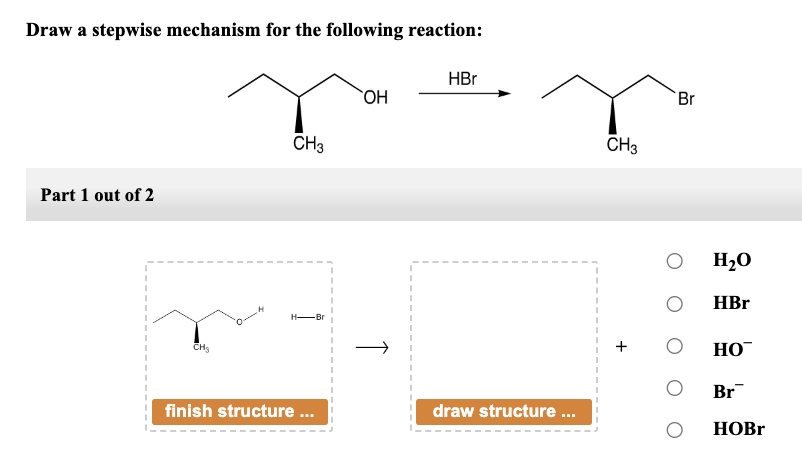
Draw A Stepwise Mechanism For The Following Reaction Hbr
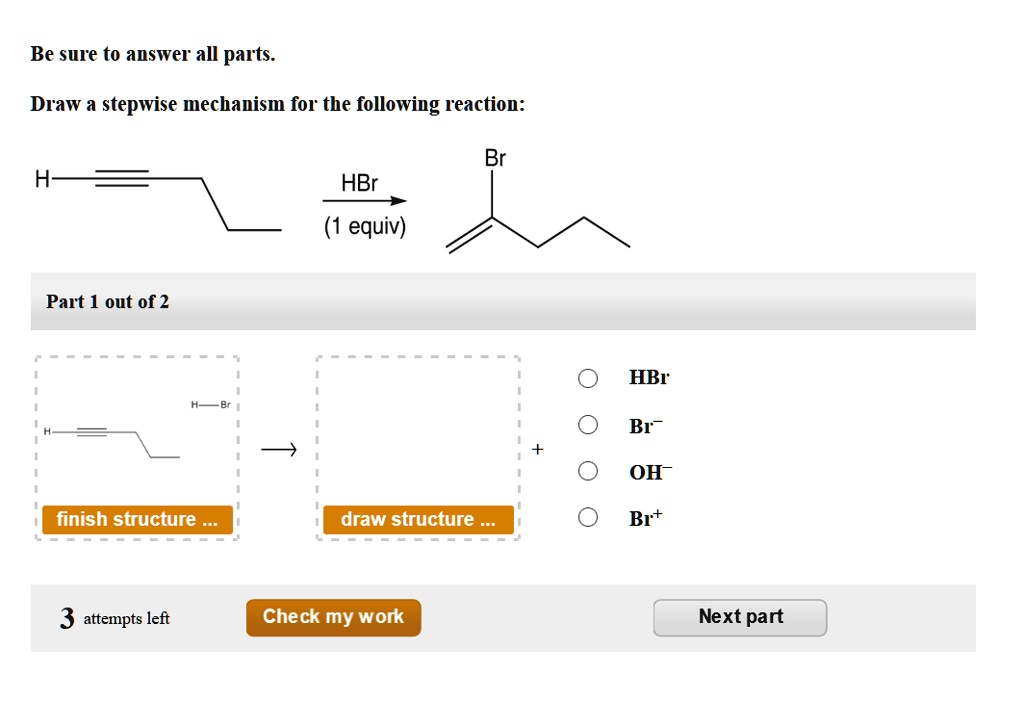
Hey there, science curious folks! Ever stared at a chemical reaction and thought, "What in the world is going on there?" I totally get it. Sometimes, those diagrams look like a secret code only chemists can crack. But guess what? Even something as seemingly complex as drawing a stepwise mechanism for a reaction with
HBr
is actually pretty darn logical, like following a recipe or understanding how a story unfolds. And believe it or not, there are reasons to care about this stuff, even if you're not planning on becoming a mad scientist!Let's imagine
HBr
as a really common, everyday ingredient. Think of it like salt and pepper for your chemical kitchen. It's a bit of a mood-setter, a little bit of a nudge, and it's involved in making all sorts of cool things happen. So, what's a "stepwise mechanism" anyway? Think of it like telling a story. Instead of just saying, "Poof! The cookies are baked," you're explaining how they got baked: first, you mixed the flour and sugar, then you added the eggs, then you put them in the oven, and so on. A reaction mechanism does the same thing for molecules – it breaks down a big change into a series of smaller, manageable steps.Now, why should you care about this? Well, understanding how reactions happen is like understanding how your car engine works. You don't need to be a mechanic to drive, but knowing a little bit can help you prevent problems or even fix a minor issue. In chemistry, knowing the "how" behind reactions helps us design new materials, develop medicines, create better fuels, and even figure out how things break down in the environment. It’s all about building, transforming, and understanding the world around us, molecule by molecule!
