our acetate ion, CH₃COO⁻. We’ve got our little CH₃ group, which is pretty standard. The interesting part is the COO⁻. We have a central carbon atom bonded to this CH₃ group. This central carbon is also bonded to two oxygen atoms. In our first resonance structure, one oxygen has a single bond and a negative charge, and the other oxygen has a double bond. In our second resonance structure, those roles are reversed: the first oxygen now has the double bond, and the second oxygen has the single bond and the negative charge.
The key thing to remember is that these two structures are equivalent. That means they are identical in energy and in their contribution to the overall structure of the acetate ion. It’s like having two identical twins. They look the same, they act the same, and they’re equally important. This is why the acetate ion is so stable. The negative charge is not truly on one oxygen or the other; it's spread out equally across both.
When we talk about resonance, we're essentially acknowledging that our simple Lewis structures, which show electrons in fixed positions, are often an oversimplification. Molecules, especially those with pi bonds and charges, can have electrons that are more fluid. These electrons aren't tied down to a single bond or a single atom; they can move around. This movement, this delocalization, is what resonance is all about. It's a way for molecules to achieve a lower energy state, making them more stable and often more reactive in specific ways.

So, when you see that double-headed arrow (↔) between resonance structures, don't think of it as a chemical reaction happening. Think of it as a statement about electron distribution. It's telling you that the electrons are spread out, and the actual structure is a blend of the possibilities shown. For the acetate ion, these two structures are the only valid resonance contributors because the two oxygen atoms are essentially identical in terms of their chemical environment and bonding potential.
Imagine you’re painting a picture. You have a canvas, and you want to depict a certain feeling or object. You might use different brushes, different colors, and different strokes. Each stroke is like a resonance structure – it’s part of the process of creating the final image. The final painting, though, is the complete artwork, a blend of all those individual strokes. Similarly, the acetate ion is the complete artwork, a blend of these two resonance structures. It’s the most accurate representation of how those electrons are truly arranged and how stable the ion is because of it.
It’s also worth noting that not all molecules exhibit resonance. It's a special property that arises when there are adjacent pi systems and lone pairs or charges that can be delocalized. Acetate is a classic, textbook example because it’s so clear and easy to visualize. When you're first learning about resonance, acetate is your best friend. It’s the friendly face of electron delocalization, showing you the ropes without too much confusion. So, next time you’re sipping on your vinegar-laced salad dressing, give a little nod to that awesome acetate ion. It’s doing some serious electron-shuffling behind the scenes to keep things stable and, well, delicious! Isn't chemistry just the coolest? Always something to discover, something to draw, something to… contemplate over coffee. Cheers!
