Draw A Structure For The Trans Isomer Of C2h2cl2
Ever found yourself staring at a chemical formula and wondering what on earth it actually looks like in 3D? It's a bit like trying to imagine a song from just reading the lyrics, right? Today, we're going to dip our toes into the fascinating world of molecular structures, specifically looking at a rather neat molecule called C2H2Cl2. Don't let the alphanumeric jumble scare you! It's a simple enough compound, and understanding its structure can be surprisingly fun and reveal some cool quirks about how atoms connect.
So, what's the big deal about drawing the structure of C2H2Cl2, and why should you care? Well, understanding molecular structure is fundamental to chemistry. It's the key that unlocks how molecules behave, how they interact with each other, and what properties they possess. For C2H2Cl2, understanding its structure helps us grasp the concept of isomers – molecules with the same chemical formula but different arrangements of atoms. This leads us to the specific isomer we're interested in today: the trans isomer.
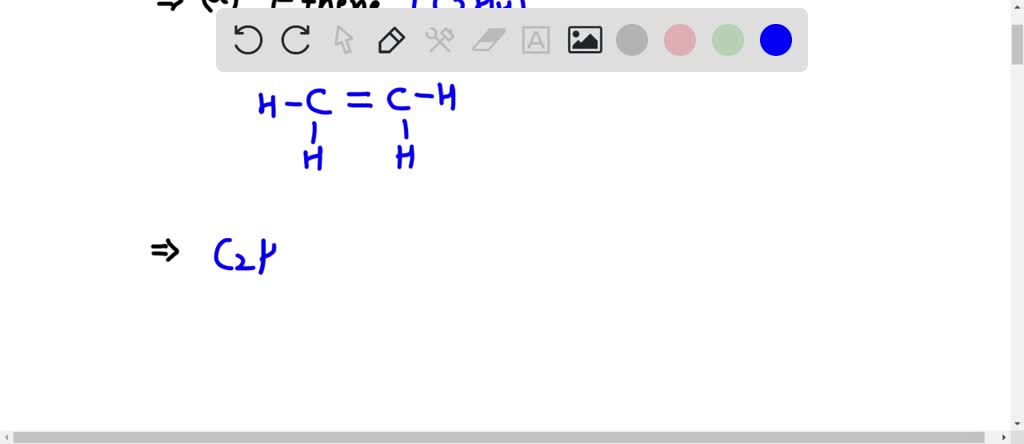
The purpose of drawing this structure is to visualize the spatial arrangement of the atoms. For C2H2Cl2, there are actually two possible arrangements for the hydrogen and chlorine atoms around the central carbon-carbon double bond. One is called the cis isomer, and the other, which we'll focus on, is the trans isomer. In the trans isomer, the two chlorine atoms are on opposite sides of the double bond, and the two hydrogen atoms are also on opposite sides. This seemingly small difference in arrangement can lead to significantly different physical and chemical properties. For instance, they might have different melting points, boiling points, or react differently with other chemicals.
You might wonder where this seemingly abstract knowledge pops up in real life. While you won't be drawing C2H2Cl2 trans isomer structures on your grocery list, the principles behind it are everywhere. In education, it's a cornerstone of chemistry courses, helping students develop critical thinking and spatial reasoning skills. Think about how medicines are designed – their effectiveness often depends on their precise 3D shape fitting into specific biological targets. This is all rooted in understanding molecular structures. Even in material science, the arrangement of atoms in polymers can dictate the strength and flexibility of plastics.
So, how can you explore this yourself? It's simpler than you think! First, let's break down C2H2Cl2. 'C2' means two carbon atoms. 'H2' means two hydrogen atoms. 'Cl2' means two chlorine atoms. The double bond between the two carbon atoms is crucial; it means they can't rotate freely. To draw the trans isomer, picture two carbon atoms bonded together with a double line. Then, attach one chlorine atom to one carbon and the other chlorine atom to the opposite side of the other carbon. Similarly, place the hydrogen atoms on the remaining opposite sides. You can even use everyday objects like colored balls and sticks to build a model – it’s a fantastic way to get a feel for the 3D shape. Searching online for "C2H2Cl2 trans isomer structure" will bring up plenty of visual examples to guide you. It’s all about seeing how atoms can arrange themselves in different, yet equally valid, ways!