Let's Meet Our Main Character: HBr
So, we've got
HBr
– hydrogen bromide. It's a molecule made of one hydrogen atom and one bromine atom, linked together. Now, bromine is a bit of a show-off. It's bigger and has more electrons than hydrogen, and it's pretty good at attracting those electrons. This means the bond between hydrogen and bromine isn't perfectly shared. The electrons spend more time hanging out with the bromine. This makes the bromine end of the molecule a little bitnegative
(we call this a partial negative charge, denoted by a little δ-) and the hydrogen end a little bitpositive
(a partial positive charge, δ+).Think of it like a tug-of-war. Bromine is the stronger person, and hydrogen is the weaker one. The rope (the electrons) gets pulled more towards bromine. This makes the hydrogen atom a bit of a
"hungry"
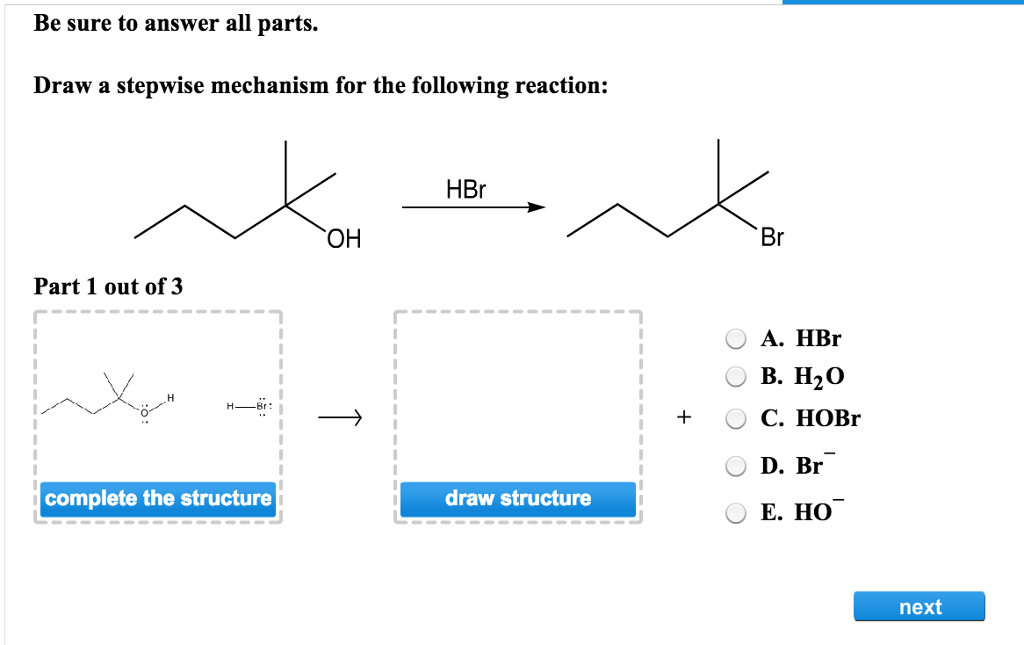
The Unveiling: What Happens When HBr Gets Involved?
Usually, when
HBr
is a reactant, it's interacting with another molecule that's a bit of an electron-lover itself. Often, this is analkene
, which is a type of organic molecule with a special double bond. This double bond is like a"party zone"
for electrons. It's rich in electron density, kind of like a chocolate fountain at a party – everyone wants to dive in!When our
HBr
molecule encounters this electron-rich double bond, the slightly positive hydrogen atom sees that electron party and thinks, "Hey, that looks like fun!" It’s attracted to the abundant electrons in the double bond.Step 1: The Electrophilic Attack – A Love at First Sight (for Electrons!)
This is where the magic begins. The double bond in the alkene, being electron-rich, acts like a
"homing beacon"
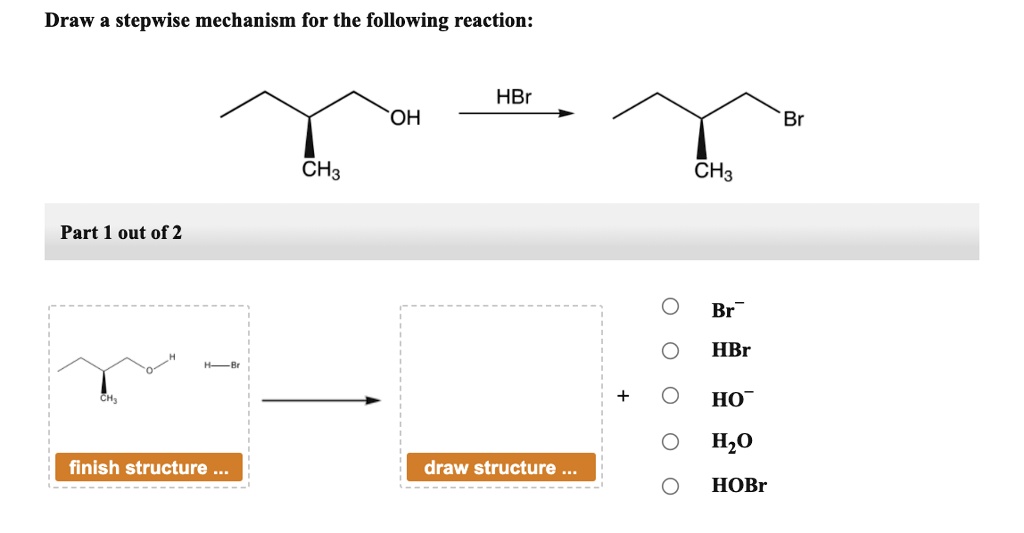
HBr
. The electrons from the double bond reach out and grab hold of that hydrogen atom. Imagine a hungry kid reaching for the last cookie – it’s an immediate attraction!When this happens, a few things change:
- The double bond between the carbon atoms in the alkene breaks. It was a double bond, now it's going to become a single bond.
- The hydrogen atom from
HBr
attaches itself to one of the carbon atoms that was part of the double bond. - Because the hydrogen left
HBr
, the bromine atom is left all by its lonesome. And guess what? It's now carrying anegative charge
(Br-). It’s like the hydrogen took its partner with it, leaving the bromine behind with a bit of an identity crisis, now a bromide ion. - The carbon atom that didn't get the hydrogen now has
"lost"
electrons from the broken double bond. This makes it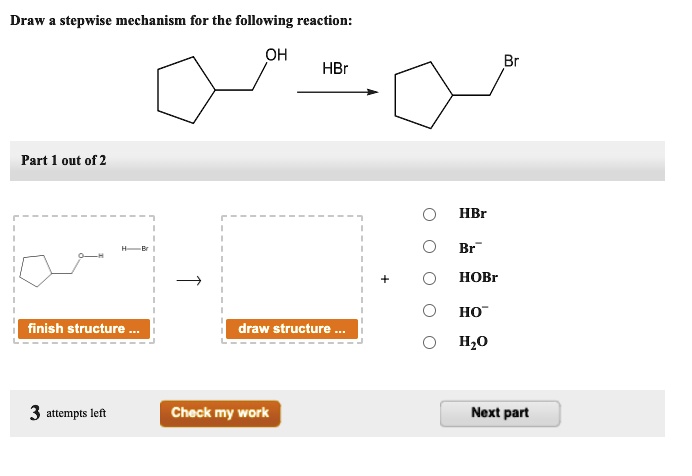 draw stepwise mechanism for the following reaction oh hbr part out of 2
draw stepwise mechanism for the following reaction oh hbr part out of 2positively charged
. We call this acarbocation
. Think of it as a carbon atom that’s feeling a bit empty and looking for some electron love.
So, in this first step, we’ve taken our alkene and
HBr
and created a carbocation and a bromide ion. It’s like a brief, intense encounter!Step 2: The Nucleophilic Attack – Finding a New Partner
Now we have our positively charged carbocation and our negatively charged bromide ion (Br-). Remember how that carbocation was feeling a bit empty and looking for electrons? Well, the bromide ion has an
excess
of electrons. It’s like a generous friend offering a helping hand.The bromide ion, with its negative charge and all those available electrons, is
attracted
to the positively charged carbocation. This is called anucleophilic attack
And voilà! A new molecule is formed. The hydrogen from
HBr
is attached to one carbon, and the bromine atom is attached to the other carbon that used to be part of the double bond. The double bond is gone, and we have a new, more stable molecule. It's like our carbocation found its perfect match and settled down.Why Does This Matter, Really?
Understanding these steps might seem like splitting hairs, but it’s incredibly important. For instance, the stability of that
carbocation
intermediate (the positively charged carbon) can actually influence which product you get if there are multiple options. It’s like choosing a shortcut on your commute – the road you take might depend on how clear the traffic is on different routes.Chemists use this knowledge to predict what will happen when they mix certain chemicals. They can design experiments to make specific products efficiently. Think about making a new plastic. You need to know how the building blocks will connect, and that’s where understanding reaction mechanisms comes in.
Even in biology, these kinds of step-by-step transformations are happening all the time inside our bodies. Our cells are constantly breaking down and building up molecules, and understanding these pathways helps us understand diseases and develop treatments.
So, the next time you see a chemical reaction, remember it’s not just a jumble of symbols. It’s a story, a series of events, and by understanding the steps, we unlock the secrets of how the world works at its most fundamental level. It’s pretty cool, right?
